Abstract
Psoriasis is an immune-mediated inflammatory disorder with a strong genetic predisposition. However, in some cases, it can emerge as an adverse effect of biologic therapy, particularly interleukin-6 inhibitors such as tocilizumab. This case report describes the unexpected onset of psoriatic dermatitis in a 16-year-old male diagnosed with polyarticular juvenile idiopathic arthritis following treatment with tocilizumab. The patient had no prior personal or family history of psoriasis, highlighting a potential paradoxical reaction to interleukin-6 inhibition. Management strategies included topical corticosteroids and vitamin D analogs, with consideration of dose adjustment or transition to an alternative biologic therapy. This case highlights the importance of recognizing dermatologic adverse effects associated with interleukin-6 inhibitors and emphasizes the need for individualized treatment strategies in juvenile idiopathic arthritis patients receiving biologic therapy.
Introduction
Juvenile idiopathic arthritis (JIA) is the most prevalent chronic inflammatory arthritis in children and adolescents, characterized by persistent joint inflammation that can lead to long-term disability. 1 Among the various subtypes, polyarticular JIA involves multiple joints and often necessitates aggressive treatment with disease-modifying antirheumatic drugs and biologic agents to achieve disease control. Tocilizumab (TCZ), a monoclonal antibody targeting the interleukin-6 (IL-6) receptor, has demonstrated significant efficacy in managing systemic and polyarticular JIA. 2 However, biologic therapies, including IL-6 and tumor necrosis factor-α (TNF-α) inhibitors, have been associated with paradoxical immune-mediated adverse effects, including psoriasiform skin eruptions.2–4
Psoriasis and psoriasiform dermatitis are well-documented side effects of TNF-α inhibitors, but their occurrence with IL-6 blockade remains rare and poorly understood. The underlying mechanism is hypothesized to involve immune dysregulation, where IL-6 inhibition may disrupt cytokine balance, leading to unopposed IL-17 activity and the emergence of psoriatic lesions.3,4 In this report, we describe a unique case of TCZ-induced psoriatic dermatitis in a JIA patient without a prior history of psoriasis, emphasizing the importance of early recognition and appropriate management of this unexpected adverse effect.
Case presentation
A 16-year-old Georgian male presented to our department in September 2021 with a 2-year history of recurrent bilateral knee pain, morning stiffness lasting ~30 min, and progressive functional limitations. He also reported intermittent fatigue but denied any systemic symptoms such as fever, weight loss, or rash. His past medical history was significant for a tonsillectomy in 2020, and there was no personal or family history of psoriasis, autoimmune diseases, or other dermatologic conditions. On physical examination, the patient exhibited bilateral knee deformities, involvement of the proximal interphalangeal joints, and restricted active and passive range of motion in affected joints. The temporomandibular joint was also involved. His skin was clear at the time of initial presentation, and no nail changes or scalp lesions were observed. Cardiac and pulmonary examinations were unremarkable, with a rhythmic heartbeat and clear lung sounds on auscultation.
A diagnostic workup, including musculoskeletal ultrasound, revealed bilateral synovial membrane thickening and joint effusion in the knees, while hand radiographs were normal. Laboratory investigations showed an antinuclear antibody titer of 1:80, a slightly elevated rheumatoid factor at 14 IU/mL (normal: 0–10 IU/mL), and an anticyclic citrullinated peptide antibody level of 20 U/mL (normal: <4.5 U/mL). A tuberculosis screening test was negative. Based on clinical, imaging, and serological findings, the patient was diagnosed with polyarticular, seronegative JIA.
The initial treatment approach included nonsteroidal anti-inflammatory drugs and intra-articular corticosteroid injections, which provided temporary relief. Methotrexate was recommended as a disease-modifying agent; however, the patient’s parents declined its use due to concerns about potential side effects. In early 2022, the patient commenced monthly intravenous TCZ at a dose of 8 mg/kg, which resulted in substantial improvement in joint mobility and morning stiffness. However, despite the beneficial effects of TCZ, the patient experienced periodic disease flares necessitating additional intra-articular corticosteroid injections for inflammatory knee effusions.
Approximately 5 months prior to this presentation, the patient developed erythematous, well-demarcated plaques with silvery-white scales on his elbows, knees, lateral tibial surface, scalp, and abdomen (Figures 1 and 2). The lesions were nonpruritic and painless but gradually increased in size. Examination confirmed features consistent with psoriatic dermatitis, which was unexpected given the absence of a personal or family history of psoriasis. A dermatology consultation was sought, and a diagnosis of TCZ-induced psoriasiform dermatitis was made based on clinical presentation and temporal association with biologic therapy. Given the unexpected development of psoriasis-like skin lesions, the treating team initiated a comprehensive reassessment of the therapeutic strategy, including discussion of potential dose modifications or transitioning to an alternative biologic therapy. During follow-up visits, the patient was prescribed topical corticosteroids (clobetasol propionate) and a vitamin D analog (calcipotriol) for symptomatic management of psoriatic plaques. Regular follow-ups were conducted every 6–8 weeks, with monitoring of joint status, inflammatory markers, and dermatologic progression. Despite topical therapy, the psoriasiform lesions persisted, prompting consideration of alternative systemic treatment options. Future management plans include either shortening the TCZ dosing interval to every 3 weeks or transitioning to an IL-17 or JAK inhibitor, depending on disease progression, treatment response, and the severity of dermatologic involvement.
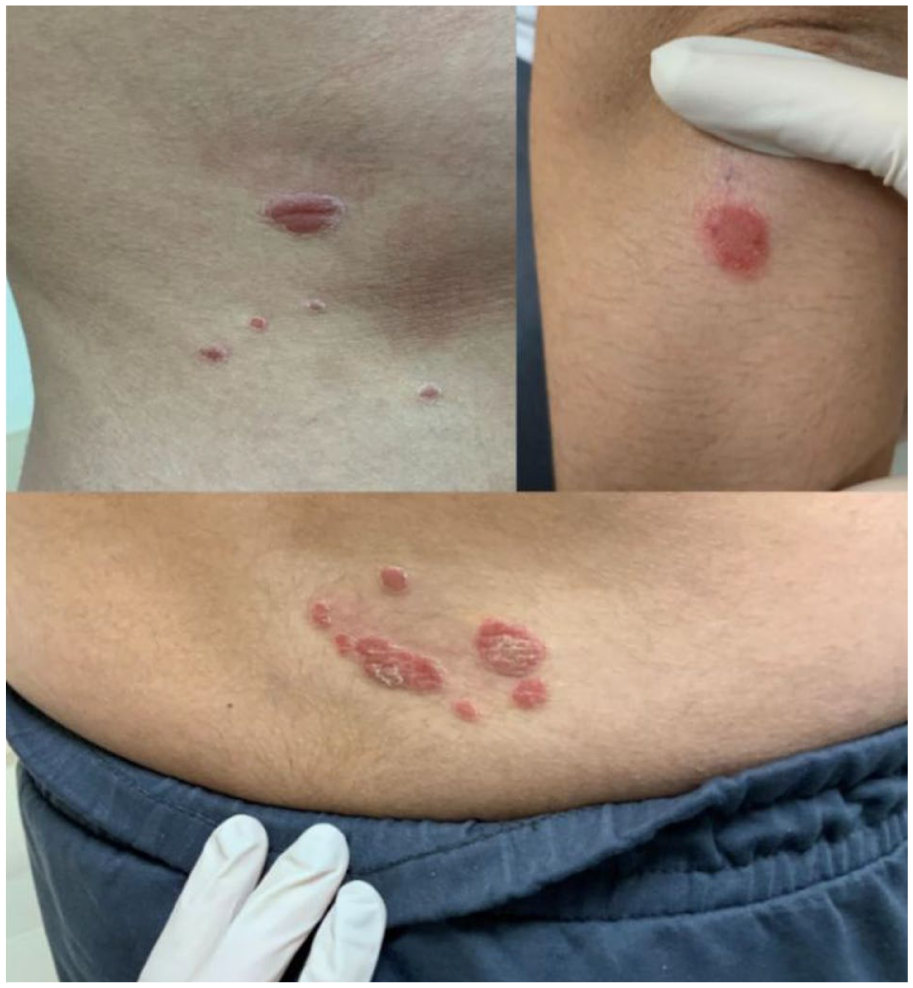
Psoriatic red skin lesions with silvery scales on the elbows and abdomen.

Psoriatic red skin lesions with silvery scales on leg (near knees) and on the lateral surface of the tibia.
Discussion
TCZ, an IL-6 receptor antagonist, exerts multiple immunomodulatory effects due to its interference with IL-6 signaling, a key pathway in chronic inflammation. Although widely used in treating JIA, TCZ has been associated with paradoxical cutaneous adverse effects, including psoriatic dermatitis, which remains a rare but clinically significant complication.5,6 This case adds to the growing, albeit limited, body of literature on TCZ-induced psoriasiform eruptions, particularly in patients without a prior history of psoriasis.
IL-6 is a pleiotropic cytokine involved in the pathogenesis of several autoimmune and inflammatory diseases. It plays a central role in promoting the differentiation of naïve CD4+ T cells into Th17 cells, which subsequently produce pro-inflammatory cytokines such as IL-17A, IL-17F, and IL-22.4,6 These cytokines are well-established mediators in the development and exacerbation of psoriasis. Thus, paradoxically, inhibition of IL-6 through TCZ may dysregulate the Th17 axis, resulting in unintended promotion of psoriasis-like lesions. 7
The precise mechanism by which TCZ induces psoriatic dermatitis remains unclear, but it is hypothesized that it involves a complex interplay between IL-6 inhibition and compensatory immune pathways. While some studies have reported increased circulating IL-6 levels following TCZ administration, likely due to receptor blockade and accumulation of the ligand, others have not observed this trend. 8 However, multiple studies have consistently shown elevated levels of soluble IL-6 receptor (sIL-6R) in patients receiving TCZ. 9 This increase may amplify IL-6 trans-signaling, which is pro-inflammatory, unlike classic signaling via membrane-bound IL-6R which tends to mediate anti-inflammatory or regenerative responses. 6 The predominance of trans-signaling may further augment Th17 differentiation and promote cutaneous inflammation characteristic of psoriasis.
Another contributing factor may be the immune shift from one cytokine axis to another. As TCZ suppresses IL-6, the immune system may compensate by increasing activity in alternative pathways, particularly the IL-23/IL-17 axis, known to be central to psoriatic pathophysiology. Increased availability of sIL-6R may also support chronic immune activation, sustaining low-grade systemic inflammation that manifests through the skin.
A literature review revealed a relevant case in which a 74-year-old male with rheumatoid arthritis developed psoriasis-like eruptions after three infusions of TCZ administered alongside methotrexate. These lesions resolved after reducing the TCZ dosing frequency, and no recurrence was noted following the switch to subcutaneous administration. 7 This suggests that modifying the mode and frequency of TCZ delivery may influence the risk of psoriatic skin reactions.
In pediatric populations, data from the BiKeR Registry 10 compared the incidence of psoriatic skin reactions among JIA patients treated with TNF inhibitors (etanercept, adalimumab), non-TNF biologics (including TCZ, abatacept, anakinra, canakinumab), and methotrexate alone. Among 4149 patients on TNF inhibitors, 676 on non-TNF biologics, and 1692 on methotrexate, 31 new-onset cases of psoriasis were recorded. The relative risk (RR) of developing psoriasis was significantly higher in patients receiving TNF monoclonal antibodies (RR 29.8, p = 0.0009) and non-TNF biologics, including TCZ (RR 25.0, p = 0.003), compared to methotrexate-treated patients. Although most studies highlight TNF blockers as primary culprits, this registry suggests that TCZ and other non-tumor necrosis factor inhibitors (TNFi) agents are also associated with elevated risk, reinforcing the need for vigilance.
For polyarticular JIA, TCZ is dosed based on body weight: patients under 30 kg receive 10 mg/kg IV every 4 weeks, while those over 30 kg, such as our patient, receive 8 mg/kg IV every 4 weeks, with a maximum dose of 800 mg. 3 Our patient was treated according to this protocol, and cutaneous symptoms emerged ~14 months into therapy, following 16 infusions.
Recent studies have further underscored the dual nature of IL-6 signaling—while classic (membrane-bound) IL-6R signaling is important for homeostatic and regenerative responses, trans-signaling via sIL-6R is largely pro-inflammatory and may be disproportionately enhanced by TCZ therapy. 6 IL-6 has also been shown to interfere with osteoblast function, mineralization, and apoptosis, which may have implications beyond the skin, including bone and joint remodeling in JIA. 5
In light of this evidence, it is plausible that the elevation in sIL-6R levels post-TCZ treatment contributes to psoriasiform skin lesions in susceptible individuals, especially in the absence of traditional risk factors such as genetic predisposition or family history. Further research, including prospective dermatologic surveillance in biologic-treated JIA cohorts, is needed to elucidate these mechanisms and guide clinical decision-making.
Conclusion
This case highlights the potential for paradoxical dermatologic reactions, such as psoriatic dermatitis, in patients treated with TCZ for JIA. Although rare, clinicians should be aware of this possibility even in the absence of a personal or family history of psoriasis. Early recognition of such adverse effects and a multidisciplinary approach involving rheumatologists and dermatologists are crucial for appropriate management. Further research is warranted to understand the underlying immunological mechanisms and to establish evidence-based guidelines for managing such cases.
Footnotes
Ethical considerations
In our university, ethics approval was not required for case reports and case series.
Consent for publication
Written informed consent was obtained from the patient for the research and the publication.
Author contributions
Rahul Yedpallikar Rajesh, Pavisankar Biju Seena, Eka Nakhutsrishvili, and Tejasri Akkineni—idea, conceptualization, resources, formal analysis, data collection, supervision, writing draft, approved final draft. Aasim Akthar Ahmed and Tarun Kumar Suvvari—visualization, resources, writing draft, and approved final draft. Tejinder Singh—supervision, formal analysis resources, writing draft and revision of draft, and approved final draft. Vimal Thomas—project administration, resources, formal analysis, writing draft and revision of draft, and approved final draft.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
