Abstract
Melanoma is an aggressive skin cancer with high mortality, particularly when not detected early. We present a case of metastatic melanoma in a patient with primary invasive melanoma and a negative sentinel lymph node biopsy who was lost to follow-up. Despite initial negative nodal involvement, the patient later developed head and neck micrometastases, resembling a “buckshot” pattern. We review current sentinel lymph node biopsy guidelines relative to tumor stage and recurrence risk. This case underscores the importance of diligent follow-up in high-risk melanoma patients, even after a negative sentinel lymph node biopsy.
Keywords
Introduction
Melanoma is a malignancy characterized by the uncontrolled proliferation of atypical melanocytes. These pigment-producing cells are located primarily in the basal epidermis, making the cutaneous form most common. However, about 5% of melanomas occur in extracutaneous sites where melanocytes are present, including ocular, mucosal, and leptomeningeal tissues and, less commonly, internal organs. 1 Melanoma is one of the most aggressive malignancies, with increasing incidence and high mortality rate. Although most patients undergo surgical excision of the primary tumor at diagnosis, many still develop metastases, the leading cause of melanoma-associated deaths. 2 We report a case of invasive primary melanoma in a patient who developed melanoma micrometastases in the head and neck within a year, despite a negative sentinel lymph node biopsy (SLNB), to emphasize the importance of early detection and ongoing monitoring on prognosis.
Case report
A 61-year-old male with Fitzpatrick skin type II presented for a total body skin exam (TBSE), due to a growing, recently bleeding lesion on his left upper arm. Examination revealed a 2.8 cm, ulcerated, dome-shaped plaque on the left upper extremity (LUE; Figure 1), and clinically atypical brown macules on the inferior thoracic spine, left lateral upper back, and superior lumbar spine. The patient denied fever, fatigue, weight changes, lymphadenopathy, arthralgias, or neurological symptoms. Family history included melanoma in his brother and breast cancer in his aunt, with no personal history of malignancy or prior TBSE. Shave biopsies were performed on the lesions on LUE and thoracic spine; the other two were monitored for evolution.
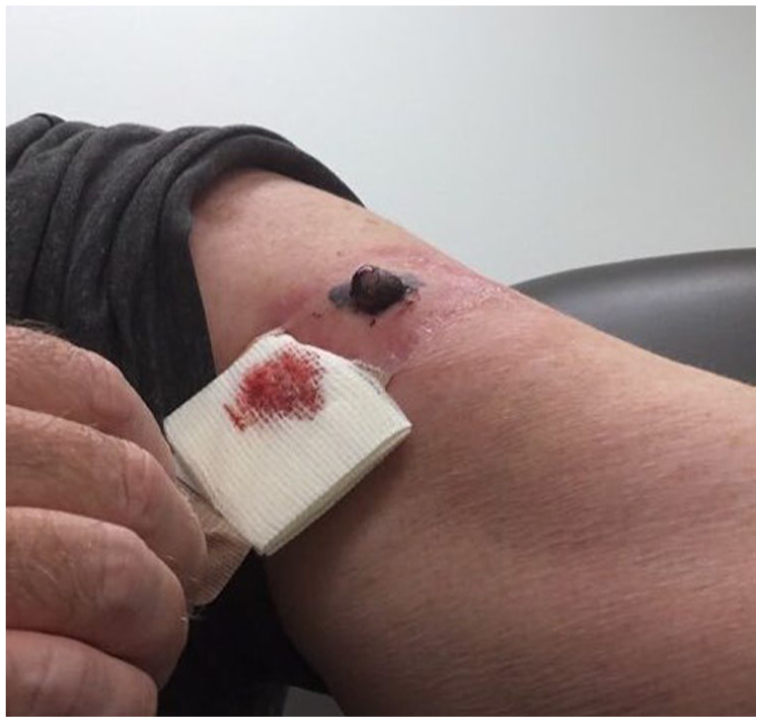
Invasive malignant melanoma on the left anterior proximal upper arm.
Histopathology of the LUE lesion revealed invasive malignant melanoma (Breslow thickness 6.4 mm, Clark level IV, pT4b staging). Melanoma in situ was present 0.8 mm from the peripheral margin, and invasive melanoma was present 1.6 mm from the deep margin. Thus, a wide local excision and SLNB were performed 20 days later. Both yielded negative results with no residual malignant melanoma and a single lymph node negative for metastatic melanoma. Immunohistochemical stains HMB-45 (human melanoma black) and Melan-A (MART-1), useful markers for melanoma diagnosis, were negative. Despite negative SLNB, the patient was referred to oncology for consideration of immunotherapy, but the patient did not follow through. He was also advised to return for TBSE every 3 months, but was lost to follow-up for 8 months.
Eight months later, the patient presented with several new 1–3 mm dark brown-to-black macules and papules on the forehead, around the mouth, and a larger 5 mm red-brown papule on the scalp (Figure 2). Also noted were two 8–9 mm dark brown-to-black macules on the back. The patient remained asymptomatic. Shave biopsies were performed on the two back lesions, with concern for primary melanoma, and two head/neck lesions, with concern for metastatic melanoma. Histopathology of the macule on the right upper back revealed superficial spreading malignant melanoma (Breslow thickness 0.7 mm, Clark Level IV, pT1a staging) with positive SOX-10 (Sry-related HMg-Box gene 10) and PRAME (preferentially expressed antigen in melanoma) stains, immunohistochemical stains of nuclear transcription factors useful for identifying metastatic melanoma. Histopathology of the papule on the left lateral mid-back lesion revealed melanoma in situ (lentigo maligna type) colonizing a nodular, infiltrative basal cell carcinoma, confirmed by positive SOX-10 staining. Biopsies of the scalp and lower lip papules showed metastatic malignant melanoma, present at the margins. These findings, along with his history, were consistent with melanoma metastases. The patient was referred again to oncology, but remained reluctant to seek care due to lack of insurance.

(a)–(c) Small macules and papules concerning for metastatic melanoma in a buckshot pattern around the mouth, face, and on the scalp, respectively.
Discussion
Most melanomas are asymptomatic at detection, with systemic signs of metastasis (weight change, dyspnea, back pain) appearing after growth progression. 3 Histopathologic examination remains critical for melanoma diagnosis and management. Key microscopic features include ulceration, mitotic activity, lymphovascular invasion, and peripheral margin status. 4 Breslow depth and Clark level are key prognostic indicators that, in addition to ulceration, aid in tumor staging. Breslow depth quantifies tumor thickness to the nearest 0.1 mm; thinner tumors (<1 mm) have a better prognosis. While Clark levels are used less often, they describe the depth of invasion, each level characterized by a deeper invasion, greater metastasis risk, and poorer outcomes. 5 Immunohistochemical stains (S-100, Melan-A, HMB-45, PRAME) aid in distinguishing melanoma from other benign pigmented lesions and malignancies. 6
SLNB guidelines
Lymph node involvement is a major prognostic indicator in melanoma. In patients with clinically-evident lymphadenopathy, evaluation with ultrasound alone is inadequate due to missing micrometastases; instead, ultrasound-guided fine needle aspiration is recommended.7,8 Yet, SLNB is still preferred. A sentinel lymph node is the first node receiving drainage from the primary tumor, making it the most likely site for early metastatic spread. SLNB, often performed with lymphoscintigraphy, provides superior staging data compared to complete nodal dissections, despite a false-negative rate of up to 21%.8,9
SLNB is typically advised for lesions >1 mm in thickness, tumor stage T2a–T4b, or lesions between 0.8 and 1.0 mm in thickness with additional high-risk features (lymphovascular invasion, ulceration, high mitoses, young age). It is not recommended for non-ulcerated lesions <0.8 mm in thickness or stage T1a, or for patients with primary melanoma and satellitosis, who are already categorized as stage III (American Joint Committee on Cancer).4,10,11 Some debate exists about whether SLNB should be performed in thin melanomas, despite low rates of SLNB-positivity, 12 though many favor it in the presence of high-risk features. 13 Decisions to perform SLNB should be individualized, weighing factors like age, comorbidities, and prior surgery in the risk-benefit discussion.
Recurrence risk with negative SLNB
A negative SLNB generally indicates a favorable prognosis due to low risk of melanoma recurrence. 14 However, recurrence still occurs in approximately 16% of SLNB-negative patients, particularly those with thicker tumors, ulceration, or high mitoses. 15 Median time to melanoma recurrence ranges from 9.3 to 35.4 months, with melanoma of the head and neck recurring earlier, indicating a worse prognosis.16,17 Notably, SLNB-negative patients with subsequent recurrence have poorer survival compared to SLNB-positive patients. 18 This supports the need for enhanced risk stratification and follow-up guidelines in select SLNB-negative patients. Patients with a prior melanoma are also at increased risk of developing subsequent primary melanomas, as noted in our patient. For instance, a 2024 Norwegian cohort study identified older men as particularly high risk for secondary primary invasive melanomas, highlighting the need for closer monitoring in the first 3 years following initial diagnosis. 19 Current guidelines recommend TBSE every 6 months for the first 2 years for patients with stages 0–IIa and every 3–6 months for the first 2 years for patients with stage IIb and higher.10,20
This case highlights the unpredictable nature of melanoma progression, especially in SLNB-negative patients. Despite early detection and a negative SLNB, the patient developed widespread head and neck micrometastases in under 1 year. This underscores the limitations of SLNB, particularly in thick melanomas or those with high-risk features, emphasizing the need for vigilant, personalized follow-up and enhanced predictive tools for effective surveillance.
Footnotes
Consent to participate
The patient provided written informed consent for the publication of this case report, including all clinical details and images.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
