Abstract
Adrenal insufficiency in systemic lupus erythematosus is rarely detected, especially in male patients. Nevertheless, such coexistence can occur, and screening for systemic lupus erythematosus should be considered in primary adrenal insufficiency with symptoms of systemic multiorgan involvement. We report a 22-year-old Asian man, initially diagnosed with bicytopenia, developed severe unintentional weight loss, skin and mucosal hyperpigmentation, along with persistent fatigue. Laboratory examination showed positive antinuclear antibody—indirect immunofluorescence, elevated anti-double-stranded DNA, extremely low morning serum cortisol, and mildly elevated thyroid stimulating hormone with normal free T4. He was diagnosed with systemic lupus erythematosus, manifesting as chronic primary adrenal insufficiency, subclinical hypothyroidism, and bicytopenia. He was treated with mycophenolic acid of 180 mg b.i.d, methylprednisolone of 4 mg q.d, and vitamin D3 1000 IU q.d. Methylprednisolone was given for its anti-inflammatory property and as a simple once-daily regimen to supplement glucocorticoid deficiency. Levothyroxine was not prescribed for our patient since his thyroid stimulating hormone was only mildly elevated, and supplementation of levothyroxine in the setting of adrenal insufficiency might precipitate an adrenal crisis. At the 6-month follow-up, he was no longer fatigued, he regained his body weight, his skin and mucosal hyperpigmentation improved significantly, his thyroid stimulating hormone level normalized (without levothyroxine supplementation), and his complete blood count stabilized, remitting him from the need for transfusion.
Introduction
Systemic lupus erythematosus (SLE), widely known for its diverse and often unpredictable clinical manifestation, has long been associated with delayed diagnosis, resulting in higher morbidity and mortality as well as diminished quality of life.1–3 Recognizing adrenal involvement in SLE is challenging due to its rare occurrence as well as vague and unspecific symptoms, particularly in the setting of chronic adrenal insufficiency.4,5 Little is known regarding the characteristics of male SLE patients with adrenal insufficiency, as such coexistence has rarely been reported in literature. 6 More data are required to improve our understanding and awareness of this unusual yet potentially lethal condition. 7 We report a case of SLE in a young male patient presenting with a combination of chronic primary adrenal insufficiency, subclinical hypothyroidism, and bicytopenia.
Case report
A 22-year-old Asian man was referred to our center due to recurrent anemia and thrombocytopenia for the past 2 years, requiring transfusion of roughly two packed red cells and six thrombocyte concentrates every 4–6 months. For 6 months, he experienced unintentional 20 kg weight loss (60 to 40 kg), progressive skin darkening, and persistent fatigue. He denied a history of previous medicine consumption, including steroid and ketoconazole. On physical examination, his vital signs were within normal limits. He was normotensive with a negative orthostatic test. His body mass index (BMI) was 14.7 kg/m2. Generalized skin hyperpigmentation was observed, especially on knuckle creases. Hyperpigmentation of tongue mucosa was also observed, which the patient had not previously noticed (Figure 1). The rest of his physical examination was unremarkable. His relevant laboratory results are described in Table 1, showing a high titer of speckled antinuclear antibody-indirect immunofluorescence (ANA-IF), high level of ds-DNA, extremely low level of morning serum cortisol, mild microcytic hypochromic anemia with low reticulocyte index, mild thrombocytopenia, and mildly elevated thyroid stimulating hormone (TSH) with normal free T4. The serum adrenocorticotropic hormone (ACTH) level measurement and cosyntropin test were unavailable in our region and thus were not performed. Bone marrow analysis showed normal number of megakaryocytes, giant platelets with abnormal granulation, and nucleated red blood cells with megaloblastoid changes and intercytoplasmic bridging. Differential count of white blood cells showed myeloblast 3%, promyelocyte 5%, myelocyte 9%, metamyelocyte 14%, stab 11%, segment 23%, promonocyte 2%, monocyte 1%, and eosinophil 6% with abnormal nuclear shape, suggesting multilineage dysplasia.

Pretreatment picture showing generalized skin hyperpigmentation (a), hyperpigmentation on tongue mucosa (b), and hyperpigmentation of knuckle creases (c).
Pretreatment laboratory result.

ANA-IF: antinuclear antibody-indirect immunofluorescence; Anti-dsDNA: anti-double-stranded DNA; WBC: white blood cell; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; TIBC: total iron-binding capacity; TSH: thyroid stimulating hormone.
According to his clinical and laboratory data, he was diagnosed with SLE manifesting as chronic primary adrenal insufficiency, subclinical hypothyroidism, and bicytopenia (Table 2). He began treatment with mycophenolic acid of 180 mg b.i.d, methylprednisolone of 4 mg q.d, and vitamin D3 1000 IU q.d. After 2 months of treatment, his anemia improved, he felt less fatigued, and his body weight increased by 5 kg. After 6 months of treatment, he no longer felt fatigued, his body weight further increased by 25 kg (his final weight was 70 kg with a BMI of 25.7 kg/m2), his skin, as well as mucosal hyperpigmentation improved significantly (Figure 2), and his TSH level normalized without levothyroxine supplementation (Figure 3, Table 3).
SLE diagnostic criteria according to 2019 ACR/EULAR. 8

ACR/EULAR: The european league against rheumatism/american college of rheumatology; ANA: antinuclear antibody; SLE: systemic lupus erythematosus; anti-β2GP1: anti-beta 2 glycoprotein 1; HEp-2: human epithelial type 2.

Posttreatment picture showing significant improvement of generalized skin hyperpigmentation (a), hyperpigmentation on tongue mucosa (b), and hyperpigmentation of knuckle creases (c).

Trend of hematologic indices in relation to the patient’s treatment.
Posttreatment laboratory result.
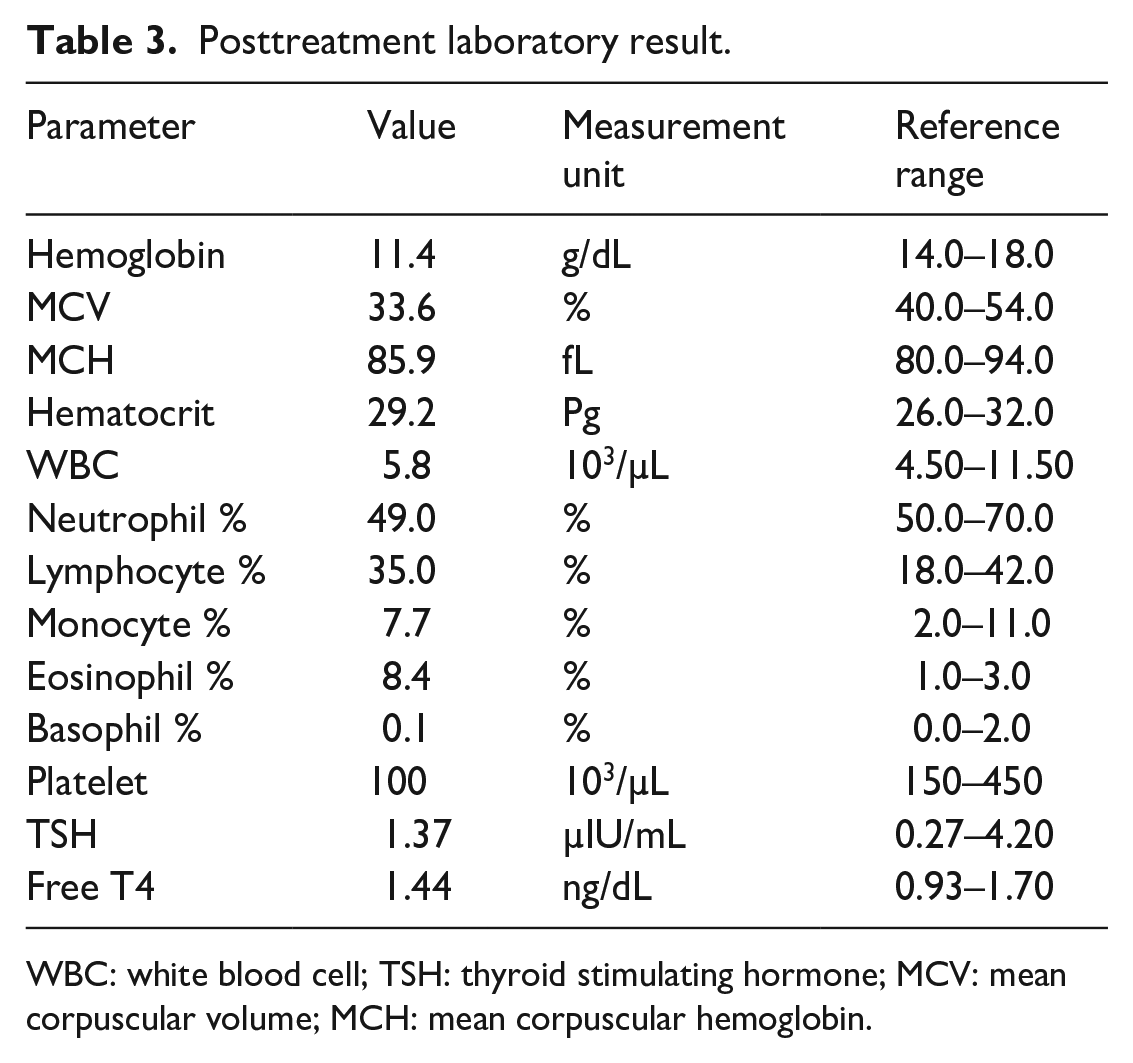
WBC: white blood cell; TSH: thyroid stimulating hormone; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin.
Discussion
The diagnosis of SLE requires evidence of multiple organ system involvement and detection of relevant immunological markers, such as ANA and anti-double-stranded DNA (anti-dsDNA). 8 Unfortunately, the initial clinical manifestation is often unclear, and considerable time might be passed before the sign of multisystem involvement becomes apparent. 1 Our patient initially experienced isolated hematologic manifestation without signs of systemic disease, which was not suggestive of systemic autoimmunity, especially in a young male patient. Roughly 1.5 years later, he began developing signs of systemic disease, such as unintentional weight loss, skin hyperpigmentation, and persistent fatigue, raising the suspicion of autoimmunity, which led to his referral to our center. As part of an autoimmune screening, ANA-IF and anti-dsDNA were examined, and both showed positive results, indicating the diagnosis of SLE according to 2019 ACR/EULAR criteria 8 (Table 2). However, he had no typical symptoms of SLE, such as arthralgia, skin rash, alopecia, and serositis. Instead, his most prominent symptoms were hyperpigmentation and severe weight loss, which were symptoms of primary adrenal insufficiency.
The diagnosis of primary adrenal insufficiency is based on the low level of morning serum cortisol, accompanied by the elevation of serum ACTH. 9 Both cosyntropin test and serum ACTH measurement were unavailable in our facility, thus were not performed. However, skin and mucosal hyperpigmentation are associated with elevated ACTH, as they are caused by the increased level of α-melanocyte-stimulating hormone, a hormone transcribed together with ACTH.10,11 Furthermore, skin and mucosal hyperpigmentation hav never been documented in secondary adrenal insufficiency. Thus, its presence strongly indicates primary adrenal insufficiency. Detection of 21-hydroxylase antibody is required to confirm autoimmunity as the etiology of primary adrenal insufficiency, which was not available in our region. Regardless, positive response toward mycophenolic acid, low-dose methylprednisolone, and strongly positive ANA-IF and anti-dsDNA suggested autoimmunity as the etiology. The aim of administering methylprednisolone was twofold. Firstly, it was for treating SLE, and secondly, as a simple once-daily regimen to supplement glucocorticoid deficiency. Hydrocortisone was not used due to its inferior anti-inflammatory profile compared to methylprednisolone. Fludrocortisone was not prescribed since it was unavailable in our country, and the patient did not show signs of mineralocorticoid deficiency.
Thyroid hormones were measured in our patient to screen for polyglandular autoimmune manifestation, showing mild subclinical hypothyroidism, which did not support polyglandular autoimmune syndrome. Elevation of TSH with normal free T4 has been documented in adrenal insufficiency, presumably due to increased pituitary stimulation by low cortisol level.12,13 TSH level usually normalizes after glucocorticoid therapy, as observed in our patient. 14 Levothyroxine was not prescribed in our patient since his TSH was only mildly elevated, and supplementation of levothyroxine in the setting of adrenal insufficiency might precipitate an adrenal crisis.15,16 We did not perform sex hormone measurements since he did not show clinical signs of hypogonadism. The patient had no signs of skeletal and growth abnormality, no gynecomastia, and he had normal hair distribution, including pubic hair.
Studies have shown that male SLE patients tend to have more severe disease and higher mortality rates than their female counterparts, albeit none have reported any association between gender and adrenal involvement.17–19 Thus, the role of gender in this coexistence is still unclear. The relative scarcity of SLE among men, combined with the rarity of adrenal involvement in SLE, might lead to a significant delay in the diagnosis of SLE in this scenario. Nevertheless, such coexistence can occur, and screening for SLE should be considered in primary adrenal insufficiency with symptoms of systemic multiorgan involvement. Adrenal involvement in SLE can be due to inflammation caused by autoimmunity or due to more specific processes such as adrenal infarction or hemorrhage in the setting of antiphospholipid syndrome (APS).6,20,21
There are several challenges in detecting adrenal involvement in systemic autoimmune disease. Firstly, adrenal insufficiency is not a component of diagnostic criteria in most systemic autoimmune diseases. Secondly, adrenal involvement can occur before, after, or simultaneously with the diagnosis of systemic autoimmune disease. Thirdly, adrenal insufficiency in this setting has a heterogenous presentation. According to a systematic review, the most common presenting clinical signs and symptoms of adrenal insufficiency in APS and SLE were abdominal pain (39.04%), followed by fever (33.33%), vomiting (23.81%), and nausea (19.05%), none of which were present in our patient. Data from this systematic review showed that adrenal insufficiency was mostly detected after or at the same time with the diagnosis of systematic autoimmunity, and only a small proportion of patients had the diagnosis of adrenal insufficiency preceding the diagnosis of APS and SLE. 22 If left undetected, adrenal insufficiency in SLE can progress to adrenal crisis, usually manifesting as life-threatening hemodynamic instability, electrolyte imbalance, and hypoglycemia. 20
Conclusion
Although a rare occurrence, chronic primary adrenal insufficiency is a possible manifestation of SLE and should be suspected in the presence of suggestive signs and symptoms, such as skin and mucosal hyperpigmentation, unintentional weight loss, and persistent fatigue. Primary adrenal insufficiency can cause elevation of TSH with normal free T4, which usually normalizes after glucocorticoid therapy.
Footnotes
Acknowledgements
The authors express gratitude to Sardjito General Hospital for providing the necessary data for this publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Consent for publication
Written informed consent was obtained from the patient for the publication of this case report and the accompanying data.
