Abstract
Penile metastasis from prostate adenocarcinoma is an exceptionally rare clinical finding, occurring in <0.3% of cases. We report an 85-year-old male with a history of Gleason 4+3 acinar adenocarcinoma, managed conservatively with long-term bicalutamide monotherapy because of his clinical stability. Eight years after diagnosis, he developed a painful, ulcerated glans lesion, surgically excised via glansectomy. Histopathology revealed solid adenocarcinoma with angiolymphatic invasion, high Ki-67 index (20–70%), PSA and CDX2 positivity, and negative CK7/CK20/TTF-1/p63 staining, confirming prostatic origin. Despite the presence of metastasis, disease progression remained indolent, supporting the feasibility of individualized, conservative therapy in selected elderly patients. Literature review highlights venous or lymphatic spread as probable pathways, with prognosis varying widely. This case underscores the importance of considering secondary malignancy in penile lesions, utilizing histopathology and immunohistochemistry for definitive diagnosis, and tailoring management to patient comorbidities and preferences to preserve quality of life.
Keywords
Introduction
Adenocarcinoma of the prostate is the most common malignancy in men and remains a major cause of cancer-related morbidity and mortality, despite the availability of multiple therapeutic approaches such as radical prostatectomy, radiotherapy, and androgen deprivation therapy (ADT). 1 Management is guided by the tumor’s clinical stage, risk of progression, and the overall health and preferences of the patient.
In the localized stage, when the disease is confined to the prostate, options include active surveillance, radical prostatectomy, or radiotherapy—sometimes in combination with a short course of ADT for intermediate- to high-risk disease. 2 In locally advanced diseases or in cases with high progression risk, the standard approach is radiotherapy combined with ADT, using gonadotropin-releasing hormone agonists or antagonists, often with antiandrogens such as bicalutamide to block the androgen receptor. 3
For metastatic prostate cancer, guidelines recommend combining ADT with next-generation agents—abiraterone, enzalutamide, apalutamide—or docetaxel, tailored to disease volume and patient condition.4,5 Bicalutamide monotherapy is considered suboptimal but can be used in elderly or frail patients to limit disease progression while preserving quality of life.
Prostate cancer metastasizes predominantly via lymphatic and hematogenous routes, most often to bone (65–85%), non-regional lymph nodes (pelvic and retroperitoneal), lungs (15–20%), liver, adrenal glands, and central nervous system (CNS). Metastases to the penis or testes are exceedingly rare, 6 with penile metastases having a prevalence of roughly 0.3%.7,8
Aggressive variants of prostate adenocarcinoma can metastasize to unusual sites such as the penis and are often associated with combined inactivation of tumor suppressor genes TP53, RB1, and PTEN—forming the Androgen Receptor-independent Prostate Cancer Tumor Suppressor Gene (AVPC TSG) signature—and activation of the PI3K–AKT pathway.9 –11 These molecular alterations correlate with more invasive tumor behavior, resistance to ADT, and poor prognosis.12,13 Other genomic changes include ERG–TMPRSS2 fusions, MYC amplification, and CHD1 loss, which enhance metastatic potential.9,14
Rare expression of non-prostatic markers such as CDX2—typically linked to gastrointestinal tumors—can occur in poorly differentiated prostate cancers, especially mucinous or ductal types, complicating diagnosis when metastases arise in atypical locations. 15
The routes of spread to the penis include retrograde venous dissemination via the Santorini venous plexus and dorsal penile veins, retrograde lymphatic spread through pelvic/inguinal nodes, direct extension through the urethra or periprostatic tissues, and, less commonly, arterial embolization. 16 Clinical manifestations include palpable nodules or masses, priapism, 17 penile pain, erectile dysfunction, skin ulceration, dysuria, stranguria, and hematuria.18 –20
Case report
An 85-year-old male with a history of acinar adenocarcinoma of the prostate (Gleason score 4+3) diagnosed in 2016 refused further staging, including abdominal/pelvic CT, MRI, bone scintigraphy, or nuclear medicine imaging with PSMA-PET or FDG-PET. He also declined PSA monitoring, limiting assessment of disease evolution.
In 2024, he presented with a whitish nodular mass on the penis, with an irregular surface, first noticed 4 years earlier. Over time, the lesion ulcerated and became painful. Clinical examination confirmed a glans lesion, and surgical excision was indicated. Even in this case, the patient refused to undergo any imaging and laboratory tests, only consenting to the surgical removal of the lesion. Written informed consent was obtained from the patient prior to treatment.
A glansectomy with urethral resection was performed under anesthesia. After tourniquet application, a circumferential incision was made around the coronal sulcus, the glans was dissected from the corpora cavernosa, and the urethra was preserved and mobilized for reconstruction. Hemostasis was achieved, and the urethra was advanced to create a neomeatus. The specimen was sent for histopathological examination.
Macroscopically, the resected specimen measured 3.5 × 2.7 × 1.7 cm, with a 2 × 2.5 × 1 cm whitish, compact, infiltrative mass. A separate urethral portion (1 × 0.8 × 0.7 cm) was also excised.
Microscopically, the tumor showed solid growth of atypical cells with prominent nuclei and occasional mucinous features. There was superficial ulceration, deep infiltration into the corpora cavernosa, and angiolymphatic invasion. The urethral margin was infiltrated (Figure 1). The Ki-67 proliferation index was diffusely 20% to 25%, with focal areas reaching 60% to 70%. Immunohistochemistry showed positivity for AE1/AE3, PSA, and CDX2, and negativity for CK7, CK20, TTF-1, GATA3, and p63 (Figure 2).
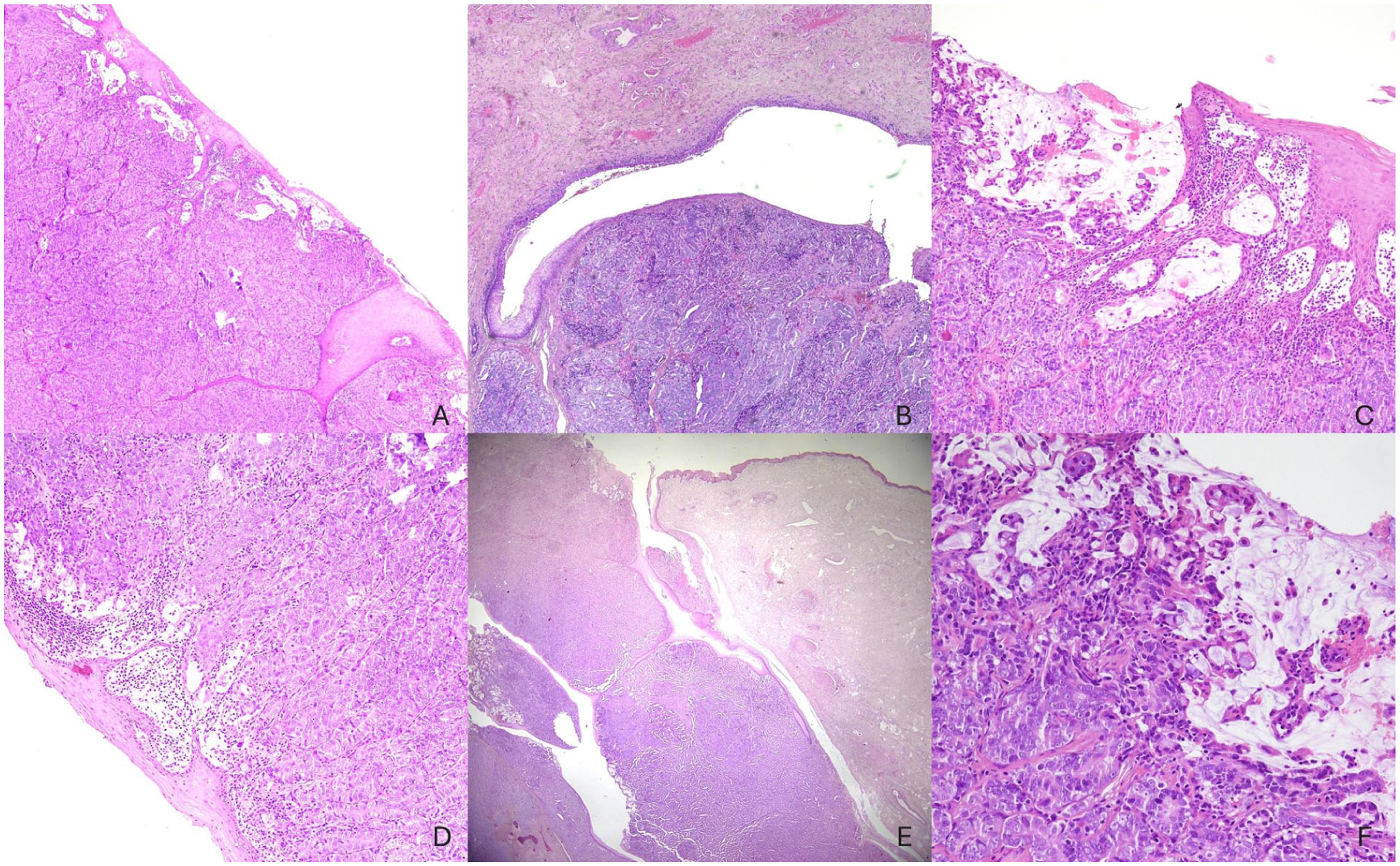
(A) Low-magnification (4×) section with deeply arranged solid neoplastic nodules infiltrating the foreskin epithelium.

(A) CK AE1/AE3: strong cytoplasmic positivity confirms the epithelial origin of the neoplasm, consistent with prostatic adenocarcinoma.
In the present case, the patient declined aggressive interventions from the outset. Hormonal therapy with bicalutamide 150 mg/day was initiated and maintained for several years with satisfactory clinical and biochemical control, even after the development of penile metastasis, indicating a relatively indolent disease course.
Discussion
Metastatic involvement of the penis from prostate adenocarcinoma is very rare (<0.3%). 21 Traditionally considered a marker of widespread disease and poor prognosis, recent literature reveals a spectrum of presentations—from indolent, isolated lesions to aggressive, symptomatic disease requiring extensive surgery.22 –25
Our patient was older than the mean age of reported cases (71 years, range 7–94). 8 Prostate cancer is the leading source of penile metastases (35%), followed by bladder (25%), colorectal (18%), kidney (7%), and lung (4%). 26 The average latency between diagnosis of primary tumor and penile metastasis is ~39 months; here, ~24 months. Lesions most often involve the glans (46%) and corpora cavernosa (39%), presenting with palpable masses, pain, hematuria, priapism, or ulceration.18,26 –28
Histologically, adenocarcinoma is the most frequent subtype (56%), typically PSA positive, as in our case. Ki-67 levels above 20% indicate higher proliferation; focal peaks of 60% to 70% suggest aggressive biology. Guerrieri et al. noted rare CDX2 immunostaining in prostate metastases, while our case showed a moderate positivity. 15
Median survival after penile metastasis diagnosis is ~22 months, yet our patient remains alive, illustrating prognostic variability. Aggressive variants with TP53, PTEN, and RB1 loss show atypical metastatic spread and poor response to ADT. Although no genomic profiling was performed, the high proliferation index and histological features suggest dedifferentiation consistent with such variants.
The exact mechanism of penile metastasis remains uncertain. Eberth first reported a case in 1870. 29 Paquin and Roland (1956) described five potential routes: 16 retrograde venous spread (most accepted), retrograde lymphatic spread, arterial embolization, direct extension, and iatrogenic implantation. In our patient, the isolated glans lesion without other detectable metastases supports a venous or lymphatic route.
Isolated penile metastases have been reported, sometimes as the first sign of recurrence. Wong et al. described an 83-year-old with a single corpus cavernosum lesion confirmed by MRI and biopsy. 30 Chau et al. reported a PSMA-PET-negative but FDG-PET-positive case treated with penectomy, achieving biochemical remission. 31 Landen et al. analyzed 72 cases, including isolated lesions with survival up to 46 months. 24 Exceptional cases show >5 years survival with localized progression. 32
Differential diagnoses include primary penile carcinoma (mainly squamous cell carcinoma) and benign conditions such as Peyronie’s disease or chronic inflammatory lesions.
Diagnosis begins with physical examination. Ultrasound helps differentiate lesion type, while MRI assesses local extent, corpora and urethral involvement. 33 PET/CT with FDG or PSMA tracers assists in detecting systemic disease, especially in low PSA states.27,34 Definitive diagnosis requires biopsy and immunohistochemistry to confirm origin. 16
Treatment is guided by disease extent, symptoms, and patient status. Most cases receive palliative ADT. 6 Our patient remained on bicalutamide monotherapy due to clinical stability and personal choice. Radiotherapy may reduce pain and tumor mass. 27 Surgery—from local excision to partial or total penectomy—can control symptoms or remove isolated metastases.34,35 Our glansectomy achieved symptom relief without significant functional compromise. Metastasis-directed therapy (MDT) is emerging, but its survival impact remains uncertain.
Prognosis is generally poor—median survival 6 to 12 months—especially in the presence of hematuria, priapism, pain, and multiple metastatic sites. Rarely, isolated lesions allow for meaningful survival extension when treated locally. 25 Our patient’s outcome supports the role of individualized, conservative therapy to preserve quality of life while maintaining oncologic control.
A key limitation of this case is the restricted follow-up period after surgical excision of the penile lesion, which limits the ability to assess long-term oncologic outcomes, recurrence patterns, or overall survival. Without extended surveillance, it remains uncertain whether the indolent clinical course observed thus far will persist or if delayed metastatic progression may occur. Additionally, the absence of post-operative imaging and biochemical monitoring (PSA) further constrains the interpretation of treatment efficacy and disease trajectory. Longer follow-up with serial clinical, biochemical, and radiologic assessment would be necessary to fully evaluate the durability of symptom control and the potential role of conservative management in similar cases.
Conclusion
This case illustrates the diagnostic importance of careful evaluation of penile lesions, as they can be the first sign of metastatic spread from advanced prostate adenocarcinoma. Histopathology and immunohistochemistry remain central for accurate diagnosis and guiding personalized management.
Even in metastatic settings, tailored conservative treatment can maintain quality of life and achieve prolonged survival.
Footnotes
Acknowledgements
The authors thank the surgical, pathology, and oncology teams involved in the care of the patient for their expertise and collaboration. The authors acknowledge support from the University of Messina through the APC initiative.
Ethical considerations
All patient data were collected anonymously and written informed consent, as part of the routine diagnosis and treatment procedures, was obtained from patients or their guardians according to the Declaration of Helsinki and the study adhered to Good Clinical Practice guidelines.
Author contributions
Conception or design of the work was done by AC, PT, and MM. Data collection was done by AC, PT, VFc, and MM. Data analysis and interpretation were done by VF and AI. Drafting the article was done by AC and PT. Critical revision of the article was done by GT, GF, VZ.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
