Abstract
Patients with antiphospholipid syndrome undergoing cardiac surgery are at increased risk for thrombotic and hemorrhagic complications. We present here the management of a 68-year-old man with antiphospholipid syndrome who was scheduled for aortic valve replacement. A heparin sensitivity test was performed after anesthesia induction, and a calibration curve for blood heparin concentration and activated clotting time was created. We set a target activated clotting time of 444 s, corresponding to a heparin concentration of 3.5 U/mL. Heparin was administered at a dose of 400 U/kg, and cardiopulmonary bypass was initiated after confirming that the target activated clotting time was exceeded. After cardiopulmonary bypass weaning, 2 mg/kg of protamine was administered, resulting in a return of the activated clotting time to the pre-heparin level. Thromboelastography measurements showed citrated kaolin reaction time/citrated kaolin with heparinase reaction time values of 11.6/9.8. No additional protamine was administered, and heparin was restarted for anticoagulation management of antiphospholipid syndrome 3 h after admission to the intensive care unit. During cardiac surgery in antiphospholipid syndrome patients, evaluating coagulation status using thromboelastography combined with the heparin sensitivity test may enable safer management compared to activated clotting time-based assessment alone.
Keywords
Introduction
Antiphospholipid syndrome (APS) is an auto-immune condition, that is, characterized by the presence of antiphospholipid antibodies and recurrent episodes of arteriovenous thrombosis. In vivo, APS presents with a hypercoagulable state, although with paradoxical prolongation of the activated partial thromboplastin time (APTT) and activated clotting time (ACT). 1 Thus, cardiac surgery under cardio-pulmonary bypass (CPB) in APS patients carries an increased risk for thrombotic and hemorrhagic complications. Thromboelastography (TEG) 6s® (Haemonetics Corp., Boston, MA, USA) is a global measure of hemostasis and is useful for monitoring the coagulation status during cardiac surgery. Although there is no consensus regarding optimal anticoagulation in APS patients, 2 we considered that the combination of ACT and TEG monitoring would be useful for determining the required dose of intravenous heparin and protamine during CPB. Here, we report aortic valve replacement (AVR) in an APS patient in whom coagulation was managed perioperatively using TEG combined with ACT titration. Written informed consent was obtained from the patient for publication of this case report.
Case
A 68-year-old man was admitted to our cardiology department with a diagnosis of heart failure. Although his symptoms improved with ventilatory management and diuretics, he required AVR surgery for aortic regurgitation. He had previously undergone aortic valvuloplasty at the age of 57 years and had experienced an embolic stroke at 63 years of age. His other medical histories included APS (positive lupus anticoagulant), atrial fibrillation, hypertension, and chronic kidney disease. The APS had been diagnosed when he was hospitalized at the time of his embolic stroke, and since then, he had been taking 3.5 mg/day warfarin. Preoperative blood tests showed an APTT of 65.3 s, prothrombin time of 21.9 s, and serum fibrinogen of 628 mg/dL. Transthoracic echocardiography showed moderate to severe aortic regurgitation with perforation and deviation of the right coronary cusp.
Perioperative management in APS patients carries the risk of both thromboembolic and hemorrhagic complications. In this patient, anticoagulation management was preoperatively performed by switching from warfarin to heparin. Further, since ACT does not always accurately reflect heparinization status in APS patients, setting the ACT target to the usual 480 s during CPB might have resulted in inadequate anticoagulation. It was considered that measuring whole blood heparin concentration as an indicator during CPB would be best, but the device was unavailable in our hospital. Therefore, we decided to perform an ACT-based heparin-dose response test using the patient’s blood and calculate the target ACT value corresponding to the blood heparin concentration.
Prior to the induction of general anesthesia, an arterial line was inserted, and blood samples were collected. Blood samples with heparin concentrations of 1–4 U/mL were prepared, and ACT was measured twice using iSTAT Celite ACT® (Abbott Point of Care, Princeton, NJ, USA), and the mean value was calculated to generate a heparin-ACT titration curve. The target ACT of 444 s, which corresponds to a heparin concentration of 3.5 U/mL, was calculated.
Anesthesia was induced and maintained with remimazolam, fentanyl, remifentanil, and rocuronium. Before CPB, 400 U/kg heparin was administered, resulting in an ACT of 470 s, thus exceeding the target value. During CPB, ACT was measured every 30 min, and the target value was maintained by supplementing heparin as needed. After CPB, 2 mg/kg protamine, which is half the normal dose according to our hospital protocol, was administered, resulting in an ACT of 147 s (Figure 1).
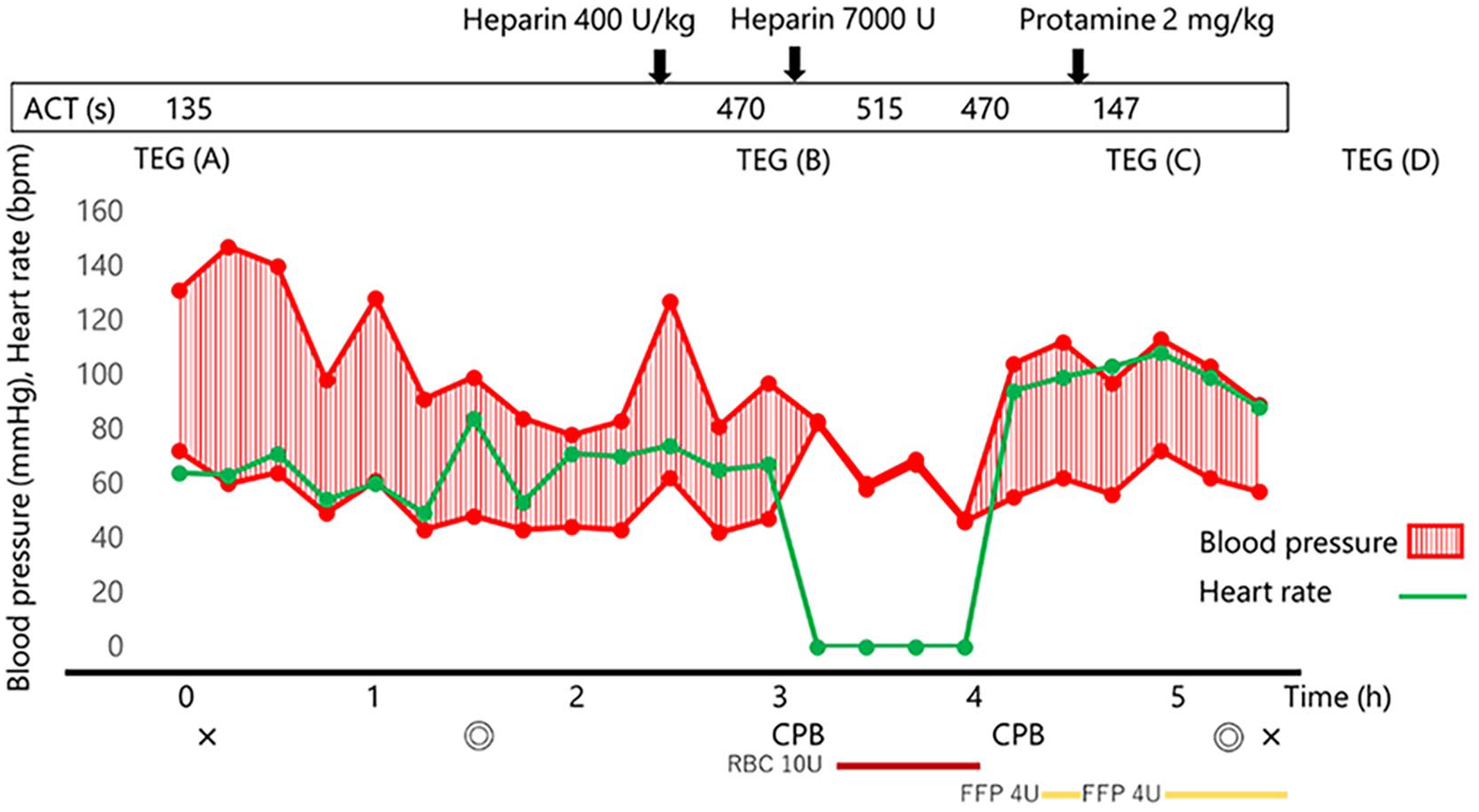
Patient’s anesthesia record and timeline of ACT.
In addition, TEG was measured at various time points, including at the time of the patient’s admission to the operating room (a), after heparin administration (b), after protamine administration (c), and 3 h after protamine administration (1 h and 20 min after admission to the intensive care unit (ICU; d); Figure 1). TEG (a) showed citrated kaolin reaction time (CK-R)/CK with heparinase reaction time (CKH-R) values of 9.2/8.7 min, indicating no obvious residual heparin after preoperative cessation of its administration, and citrated rapid TEG-maximum amplitude/citrated functional fibrinogen-maximum amplitude values of 73.7/56.3 mm, indicating a hypercoagulable state (Table 1). Following heparin administration, the anticoagulant effect of heparin was seen (TEG (b)), while after protamine administration, CK-R/CKH-R decreased to 11.6/9.8 min. Although we were concerned about residual heparin, no additional protamine was administered, considering the potential for thrombotic complications.
TEG analysis.

TEG analysis at A: time of patient’s admission to the operating room, showed hypercoagulation; at B: after heparin administration, showed inhibition of coagulation activity; at C: after protamine administration, indicated the possibility of residual heparin; and at D: 3 h after protamine administration, indicated the possibility of heparin rebound; CK-R: citrated kaolin reaction time; CKH-R: citrated kaolin with heparinase reaction time; CRT-MA: citrated rapid TEG-maximum amplitude; CFF-MA: citrated functional fibrinogen-maximum amplitude; TEG: thromboelastography.
The surgery was completed uneventfully. The total amount of fluid administered was 2545 mL, and the blood loss volume was 2307 mL. The patient received a transfusion of 10 units of red blood cell concentrate and 8 units of fresh frozen plasma. After the surgery, the patient was admitted to the ICU. At 1 h and 20 min after admission to the ICU, TEG (d) indicated heparin rebound, although there was no increase in bleeding and no additional protamine was administered. Thereafter, heparin was restarted for anticoagulation management of APS 3 h after admission to the ICU. As the hematologist suggested that the target value of APTT is 65–90 due to the high baseline APTT of this patient, we followed the advice during heparin administration. The patient was discharged from the ICU on postoperative day 2 (POD 2) after resuming warfarin, without any hemorrhagic or thrombotic complications. The patient was discharged on POD 16.
Discussion
APS is diagnosed using revised Sapporo criteria, based on the clinical presentation of thrombosis or pregnancy complications, and laboratory findings of antiphospholipid antibodies (lupus anticoagulant, anticardiolipin, or anti-β2GPI antibodies). 3 Although reports of cardiac surgery under CPB in patients with APS are limited, high morbidity rates have been reported, with a 30% incidence of bleeding or thrombotic complications. 4
In patients with APS, while hypercoagulable states are seen in vivo, in vitro tests, such as ACT and APTT, are paradoxically prolonged. Therefore, the anticoagulation management of patients with APS during cardiac surgery is difficult. Several monitoring methods, such as ACT, blood heparin concentration, blood viscoelasticity, and anti-Xa activity, have been proposed for monitoring anticoagulation during cardiac surgery in patients with APS, although all of them have various drawbacks. 5 Since ACT is often prolonged in APS patients, using it as an indicator for anticoagulation during CPB is difficult. However, some ACT instruments, such as iSTAT, which was used in this study, might be less sensitive to antiphospholipid antibodies.4,6 Yet, since patients with APS have various clinical manifestations and a range of severities, the accuracy of ACT monitoring in these patients is unclear. Blood heparin concentration is the most reliable monitor of coagulation during APS, and safe management with a target blood heparin concentration of 3.0–4.0 U/mL has been reported.5,6 However, although blood heparin concentrations can be measured using the Hepcon Hemostasis Management System Plus (Medtronic, Inc., Minneapolis, MN, USA), this device is not available at all institutions. The heparin-dose response test, which allows calculation of the ACT value corresponding to the target blood heparin concentration, is an alternative anticoagulation monitoring method. However, it should be noted that ACT is known to be prolonged during CPB due to hemodilution and hypothermia, which may result in inadequate heparin dosage when ACT is used as an indicator. Viscoelastic testing, which assesses residual heparin and the interaction between coagulation factors and platelets, is also useful for the management of coagulation after CPB. 7 In addition, although the reliability of TEG in patients with APS complications is not clear, it has also been reported to be useful.8,9 Further, although anti-Xa activity is the most reliable monitor of the anticoagulant effect of heparin, it has the disadvantage of being time-consuming to measure.
Although there is currently no recommended coagulation monitoring for patients with APS undergoing cardiac surgery, the case reports are limited. The reliability of TEG in APS patients is controversial, but it helps detect residual heparin. TEG-guided transfusion strategies have been reported to optimize transfusions in cardiac surgery, 10 but it is unclear whether they can be fully applied to APS patients. In this case, transfusions may have been excessive because we focused on the clinical findings.
Conclusion
In patients with APS, evaluating blood heparin concentration and TEG, combined with clinical findings, will likely enable safe management of cardiac surgery. In our case, the perioperative use of TEG contributed to perioperative anticoagulation management without thrombotic/hemorrhagic complications. Combined evaluation of multiple coagulation parameters might enhance the safety of anticoagulation management in APS patients undergoing cardiac surgery under CPB.
Footnotes
Acknowledgements
The authors would like to thank Forte Science Communications, Tokyo, Japan, for English language editing.
Ethical Considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report and the accompanying images.
Author Contributions
T.S. and M.H. contributed to the anesthesia management of the patient, conceptualization of the case report, and writing of the original draft. K.B. contributed to the anesthesia management of the patient. Y.I. edited the manuscript. K.O. was the overall supervisor of this case. All authors read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
