Abstract
Systemic lupus erythematosus predominantly affects women of all ages, particularly targeting those of reproductive age, and can increase the risk of adverse pregnancy outcomes, including uterine atony, which is rarely documented. This case report examines uterine atony in a systemic lupus erythematosus patient and its microscopic features. A 37-year-old woman with systemic lupus erythematosus, in her fourth pregnancy, experienced vaginal bleeding, and severe hypotension after delivering at 32–33 weeks. Despite treatments including fluids, oxytocin, misoprostol, and methylergometrine, uterine contractions did not improve, leading to a supravaginal hysterectomy. Histopathology revealed hypertrophic myocytes and extensive fibrosis. This case suggests that systemic lupus erythematosus may increase the risk of uterine atony and postpartum hemorrhage due to uterine fibrosis and reduced response to uterotonics.
Introduction
Uterine atony is a major cause of postpartum hemorrhage (PPH), leading to maternal mortality worldwide. This condition can be triggered by various risk factors, including prolonged or excessively rapid labor; uterine distension due to factors such as multiple pregnancies, excessive amniotic fluid, or a large fetus; uterine fibroids; chorioamnionitis; and extended use of oxytocin. Inadequate uterine contractions, which can be localized or widespread, are also associated with other causes, such as retained placental tissue, placental abnormalities, blood clotting disorders, and uterine inversion. In addition, a body mass index exceeding 40, or class III obesity, is recognized as a risk factor for postpartum uterine atony. 1
Systemic lupus erythematosus (SLE) is a complex autoimmune disorder characterized by inflammation that affects multiple systems, with various contributing factors. If left untreated, SLE can lead to irreversible organ damage. 2 SLE is most frequently diagnosed in women of reproductive age. 3 The incidence of SLE in women is higher during this period, with a female-to-male prevalence ratio of 7–9:1. 4 While SLE does not affect fertility, it decreases pregnancy success rates and increases the risk of poor pregnancy outcomes. 5 Experts recommend that conception should occur within 6 months after SLE remission. Potential complications for both the mother and fetus can include miscarriage, preterm birth, preeclampsia, and neonatal lupus syndrome. 6
The incidence of uterine atony in pregnant women with SLE is rarely reported. This case report explores the correlation between uterine atony and SLE as well as the anatomical and pathological characteristics of the uterus in individuals with uterine atony and concurrent SLE.
Case report
A 37-year-old woman, G4P3A0, at 7.5 months of pregnancy, presented with complaints of increasingly frequent and intense cramping over the past 8 h before admission to the hospital. She noted significant fluid discharge from the birth canal 4 h prior to arrival. The mother reported feeling fetal movements. The patient has had SLE for about 9 years and regularly attends the rheumatology clinic. During her pregnancy, she was prescribed methylprednisolone 2 mg every other day, azathioprine 50 mg twice daily, calcium 500 mg twice daily, folic acid 1 mg daily, and aspirin 80 mg daily. She denied any other chronic illnesses such as hypertension, diabetes mellitus, asthma, or heart disease and reported no recent history of fever, cough, cold, or sore throat.
The patient arrived at the emergency department in the second stage of labor and received assistance for spontaneous delivery along with active management of the third stage. During the fourth stage, a physical examination revealed a blood pressure of 125/80 mmHg, a pulse rate of 104 beats/min, and a respiratory rate of 20 breaths/min. The patient was administered 20 IU of oxytocin via IV infusion at a rate of 20 drops/min and received a uterine massage. Ten minutes after placental delivery, active bleeding of 200 cc was observed with inadequate uterine contractions, prompting the rectal administration of three tablets of misoprostol and an IV injection of 0.2 mg methylergometrine. Fifteen minutes post-placenta delivery, blood pressure was 118/75 mmHg, and pulse rate was 120 beats/min. A two-line IV was established, a 1000 mL 0.9% NaCl loading dose was administered, and a second 0.2 mg IV methylergometrine injection was given. Thirty minutes later, estimated blood loss reached 500 mL, blood pressure dropped to 86/50 mmHg, pulse rate remained at 120 beats/min, respiratory rate increased to 22 breaths/min, and uterine atony persisted. An exploratory laparotomy was then decided.
In the operating room, an exploratory laparotomy revealed a uterus of a 20-week pregnancy, with no contractions and active vaginal bleeding, confirming uterine atony. A supravaginal hysterectomy was performed. Predelivery laboratory tests showed hemoglobin at 10.4 g/dL, hematocrit at 29.2%, platelets at 115,000/µL, and white blood cells at 12,210/mm3. Postpartum, hemoglobin remained at 10.3 g/dL initially, dropping to 8.4 g/dL and then to 7.5 g/dL due to ongoing bleeding from uterine atony. Platelet count decreased to 90,000/µL. The patient experienced hemorrhagic shock, with the lowest blood pressure recorded at 70/55 mmHg. Diagnosis included hemorrhagic shock due to uterine atony, P4A0 premature spontaneous labor, SLE with mucocutaneous and musculoskeletal involvement under therapy, anemia, thrombocytopenia, and stage I acute kidney injury. The patient was referred to Internal Medicine and Anesthesiology for further management.
The patient received 5 units of fresh frozen plasma and 9 units of packed red cells. Persistent bleeding resulted in emergency hysterectomy surgery. Post-hysterectomy, the hemoglobin was 8.5 g/dL. After the surgery, the patient’s condition improved, with hemoglobin reaching 12.3 g/dL and bleeding from uterine atony ceasing. The patient’s platelet count normalized to 260,000/µL and was discharged after a 5-day hospital stay. The patient was followed up in an outpatient setting after discharge. Both the patient and the baby showed no complaints and remained hemodynamically stable.
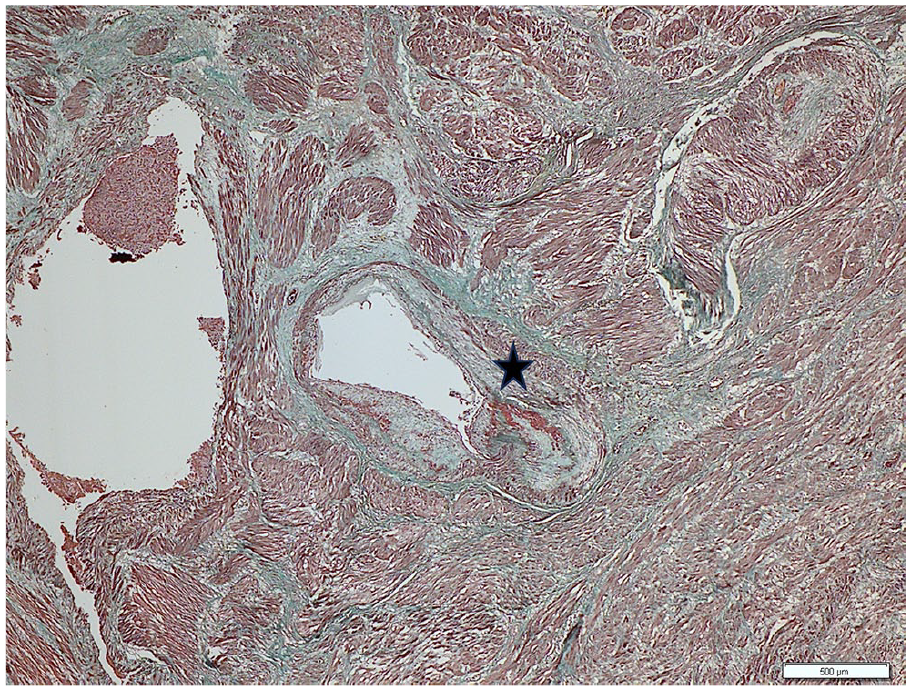
The uterine tissue obtained from the supravaginal hysterectomy was sent to the anatomical pathology laboratory for histopathological examination (Figures 1 and 2). The uterine myometrium showed hypertrophic and hyperplastic myocytes arranged in fascicles, with nuclei within normal limits. The connective tissue showed partial edema and fibrosis, along with infiltration of inflammatory cells, including lymphocytes, polymorphonuclear cells, and histiocytes. In addition, dilated and congested blood vessels were noted. Medium-sized blood vessels exhibited partial erosion in the tunica intima with normal nuclei, while the tunica media and adventitia showed partial fibrosis. No chorionic villi or decidual tissue were observed, and no signs of malignancy were detected.

Histological picture of the patient’s uterine myometrial layer stained with HE. Medium-sized blood vessels in the tunica intima, showing partial erosion with nuclei within normal limits. The tunica media and adventitia exhibit partial fibrosis. This histological appearance is observed at 200× magnification using HE staining, as indicated by the black asterisk.
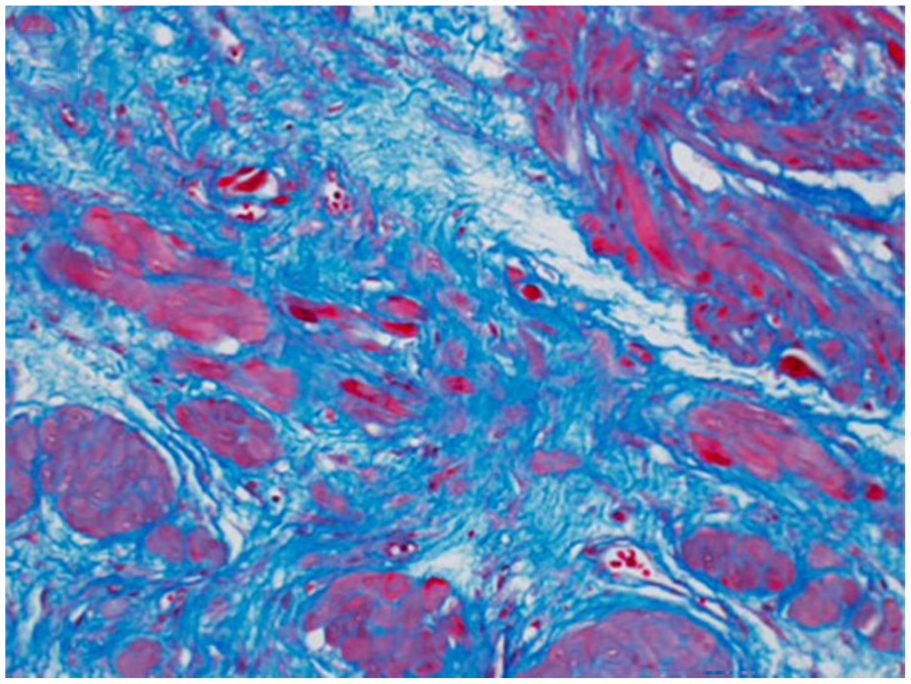
Histological picture of the patient’s uterine myometrial layer stained with MC. Fibrosis was evident in the tunica media of the blood vessels and the surrounding area. Myocyte cells are stained magenta, displaying a normal fascicular arrangement. The tunica media of the blood vessels appears green. In the connective tissue surrounding the vessels, there is a broader green-stained area. This histological appearance is observed at 200× magnification using MC staining, as indicated by the black asterisk.
Discussion
Correlation between uterine atonia and SLE
PPH is a common and serious complication of childbirth, affecting 1%–10% of deliveries and leading to ~80,000 maternal deaths annually. 7 In Indonesia, postpartum bleeding is primarily caused by the retained placenta (35.9%), followed by placental retention (25.0%), vaginal laceration (25.0%), and uterine atony (12.5%). 8
Uterine atony occurs when the muscle cells of the uterus fail to contract adequately in response to the natural oxytocin released during childbirth, preventing the spiral arteries from constricting and stopping blood flow. Risk factors include rapid or prolonged labor, uterine enlargement, fibroids, chorioamnionitis, high doses of magnesium sulfate, and obesity. Inadequate uterine contractions may also result from abnormal placental attachment, clotting disorders, or uterine inversion. 9
Despite experiencing postpartum uterine atony, the patient did not present the typical risk factors for the condition aside from having SLE. The patient delivered her baby spontaneously without complications, even though the birth was premature. Premature birth is a known risk for patients with SLE and is associated with disease activity—both clinically and through serological markers such as elevated anti-dsDNA titers and low complement levels—as well as high-dose steroid use, hypertension, and thyroid disorders. 10
Although uncommon, the correlation between SLE during pregnancy and uterine atony—potentially due to structural abnormalities in the myometrium—has been reported over the past decade, with most cases originating from Asian countries. These patients, typically aged 29–36 years, had been diagnosed with SLE and treated with corticosteroids for over 5 years, including during pregnancy. Notably, most cases involved nulliparous women, suggesting that SLE may contribute to uterine wall thinning. All reported patients underwent cesarean section followed by hysterectomy due to severe PPH.11–16 Our case differs in several key aspects. The patient had a history of three vaginal deliveries, distinguishing her from previously reported nulliparous cases. While multiparity can be a predisposing factor for uterine atony, it is not necessarily the underlying cause of the complications. 17 Ultimately, a hysterectomy was performed due to persistent uterine atony, similar to prior cases. However, a significant difference is that PAS (Placenta Accreta Spectrum) was absent in any of her pregnancies, further highlighting the variability in how SLE affects pregnancy outcomes. SLE patients may also have a poor response to oxytocin and experience uterine wall thinning, contributing to uterine atony. 12
Various theories have been proposed to explain how SLE influences pregnancy. Hormonal factors, particularly estrogen, are believed to play a role in uterine atony in SLE patients, as estrogen regulates myometrial thickness. Several studies have shown that pregnant patients with SLE have lower estrogen levels compared to those with normal pregnancies throughout gestation. Moreover, estrogen plays a role in myometrial thickening, primarily due to its ability to stimulate cell growth and proliferation, which, when unopposed by progesterone, can lead to abnormal thickening of the uterine lining. Reduced estrogen levels in SLE patients can disrupt myometrial hypertrophy and endothelial proliferation, potentially resulting in uterine wall thinning.11,18,19 Although estrogen levels were not measured in this case, myometrial thinning due to low estrogen is suspected to have contributed to uterine atony.
The patient’s long-term steroid use, including methylprednisolone, may have contributed to myometrial thinning. Steroids can disrupt protein synthesis and estrogen receptor activity, leading to structural changes in the uterine wall. 11 In this case, estrogen plays a crucial role in stimulating myometrial hypertrophy and endothelial proliferation during pregnancy. The antiestrogenic effect in SLE is thought to significantly influence these structural alterations. 20
Furthermore, the presence of antiphospholipid antibodies, frequently found in SLE, has been associated with microvascular thrombosis and endothelial injury; this alteration may reduce nitric oxide availability and alter vascular tone. This vascular impairment could further compromise the myometrial response to oxytocin, leading to uterine atony. 21 Clinicians should remain vigilant for signs of severe uterine wall thinning, as it can lead to uterine atony.
Pathological picture of uterine anatomy in uterine atony patients with SLE
In this case, histopathological analysis of the patient’s uterine tissue revealed hypertrophic and hyperplastic myometrial cells arranged in bundles with normal nuclei. The myometrial stroma showed partial fibrosis and scattered lymphocytic inflammation, while medium-sized blood vessels exhibited erosive changes in the intima, with fibrosis in the media and adventitia.
The histopathological findings in this case do not suggest signs of steroid-induced muscle damage, such as atrophy and centralized nuclei variation. Previous studies have linked SLE with myometrial fibrosis and vasculitis, which can contribute to PPH. SLE may damage uterine microvascular structures, leading to histopathological changes in the myometrium.11,13
Pregnancy requires an adequate immune response to maintain the maternal-fetal connection, but autoimmune disorders, such as SLE, increase the risk of complications. 22 Steroid treatment, such as prednisolone, reduces uterine natural killer cells and affects estrogen, potentially leading to myometrial thinning. Studies have shown that long-term steroid use in SLE patients can contribute to myometrial thinning (Figure 3) and uterine fibrosis (Figure 4), which may play a role in PPH.12,13 These findings are in line with the histopathological features observed in this case. However, it remains uncertain whether the thinning of the uterine wall resulted from long-term steroid use, the underlying pathophysiology of SLE, or a combination of both. A single case is insufficient to establish a definitive association, and most pregnant women with SLE or those taking prednisolone do not present with a thin uterine myometrium. Therefore, additional studies are needed to clarify the connection between SLE, steroid use, and uterine atony.

(A) Histopathological examination of the myometrium layer in pregnant women who experienced SLE and had a history of long-term treatment with steroids shows very thin and patchy uterine muscle. (B) Histopathological examination highlighted by immunostaining for α-SMA. 12
Histopathological assessment (Mason trichrome staining at ×200 magnification) showed a corpus myometrium-fibrosis ratio of 4:6. 13
Conclusion
This case highlights the increased risk of complications, including uterine atony and PPH, in pregnant women with SLE, particularly those on long-term steroid treatment. Histopathological findings suggest that steroid-induced fibrosis and vasculitis in the uterus, along with myometrial thinning, may contribute to these complications. SLE’s impact on the immune system and uterine health underscores the need for careful management of pregnancy in affected patients. Further research is necessary to better understand the relationship between SLE, steroid use, and uterine atony to improve outcomes for both mother and fetus.
Footnotes
Ethical considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for publication
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Author contributions
A.I.H. assessed and acquired the patient’s data. A.I.H. drafted the manuscript. R.A., A.S., H.H.S., F.F., and B.M.D. corrected and completed the manuscript. All authors contributed to the drafting of the article and agreed on the final version to be published.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
