Abstract
Dipyrone (metamizole) is a widely used non-opioid analgesic. Its use is restricted due to the risk of side effects such as liver toxicity. We report the case of a patient who accidentally received an oral dose of 12.5 g (132 mg/kg) dipyrone. The patient was monitored for the next 62 days and remained asymptomatic, showing no signs of gastrointestinal, hepatic, or other toxicity. Dipyrone was not detectable in the plasma, while the plasma concentrations of its active metabolites 4-methylaminoantipyrine and 4-aminoantipyrine were elevated. The pharmacokinetics were best described by nonlinear one-compartment models with Michaelis–Menten elimination. The terminal half-life of both metabolites was 2.9 h. These results demonstrate the rapid absorption of dipyrone and the extremely short window for effective gastric decontamination. Due to its specific pharmacokinetic profile, the high systemic exposure to active dipyrone metabolites did not result in acute organ toxicity in this case. Therefore, intensive medical monitoring may not always be mandatory after dipyrone overdose in otherwise clinically stable patients, although agranulocytosis remains as idiosyncratic long-term risk.
Keywords
Introduction
Dipyrone (metamizole) is widely used in pain management, particularly in Europe and South America. 1 It exhibits analgesic, antipyretic, and spasmolytic properties. 2 However, dipyrone is also associated with rare but serious adverse effects, most notably agranulocytosis.3 –6 Animal and case studies also suggest potential hepatotoxicity, primarily indicated by isolated increases in alanine aminotransferase (ALT), without corresponding changes in gamma-glutamyl transferase (γ-GT) or aspartate aminotransferase (AST). 7 While overdoses of dipyrone are typically mild and gastrointestinal, data on the effects of massive single-dose ingestion are limited. Despite frequent use, the relationship between dose, metabolite accumulation, and clinical toxicity remains insufficiently characterized in humans. Dipyrone is rapidly hydrolyzed in the gastrointestinal tract to form its active metabolites 4-methylaminoantipyrine (4-MAA) and 4-aminoantipyrine (4-AA). Therapeutic interventions such as activated charcoal are therefore likely to be effective only within the initial hours post-ingestion.1,8 This case report describes the accidental administration of 12.5 g of dipyrone. Hepatic, renal, and hematological toxicity were assessed by laboratory surveillance. Additionally, we performed a pharmacokinetic analysis based on measured plasma concentrations of dipyrone metabolites.
Case
Case history
We report the case of a 60-year-old male patient (178 cm, 95 kg, BMI 30 kg/m2) who presented with acute recurrent pain in the right leg. The time line of the case is shown in Figure 1. The patient had a known history of arterial hypertension, hyperlipidemia, and peripheral arterial disease. He presented to the vascular surgery department due to recurrent pain in his right leg. Since the onset of pain in the right leg 2 days prior, the patient had taken 400 mg ibuprofen at irregular intervals. Sonography suspected an occlusion of a P3 bypass created a year ago. Digital subtraction angiography confirmed the occlusion of the distal P3 bypass, and the patient immediately received intra-arterial lysis therapy with alteplase and heparin for the next 24 h.

Timeline of diagnostic and therapeutic interventions.
Arrival at the intensive care unit and medication error
The patient was admitted to the intensive care unit (ICU) for monitoring, laboratory tests, and pain management. The following day, angiography was repeated, and after an additional balloon angiography, restoration of blood flow in the original fem-pop bypass was confirmed. After the angiography was completed and blood flow was restored, the patient repeatedly complained about pain in his right leg. When dipyrone was administered for pain relief, a communication error occurred. The physician intended to administer dipyrone orally as a tablet with a dose of 500 mg. However, since the medication was only available in drop form at the ward at that time (one drop of 0.05 ml containing 25 mg of dipyrone) and the unit “mg” was not specified, the nurse misinterpreted the dose instruction as 500 drops. The nurse felt that counting the drops was too prone to error, so she converted the dose of 500 drops to a dose of 25 ml, and this dose of 12.5 g (132 mg/kg) of dipyrone was then administered. The error was noticed shortly afterwards, and 25 g of activated charcoal and a laxative were administered exactly 1 h after dipyrone ingestion. The error was subsequently reported to and evaluated by the internal critical incident reporting system. As a result, an automatic note was added to the digital medical record regarding dipyrone use, indicating the importance of verifying whether drops or tablets should be administered. The entire intensive care team was trained to repeat verbal instructions and to ask for clarification when in doubt.
Further course
The patient remained in the ICU and was monitored for another 48 h during which time there were no complications. He did not report feeling unwell and showed no signs of nausea, vomiting, or other gastrointestinal symptoms. Both blood pressure and heart rate remained within the normal range. The patient did not sweat and or experience pain, and was discharged to the normal ward and then sent home a few days later. We informed the patient about the details of the overdose and potential toxicity, and scheduled follow-up appointments for 14 and 62 days after exposure. He never complained of any symptoms. During the follow-up period, he did not require analgesics or other hepatotoxic medications.
Blood tests
Laboratory values, including leucocytes, neutrophilic granulocytes, ALT, AST, γ-GT, and bilirubin were closely monitored throughout the entire stay in the ICU and on the ward for 5 days after the dose (Table 1). Leukocytes and neutrophilic granulocytes were controlled again during follow-up. All cell lines were measured serially once a day as standard practice in our laboratory. The only abnormalities in the laboratory values were γ-GT, which was already elevated the day before exposure, and protein and albumin, which were slightly reduced.
Blood test results.
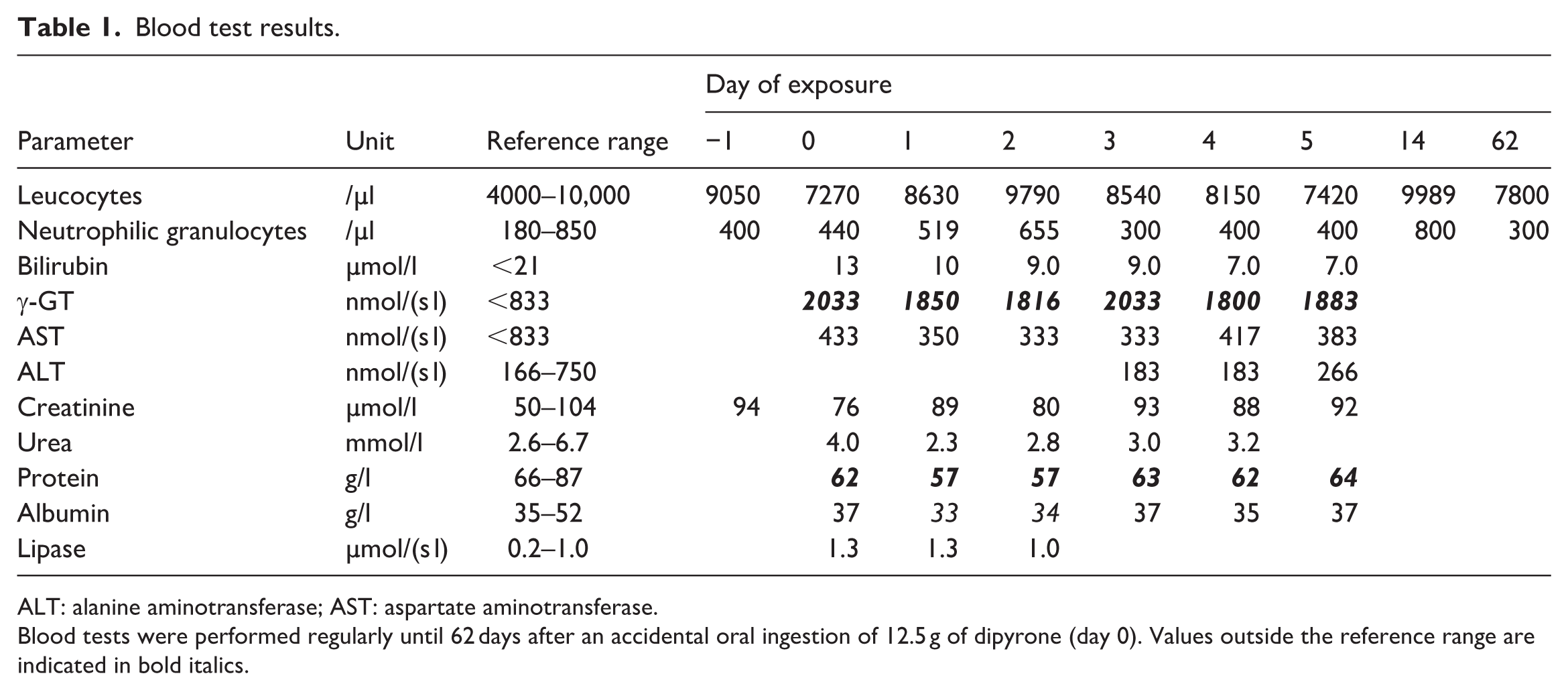
ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Blood tests were performed regularly until 62 days after an accidental oral ingestion of 12.5 g of dipyrone (day 0). Values outside the reference range are indicated in bold italics.
Plasma concentrations of dipyrone and metabolites
During the first 48 h after ingestion, we closely monitored the plasma concentrations of dipyrone and its metabolites 4-MAA and 4-AA to identify any toxic concentrations and associate them with any side effects. Seventeen venous blood samples were drawn every 2–4 h. Fifty microliters of plasma were sufficient for bioanalysis by ultra-performance liquid chromatography and tandem mass spectrometry. Deuterium labelled 4-MAA-D3 was used as internal standard. The lower limit of quantification (LLOQ) was 0.1 µg/ml for both metabolites. Intra-day and inter-assay variability were <10%. As the first sample was taken 2.5 h after application, no dipyrone was detected in the samples due to its rapid absorption and metabolization. The last concentration of 4-AA was below the LLOQ, and was excluded from the pharmacokinetic analysis.
Pharmacokinetic modeling
Pharmacokinetic modeling evaluated multi-compartment models with either linear elimination or nonlinear Michaelis–Menten elimination. Linear models were parameterized using clearance and volume of distribution. Nonlinear models were parameterized using the maximum elimination rate Vmax, the Michaelis–Menten constant KM (i.e. the concentration at which the elimination rate is half of Vmax), and the volume of distribution. The rate of hydrolysis of dipyrone to 4-MAA was fixed to 20/h, which corresponds to complete hydrolysis within 10 min. Since no plasma samples were available during the first 2.5 h, the absorption rate of 4-MAA was fixed to 3/h, which corresponds to a Tmax of 1.5 h, as reported in the literature. 9 Pharmacokinetic modeling was performed using NONMEM 7.5.0 (ICON plc, Dublin, Ireland) with the first-order estimation method, and additive and proportional residual errors. Model selection was based on the goodness of fit assessed by the Bayes Information Criterion (BIC). The time courses of the 4-MAA and 4-AA plasma concentrations were best described by one-compartment models with nonlinear Michaelis–Menten elimination (Figure 2, solid blue lines, BIC = 16.1), while models with linear elimination were inadequate (Figure 2, dashed blue lines, BIC = 78.6). Additional peripheral compartments did not improve the fit (BIC = 17.0). Vmax was 10.7 ± 0.3 mg/min (estimate ± standard error) for 4-MAA and 3.6 ± 0.2 mg/min for 4-AA, respectively. KM was 20.2 ± 1.5 µg/ml for 4-MAA and 3.5 ± 0.4 µg/ml for 4-AA, respectively. Sensitivity analysis revealed that the value of the absorption rate of 4-MAA when fixed to values between 1/h and 5/h had no significant influence on the estimates of the other pharmacokinetic parameters (coefficients of variation between 0.6% and 5.9%). The peak concentrations of both 4-MAA (81.1 µg/ml) and 4-AA (8.79 µg/ml) markedly exceeded the saturation threshold defined by the Michaelis–Menten constant KM. The terminal half-life of both metabolites was 2.9 ± 0.2 h.

Pharmacokinetic modeling of 4-MAA and 4-AA after accidental ingestion of 12.5 g dipyrone. Measured concentrations of 4-MAA (a) and 4-AA (b) are shown as black circles. The blue lines show the predicted concentrations for pharmacokinetic models with either linear elimination (dashed lines) or nonlinear elimination (solid lines).
Discussion
This case report describes a massive accidental oral overdose of dipyrone, followed by clinical observation and pharmacokinetic monitoring. Despite exceeding the typical therapeutic dose by more than 20-fold, the patient exhibited no clinical symptoms or signs of toxicity during the 62-day follow-up period. In particular, laboratory parameters indicative of hepatic, renal, and bone marrow function remained unaltered. This finding is noteworthy given other reports suggesting dipyrone-induced hepatotoxicity and the known risk of agranulocytosis.
Shortly after ingestion, the concentrations of both metabolites were significantly elevated, while dipyrone was undetectable. This pharmacokinetic profile is consistent with existing data. The parent compound undergoes rapid and nearly complete non-enzymatic hydrolysis in the gastrointestinal tract, rendering it undetectable in plasma within 1-h after ingestion. 9 The active metabolites 4-MAA and 4-AA showed a nonlinear Michaelis–Menten elimination, which has also been reported in previous studies and which is most likely caused by the saturation of the hepatic metabolic pathways at high concentrations. 9 Therapeutic ranges for 4-MAA and 4-AA have not been explicitly stated, but after repeated oral administration of a typical dose of 1 g dipyrone three times daily, plasma concentrations ranged between 1 and 10 µg/ml for 4-MAA and between 1 and 5 µg/ml for 4-AA, respectively. 9 In the first 12 h of the present case, these ranges were markedly exceeded particularly for 4-MAA, although the concentrations decreased relatively quickly (Figure 2).
In this case, almost all laboratory and vital parameters remained within the baseline values throughout the entire monitoring period. The absence of acute liver or kidney damage is noteworthy but does not contradict preclinical studies indicating hepatocellular damage, which is often characterized by elevated ALT levels. Since the baseline liver values ALT and AST are missing in this particular case, the assessment of liver function cannot be reliably evaluated in this individual case, which is why a generally valid conclusion on hepatic toxicity cannot be drawn. Additionally, no signs of agranulocytosis were observed, which confirms previous findings that this side effect is idiosyncratic and occurs independently of the dose, route of administration, or duration of treatment.1,7 One limitation in this regard could be that the observation period of 62 days was insufficient to rule out agranulocytosis. A retrospective review revealed that side effects, particularly gastrointestinal symptoms, occurred at doses of 7.5 g or higher. 4 Interestingly, no side effects were observed in our case, even at a dosage almost twice as high. Similarly, it has been reported that 81% of patients who underwent gastrointestinal decontamination within 1.5 h after a median dipyrone overdose of 10 g were asymptomatic. 4 Generally, the exact mechanisms of dipyrone toxicity are unclear, and agranulocytosis in particular seems to obey an all-or-nothing mechanism. The delayed administration of activated charcoal in the present case likely rendered it ineffective, as dipyrone is very rapidly hydrolyzed to 4-MAA (T1/2 = 2 min), and 4-MAA has a bioavailability of 89% and an absorption half-time of 15–20 min. 9 Accordingly, dipyrone was not detectable in the first blood sample at 2.5 h post-ingestion. These data further support existing recommendations that gastrointestinal decontamination is only useful within a 1-h post-ingestion. 4
Also noteworthy is the patient’s mild hypoalbuminemia, which can be explained by proinflammatory processes that had probably already begun in this clinical case. 10 However, since the plasma protein binding of 4-MAA and 4-AA is only about 60% and 50%, respectively, it is unlikely that this alteration had any influence on the pharmacokinetics. 11
One limitation of this case report is its retrospective nature. Therefore, baseline values such as ALT are missing. Regarding the pharmacokinetic analysis, the absence of early post-ingestion samples prevented the complete characterization of the absorption phase and peak exposure of 4-MAA and 4-AA. Furthermore, this is an isolated case involving a Caucasian man which limits its generalizability.
A previous study showed that out of 568 intravenous administrations, 69.7% had at least one clinical error, 25.5% of which were serious. 12 Physicians are obligated to ensure accurate dosing and clear communication in order to prevent potential harm. Reporting and discussing this incident is therefore not only consistent with professional ethics but also contributes to a culture of safety and ethical responsibility in clinical practice.
Conclusions
This case demonstrated that even a massive single oral overdose of 12.5 g of dipyrone can occur without clinical symptoms and without laboratory evidence of hepatic, renal, or hematological toxicity. In this case, oral administration of dipyrone resulted in an extremely high, albeit brief, systemic exposure to its active metabolites. The pharmacokinetic findings indicate the rapid hydrolysis of dipyrone and the nonlinear elimination of its metabolites. Administration of activated charcoal is likely useful only within a very early time window after ingestion. Based on this experience, one might hypothesize that admission to an ICU is not always mandatory in otherwise stable patients, if there are no clinical or laboratory abnormalities. Nevertheless, close monitoring and laboratory surveillance are essential and must be carried out in the normal ward. The present findings should be interpreted with caution, as this is an isolated case that cannot be generalized. Agranulocytosis remains as idiosyncratic long-term risk after dipyrone overdose. Although the reported dosing error had a relatively benign outcome, this case underscores the importance of unambiguous verbal prescriptions that clearly define dosage form and dose unit.
Footnotes
Acknowledgements
The authors thank Dr. med. Dr. rer. nat. Christian Führling, Institute of Experimental and Clinical Pharmacology and Toxicology, Friedrich–Alexander-Universität Erlangen–Nürnberg (FAU), for his support and valuable discussions.
Ethical considerations
Approval by the institutional ethical committee was waived.
Consent for publication
Written informed consent for publication was obtained from the patient.
Author contributions
AW: conceptualization, project administration, formal analysis, investigation, writing – original draft, review, editing. SS: conceptualization, project administration, formal analysis, investigation, writing – original draft. HI: conceptualization, formal analysis, pharmacokinetic modeling, writing – original draft, review. SF: conceptualization, formal analysis, writing – original draft, review. FV: formal analysis, writing – original draft, review. RCEF: writing – original draft, review, editing. IC: conceptualization, project administration, formal analysis, investigation, writing – original draft, editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge support of publication costs by Friedrich–Alexander-Universität of Erlangen–Nürnberg (FAU) within the funding program Open Access Publishing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting this study’s findings are available upon reasonable request from the corresponding author.
