Abstract
Patients with Takayasu arteritis may present with aortic dissection; however, pericardial effusion and atrioventricular block are rare manifestations of the disease. We present the case of a 51-year-old male with a 20-year history of ulcerative colitis. Two weeks before surgery, the patient experienced fever, and computed tomography revealed pericardial effusion along with thickening of the aorta, including its cervical branches. Based on clinical presentation and imaging findings, a diagnosis of Takayasu arteritis was established. Additionally, 8 days before surgery, the patient developed a complete atrioventricular block. We performed coronary computed tomography angiography to assess potential coronary artery involvement, which incidentally revealed an ascending aortic dissection. The patient subsequently underwent ascending aortic replacement surgery. The patient subsequently underwent ascending aortic replacement surgery. Although the relationship between these findings and the development to aortic dissection remains unclear, we report two atypical complications of Takayasu arteritis that subsequently progressed to aortic dissection as a third complication. This case emphasizes the importance of identifying atypical manifestations of Takayasu arteritis, as early recognition may facilitate timely diagnosis and intervention.
Keywords
Introduction
Takayasu arteritis (TA) is a rare inflammatory disease affecting large vessels, often leading to aortic involvement. During the acute phase, inflammation can cause intimal thickening, while in the chronic phase, fibrosis leads to vessel rigidity. Although fibrosis may reduce the risk of aortic dissection (AD), patients with TA remain at higher risk for AD, particularly during the active phase. Cardiac manifestations, such as pericardial effusion and atrioventricular block (AVB), although atypical, can occur and may signal heightened vascular inflammation. This case highlights the importance of recognizing these uncommon symptoms of TA, which may precede severe complications like AD.
Case presentation
We present a 51-year-old male patient with a previous history of hypertension and ulcerative colitis dating 20 years prior to presentation who was treated with steroids and immunosuppressants. The patient previously experienced self-interruptions leading to symptom exacerbations four times. He presented with fever, and 2 weeks prior to surgery, computed tomography (CT) scan revealed pericardial effusion (Figure 1) and wall thickening from the ascending aorta to the aortic arch, involving all cervical branches. The blood examination findings were as follows: a high C-reactive protein level (23.07 (normal range: <0.14 mg/dl)) and erythrocyte sedimentation rate (>140 (normal range: 2–10 mm/h)). TA was suspected, and based on imaging findings, the disease was classified as Type I according to the Numano classification, which involves the aortic arch and its branches. Then, fluorodeoxyglucose-positron emission tomography was performed and showed high uptake in the thickened areas seen on CT (maximum standardized uptake value: 2.9), as well as in the rectum and colon (Figure 2).

Computed tomography (CT) 12 days before the surgery. CT scan revealed pericardial effusion (White arrow) (a) and wall thickening from the ascending aorta (White arrow) (b) to the aortic arch (White arrows) (c) and all cervical branches (White arrows) (c, d).

Positron emission tomography – computed tomography (PET-CT). PET-CT scan showed high uptake in the areas from the ascending aorta (a) to the aortic arch (a and b) and all cervical branches (b and c). Maximum standardized uptake value was 2.9.
Based on both the clinical symptoms (fever of ⩾38°C) and the imaging findings, including the CT scan, the patient was diagnosed with TA. He had no history of arrhythmia before TA. However, 8 days prior to surgery, the patient developed complete AVB, and a temporary pacemaker was placed. Coronary artery CT scan was performed to closely examine the coronary arteries. Ascending AD without pericardial effusion was discovered incidentally (Figure 3), and ascending aortic replacement was performed.
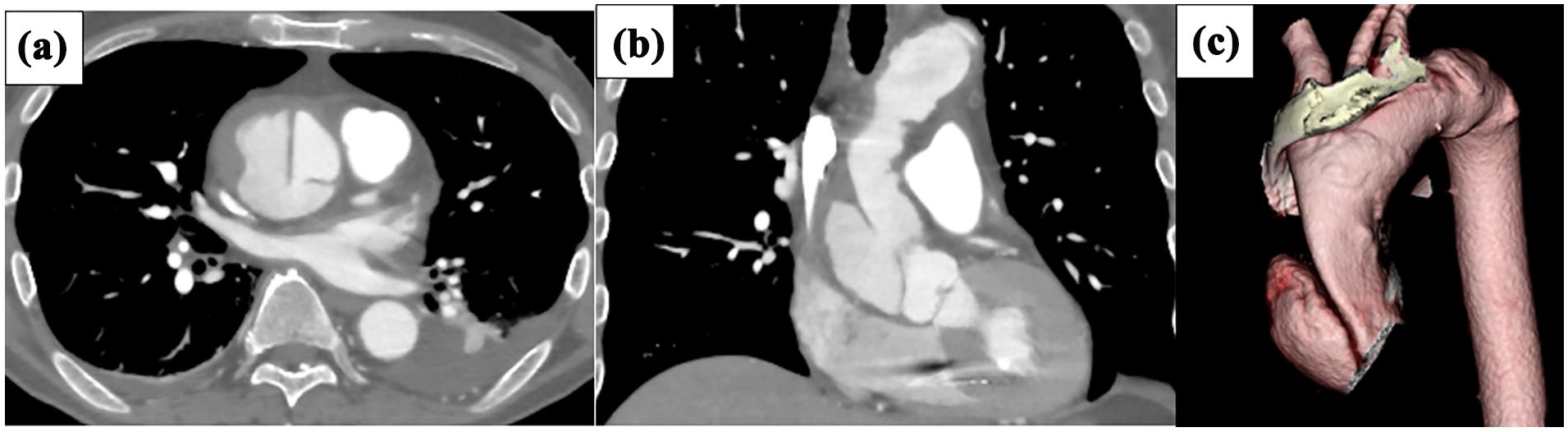
Computed tomography. CT showed intimal tear was at ascending aorta. (a) Coronal view, (b) Sagittal view, and (c) 3D view.
Via a median sternotomy, cardiopulmonary bypass was established with cannulations on the bicaval drainage and right femoral artery due to the thrombus in the ascending aorta (Figure 3). A low volume of serous pericardial effusion was observed during pericardial incision. Cold blood retrograde cardioplegia was administered every 20 min. Subsequently, the arch at zone 1 was transected with selective cerebral perfusion at minimum laryngeal and bladder temperatures of 21.3°C and 24.6°C respectively. Reconstructed the ascending aorta with a 26-mm one-branch graft (J-graft, Japan Lifeline, Japan; Figure 4). The complete cardiac arrest time for the distal body was 44 min. The total cardiopulmonary bypass and cardiac ischemic times were 168 and 111 min, respectively. After the surgery for AD, he no longer required a temporary pacemaker. On postoperative day 10, the patient was discharged without any neurological deficits or infection. After following the patient for 2 years, we found that his TA remained under control. The pathology of the resected aortic specimen showed inflammatory cell infiltration, granulation tissue formation, and elastic fiber rupture, which was consistent with TA even though steroid treatment may have suppressed inflammatory cell infiltration and cell formation.
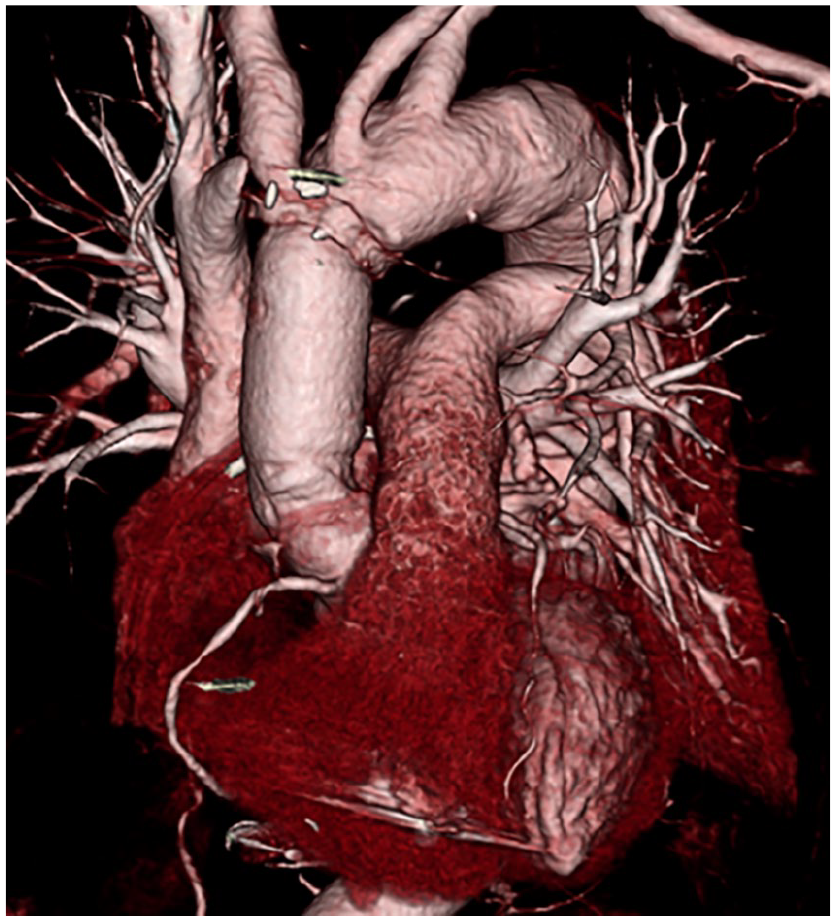
Postoperative computed tomography.
Discussion
TA affects all layers of the arterial wall. In the acute inflammatory phase, lymphocytic infiltration with giant cells in the media and fibroblast proliferation leading to intimal thickening in large vessels. In the chronic fibrotic phase, collagen replacement of elastic tissues within the vessel walls results in thickening of all vascular layers, ultimately leading to diffuse, rigid intimal fibrosis. This fibrosis may reduce the risk of AD in patients with TA. 1 However, during the acute phase, patients with TA remain susceptible to AD.1,2 The incidence of AD in TA is approximately 0.87%, 2 which is 100 times higher than in the general population (0.0077%). 3 Furthermore, the mortality rate of TA patients presenting with AD are higher than that of those without TA. 2
This case is particularly unique because pericardial effusion and AVB developed at the time of TA diagnosis, followed by the subsequent occurrence of AD. Pericardial effusion is a rare but recognized manifestation of TA. 4 Inflammation of the ascending aorta can lead to exudative effusion within the pericardial sac.5,6 Moreover, the patient did not present with typical symptoms such as chest and/or back pain but instead developed complete AVB shortly after being diagnosed with TA. It is worth noting that painless AD can occur in approximately 6% of cases, as reported in the literature,7,8 which may explain the atypical presentation in our patient. AVB also reported as a complication of TA,9,10 which subsequently improved following steroid therapy. Given that symptoms of TA are more likely to manifest during the active phase, and diagnosis often coincides with heightened vascular inflammation, both pericardial effusion and AVB, although atypical, may represent cardiac manifestations of TA. Although the association between these findings and the development to AD remains unclear, we encountered two atypical complications of TA that subsequently progressed to AD as a third complication. This case highlights the importance of recognizing atypical manifestations of TA, as they may aid in early diagnosis and prompt intervention.
Conclusions
This case emphasizes the importance of recognizing atypical cardiac manifestations of TA, such as pericardial effusion and AVB, which may precede AD. Early identification of these uncommon complications can aid in prompt diagnosis and intervention, potentially preventing further progression to more severe outcomes.
Footnotes
Ethical considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to participate
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
