Abstract
Central venous catheterization is a common practice for vascular access in different patients. Various complications associated with catheter insertion, including infection, migration, and retention, have been reported. In this report, we present an Asian (Persian) case of a 53-year-old male with a retained guidewire that protruded spontaneously from the left parietal skull 6 months after catheterization in the femoral vein. We also discuss guidewire retention’s different types, causes, and possible complications. Central venous lines are fundamental instruments that are frequently used for treatment goals; hence, loss of guidewire is a preventable complication. Preparing checklists and the attendance of two experts during the guide wire insertion procedure are two ways to reduce the loss of the guidewire.
Background
Central venous catheterization (CVC) is one of the most valuable procedures for blood transfusion, fluid, and drug administration. Complications such as infection, misplacement, arterial puncture, pneumothorax, hematoma, hemothorax, cardiac arrest due to unknown etiology, and inferior vena cava (IVC) trauma were demonstrated as adverse events following CVC.
In a systematic review following the evaluation of 76 cases to report guide wire retentions (GWR), an increased rate of GWR was found up to 2014, which was attributed to the high workload, improper education, insufficient clinical training, and lack of supervision during insertions. 1 In addition, based on the Joint Commission data, up to 2018, 73 cases were reported with GWR, which caused death in four of them. 2 Recently, some literature focusing on the complications was conducted to prevent GWR.3,4 Nevertheless, GWR occurs in 0.02% of cases (1 in 5000 cases).5–7 Here, we reported a case of GWR after cannulation of the femoral vein, which was spontaneously relieved with a rare presentation.
Case presentation
An Asian (Persian) 53-year-old male with a known case of dystrophic myopathy was referred to the emergency department with symptoms of spontaneous protrusion of a metal wire-like foreign body from the left parietal scalp. By manipulating this foreign body by the patient, the metal wire gradually began to come out of the scalp area. The wire was pulled out from the scalp area with little resistance (Figure 1). The patient had no history of craniofacial surgery or intravascular interventions. He reported femoral vein catheterization for plasmapheresis 6 months earlier, which was followed by infection of the insertion site and 2 weeks of hospitalization for antibiotic therapy but with partial treatment. He also reported a history of cervical canal stenosis surgery.
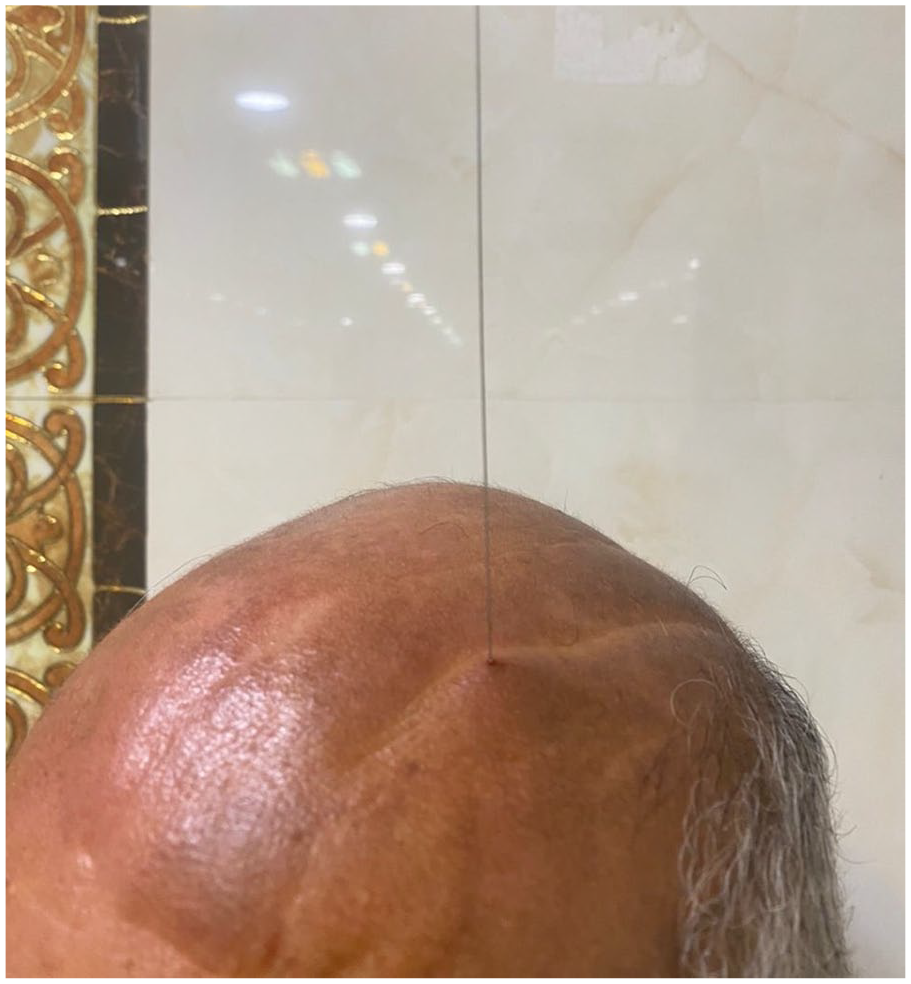
The protrusion of guidewire from the left parietal scalp of the patient.
The computed tomography (CT) scan imaging indicated a hyperdense wire-like foreign body extending from the soft tissue of the neck in the vicinity of the third cervical vertebra toward the base of the skull, entering the intracranial space right from the foramen ovale, passing through the brain parenchyma (Figure 2). It passed through the areas of the basal nuclei, migrating toward the parietal bone of the skull, and protruded from the scalp by penetrating the skull bone. There was no evidence of bleeding, hematoma, or damage to the brain parenchyma in the CT scan (Figure 3). The GWR came out of the head without any specific symptoms. After 1 h the guide wire extracted more spontaneously; under cardiovascular monitoring and critical care, the GWR with little manipulation was removed completely by a neurosurgeon. A brain CT scan after the removal of the guide wire was completely normal, and 1 day later the patient was discharged without any complications and in good condition. The patient was followed up 1 month later, and no new complications were identified. The last follow-up was 2 years later, and the patient was symptoms-free.

(a) The detected guidewire in the CT scan; (b) three-dimensional view of CT scan.

Entrance of the guidewire from foramen ovale toward intracranial space.
Discussion
Despite the progression in medical skills and growing awareness, medical errors still are reported frequently. The most common complication of catheter insertion is a-trio-ventricular arrhythmias, which mostly occur during subclavian or internal jugular vein catheterization, such as premature atrial or ventricular contractions. These arrhythmias are induced mainly by endocardium stimulation and are resolved by a few millimeters’ extraction of guidewire. 8 Intravascular GWR during CVC is an uncommon yet extremely important complication that might be completely asymptomatic and found in post-procedure routine X-ray imaging incidentally or may cause devastating insults like arrhythmias, vascular trauma, or thrombosis.4,9 According to these fatal complications resulting from intravascular GWR, retrieval of the guide wire should be done as soon as possible.
Since GWR is considered a “never event” in medical practice, it has caused increased attention from researchers. Sven-Ivar Seldinger first described Seldinger’s technique or central venous (CV) access procedures using a Guidewire in 1953. 10 A complication rate of 12% following the CV line insertion procedure was reported.11–13
Intravascular retention of fragments or parts of the catheter and possible emboli may cause severe complications and even death. It was also noted that retention of the whole guidewire may cause fewer complications than fragments or parts of the guidewire. 14
Several factors, such as high workload and operator distraction during insertion, were reported as the main risk factors for GWR. 1
We systematically searched the electronic databases to create a comprehensive and valuable report. We brought about 34 studies in which 104 patients (62 (59.6%) male‚ 42 (40.4%) female) undergoing CVC were complicated by the loss of guidewire in different sites. The most frequent insertion sites of CVCs in the literature review were as follows: internal jugular vein 16 (47%), femoral vein 12 (35.3%), and subclavian vein 7 (20.5%; Table 1).
Characterization of included studies.
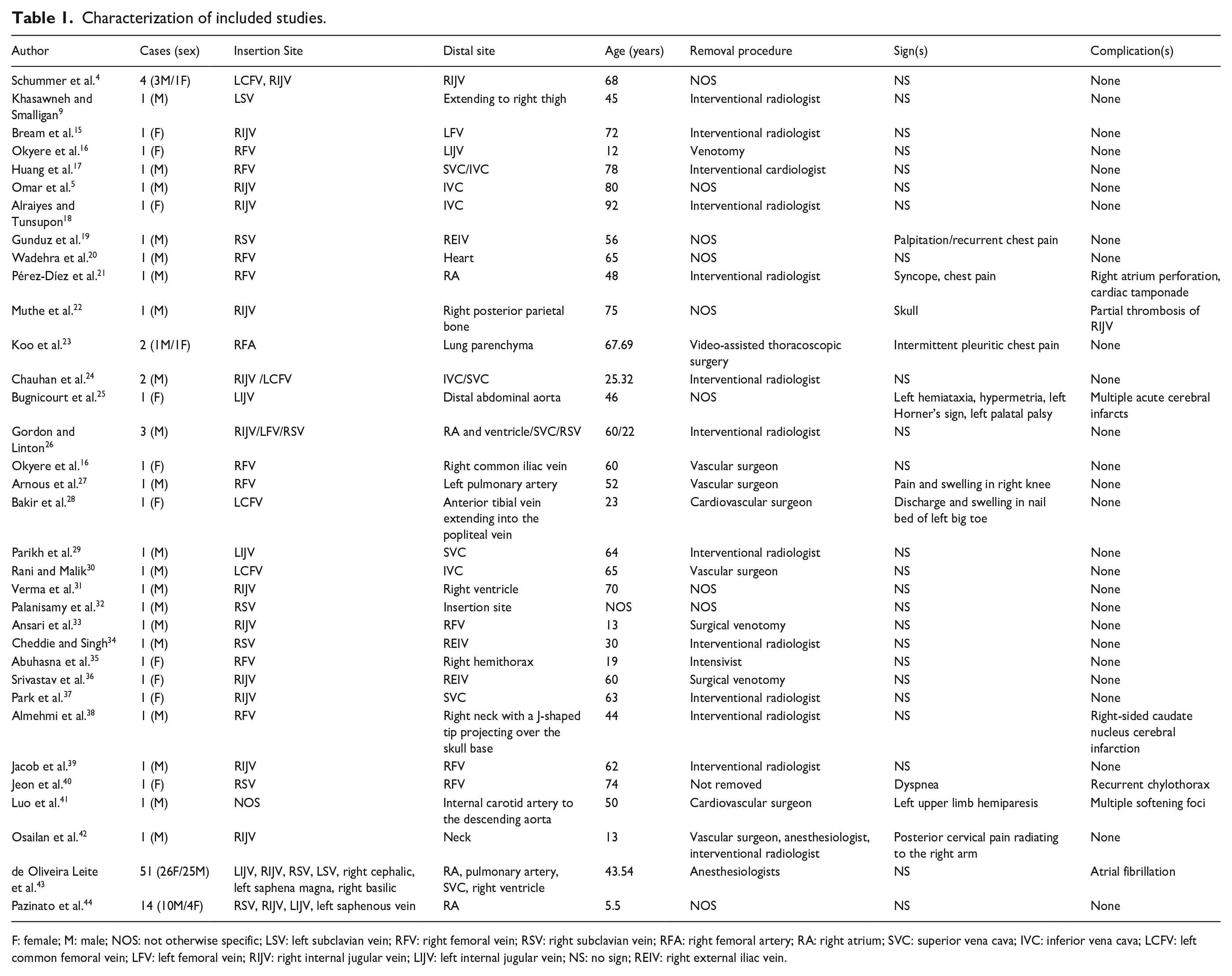
F: female; M: male; NOS: not otherwise specific; LSV: left subclavian vein; RFV: right femoral vein; RSV: right subclavian vein; RFA: right femoral artery; RA: right atrium; SVC: superior vena cava; IVC: inferior vena cava; LCFV: left common femoral vein; LFV: left femoral vein; RIJV: right internal jugular vein; LIJV: left internal jugular vein; NS: no sign; REIV: right external iliac vein.
These studies reported different sites of guidewire entanglement, which we call a distal site. Twenty-nine studies reported intravascular loss of guidewire and entanglement in large vessels, such as internal jugular vein (5 (14.7%)), superior vena cava (6 (17.6%)), IVC (4 (11.7%)), femoral vein (4 (11.7%)), external (3 (8.8%)), common (1 (2.9%)), and internal (1 (2.9%)) iliac vein, distal abdominal aorta (1 (2.9%)), subclavian vein (1 (2.9%)), pulmonary artery (1 (2.9%)), and also popliteal vein (1 (2.9%); Table 1). Six (17.6%) of them reported the heart (atrium or ventricle) as the distal site, 3 (8.8%) of them reported lung parenchyma and hemithorax as the distal site.
Of 104 retained guidewires, 51 (49%) were removed by anesthesiologists, 17 (16.3%) were removed by surgeons, 15 (14.4%) were removed by interventional radiologists/cardiologists, 5 (4.8%) were removed after exploration of the insertion site, 2 (1.9%) with video-assisted thoracoscopic surgery from lung parenchyma, and 1 (1.9%) was not removed (Table 1).
Similar to our case, a case was reported with spontaneous guidewire protrusion after 3 years of retention in India. 22
Inadequate precision on postprocedural radiographs is one of the most frequent reasons for GWR misidentification. Postprocedural radiographs should be conducted routinely following CVC. However, considerable reports of GWR were not detected on postprocedural radiographs. 45
A considerable diversity in retention patterns makes it impossible to have a treatment protocol following GWR. The time of identification is the first challenge. In the majority of previous literature, GWR was diagnosed immediately. 2 However, it cannot be considered with certainty as a “never-ever event.” Migration or remaining of the guidewire is the second challenge. Migration was more commonly reported and followed by cases that remained near the insertion site. 2 In our case, the guidewire protruded from the skull, whereas migration to the chest was more prevalent.16,17,46–48 The third challenge is the choice of the treatment intervention.
There is controversy among specialists about the treatment strategy for GWR. Studies suggest interventional approaches for guidewire retrieval should be conducted first before surgery, and not surgical approaches unless interventional techniques fail.
In cases in which guidewire retrieval could not be done successfully, long-term administration of anticoagulants in combination with prophylactic antibiotic therapy is suggested. However, the exact duration of anticoagulant administration has not been declared. 34
In patients with palpable and superficial guidewire retention (in jugular or femoral veins), performing direct venotomy for guidewire removal is reasonable. Although in centers with professional interventional equipment such as Dormia basket, gooseneck snare, and endovascular forceps, percutaneous retrieval of the guidewire is preferred. 49
In China, Zhao et al. identified a patient with a broken guidewire 1.5 months after discharge of the patient; ~1 year after surgery, the patient was referred with a swollen ankle and headache; the CT scan findings revealed a broken guidewire. 50 In another case, the physicians decided not to remove the guidewire due to signs of sufficient flow throughout the vessels, such as palpable pulse and absence of stenosis evidence. 6 In another case, the prophylactic treatment strategy was conducted, including antiplatelet therapy and follow-up with periodic Duplex studies. 6
Studies mostly reported no signs and symptoms and also no or minor complications like partial vascular thrombosis, in which guidewire was lost in large vessels, but indeed, infrequent yet catastrophic complications have been reported, including guidewire entanglement in the atrium or ventricle, causing cardiac arrhythmia, thromboembolism, chest pain, palpitation, and also such fatal condition, like atrial perforation and cardiac tamponade.
Another extremely infrequent complication was a cerebrovascular accident because of guidewire migration and retention in the brain vascular system, or brain parenchyma. For example, Bugnicourt et al. reported a 46-year-old female presenting with left hemiataxia, Horner’s syndrome in neurological examination, and then, after further investigation and clinical imaging workup, was found to have multiple acute cerebral infarcts in the vertebrobasilar territory. 25
To our knowledge, there are at least three reported studies in the literature of cerebrovascular complications related to GWR itself. First was a 50-year-old male with left upper limb Paresis due to cerebral infarction in the left frontal lobe, right parietal lobe, and right cerebellar hemisphere that was due to guidewire entanglement in the aortic arch and intra-aortic foreign body formation, and resulted in cerebral ischemia and multiple foci infarcts.
The second was a 46-year-old female who underwent left internal jugular catheterization and presented with difficulty in swallowing neurological examination revealed left hemiataxia, hypermetria, left Horner’s sign, and right deviation of the uvula, indicating left palatal palsy and hypoesthesia of the left hemi-face.
Further imaging studies revealed multiple acute cerebral infarcts in the vertebrobasilar territory and posterior cerebral infarction. They concluded that loss of the guidewire resulted in delayed vertebral artery thrombosis and caused the mentioned complications.
The third one was a 44-year-old male who underwent right femoral vein catheterization for emergent dialysis. In this case, the retained guidewire resulted in right-sided caudate nucleus cerebral infarction revealed after brain imaging.
In one study, the retained and lost guidewires entered the cranium and brain parenchyma. Muthe et al. reported a 75-year-old male who had a known case of hypertension, diabetes mellitus, and alcohol addiction and had a history of hemodialysis due to acute kidney injury and uremic encephalopathy 3 years earlier. 22 He was presented with a metallic thin wire protruding out of his skull. Further imaging studies revealed a linear hyperdense foreign body extending from the distal part of the right internal jugular vein, entering the cranium, coursing through brain parenchyma (right posterior parieto-occipital lobe), and exiting the skull by penetrating the right parietal bone. Partial thrombosis of the internal jugular vein was strangely the only reported complication.
In cases with spontaneous guidewire exclusion, the lower or even absence of complications is expected. In cases with diagnosed GWR in critical parts of the body, the removal must be conducted cautiously. There is a report of unsuccessful removal of the guidewire, which led to hemorrhagic shock. 6 In this line, Abe et al. introduced a novel technique entitled the “jailed-wire technique” for safely removing the guidewire. 51 In this technique, an inflated balloon was used for guidewire exclusion. 52
Finally, to prevent this complication, some precautions should be taken during the procedure.
The guidewire should be visible at the proximal end before catheter advancement.
Hold onto the proximal of the guidewire during the intravenous advancement of the catheter and the whole insertion procedure until the complete removal of the guidewire is achieved.
Avoiding simultaneous advancement of guidewire and catheter.
Conclusion
CV lines are fundamental instruments that are frequently used for treatment goals; hence, loss of guidewire is a preventable complication. Preparing checklists and the attendance of two experts during the guidewire insertion procedure are two ways to reduce the loss of the guidewire. Also, the postprocedural radiography should be interpreted casually to rule out GWR.
Footnotes
Acknowledgements
We would also like to show our gratitude to the Clinical Research Development Unit, Golestan Hospital, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran for sharing their pearls of wisdom with us during this research. We thank all our colleagues at Allied Health Sciences School, Ahvaz Jundishapur University of Medical Sciences.
Ethical considerations
Ethical approval to report this case was obtained from the ethical committee of Ahvaz Jundishapur University of Medical Sciences (IR.AJUMS.HGOLESTAN.REC.1402.173).
Consent to participate
Written informed consent was obtained from the patient to participate in this article.
Consent for publication
Written informed consent was obtained from the patient to publish this case report and any accompanying images. A copy of the written permission is available for review by the Editor-in-Chief of this journal.
Author contributions
M.B. designed the study, analyzed the original survey data, and created the first draft manuscript. S.R. and H.S. reviewed the data interpretation from the original studies and contributed to manuscript revisions.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare no conflict of interest. All procedures performed in studies involving human participants were by the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or compared ethical strands.
Data availability statement
The datasets generated and/or analyzed during the current study are available in the PubMed, Web of Science, Scopus, EM Base repository.
