Abstract
Objectives:
To compare the efficacy and safety of intravenous and intramuscular oxytocin in preventing atonic primary postpartum haemorrhage in the third stage of labour.
Methods:
A double-blind randomised clinical study on consenting women without risk factors for primary postpartum haemorrhage in labour at term. Two hundred and thirty-two women were randomly allotted into intravenous (n = 115) and intramuscular (n = 117) oxytocin groups in the active management of the third stage of labour. All participants received 10 IU of oxytocin, either IV or IM, and 1 ml of water for injection as a placebo via a route alternate to that of administration of oxytocin within 1 min of the baby’s delivery. The primary outcome measures were mean postpartum blood loss and haematocrit change. Trial Registration No.: PACTR201902721929705.
Results:
The baseline socio-demographic and clinical characteristics were similar between the two groups (p > 0.05). There was no statistically significant difference between the two groups with regards to the mean postpartum blood loss (254.17 ± 34.85 ml versus 249.4 ± 39.88 ml; p = 0.210), haematocrit change (2.4 (0.8%) versus 2.1 (0.6%); p = 0.412) or adverse effects (p > 0.05). However, the use of additional uterotonics was significantly higher in the intravenous group (25 (21.73%) versus 17 (14.53%); p = 0.032).
Conclusion:
Although oxytocin in both study groups showed similar efficacy in terms of preventing atonic primary postpartum haemorrhage, participants who received intravenous oxytocin were more likely to require additional uterotonics to reduce their likelihood of having an atonic primary postpartum haemorrhage. However, both routes have similar side effect profiles.
Keywords
Introduction
Postpartum haemorrhage (PPH) remains the commonest complication of the third stage of labour, and the major causes of PPH include uterine atony, tissue trauma and retained placenta.1,2 Uterine atony is the most common cause of primary PPH, occurring in 75%–90% of cases. 3 Although atonic primary PPH occurs in parturients with significant risk factors, the World Health Organisation (WHO) emphasised that the majority of parturients with atonic PPH had no observed risk factors. 4
An evidence-based set of interventions known as active management of the third stage of labour (AMTSL) has been recommended for reducing the incidence of uterine atonic primary PPH. 5 These interventions include parenteral administration of an oxytocic following the delivery of the baby, controlled umbilical cord traction and uterine massage. These ultimately enhance myometrial contractions and the delivery of the placenta, thereby reducing postpartum blood loss (PPBL).4–7 Following data assessment, the International Confederation of Midwives (ICM) and the International Federation of Gynaecologists and Obstetricians (FIGO) have, in a combined declaration, suggested the routine use of AMTSL for every parturient. 7 The FIGO proposed in 2022 that all birth attendants, regardless of where they practice, should routinely use AMTSL and that only those who have not had any uterotonic and uterine tone assessments should receive a uterine massage. 8 The WHO reported in 2020 that AMTSL is linked to a significant decrease in the incidence of PPH when compared to expectant management. 9 When contrasted with expectant management of the third stage of labour (which involves no use of oxytocic), AMTSL reduced PPH by 60%–70%, maternal postpartum anaemia by 50%, blood transfusion by 65% and use of therapeutic uterotonics by 81%.10–12 The benefits of AMTSL in preventing and reducing primary PPH following vaginal delivery for women without significant risk of PPH have been reported in a meta-analysis. 13

Consort flow chart.
The prophylactic uterotonics extensively studied in the AMTSL at different dosages and modes of administration include oxytocin, ergometrine, syntometrine, carbetocin and misoprostol.14–17 Nevertheless, oxytocin is considered the preferred uterotonic drug,5–7 and the recommended first-line oxytocic to prevent PPH in the third stage of labour. 6 This is because oxytocin is effective within 1–3 min post-injection, has comparable efficacy but lesser adverse effects when compared with other oxytocics and can be given to all pregnant women.5,18 Cotter et al., 19 in a systematic review comparing prophylactic oxytocin with no uterotonics, with or without AMTSL, reported that oxytocin was effective in terms of decreasing PPH and the need for additional uterotonics.
Although the WHO recommends the use of 10 IU of oxytocin either intravenous (IV) or intramuscular (IM) in the AMTSL, 8 they stated that there is clear evidence in favour of IV oxytocin with regards to a reduction in the risk of PPH, severe PPH, the need for blood transfusion and severe maternal morbidity. However, there is no difference in the side effect profile when compared to IM oxytocin, and the feasibility of IV administration may vary from one setting to another. 9 A randomised control trial has shown that a bolus IV oxytocin injection of 10 IU was more effective than a dilute oxytocin infusion and had no associated adverse haemodynamic outcomes when used for PPH prophylaxis in patients delivering vaginally. 20 Therefore, administering IM oxytocin to parturients already on IV lines or in centres such as ours, where there are skills for IV administration in the third stage of labour for PPH prevention, may not be reasonable in line with the practice of evidence-based obstetrics.
Although oxytocin has remained a frontline uterotonic in the AMTSL, only a few studies have compared its IM and IV routes of administration for atonic PPH prevention before and after the recommendations by these organisations. 21 Many studies on prophylactic uterotonics in the AMTSL have rather focused on comparing oxytocin and ergometrine effects in preventing PPH.14–17 Oladapo et al., in a recent Cochrane review, concluded that in order to avoid PPH during vaginal delivery, oxytocin administered intravenously works better than when administered intramuscularly. The injection of oxytocin intravenously raises no new safety issues, and the adverse effect profile is similar to that of IM administration. They recommended that research should examine if the intervention is feasible, acceptable, and resource-efficient, especially in low-resource environments. 22 A study comparing the efficacy and adverse effects of IV and IM oxytocin for preventing atonic primary PPH in the third stage of labour is therefore desirable, especially in a resource-poor setting like ours. Thus, this study was aimed at comparing the efficacy and adverse effects of oxytocin administered via IV and IM routes for preventing atonic primary PPH in the third stage of labour.
Methods
Study design
The study was a double-blind, randomised, controlled study.
Study setting
The study was conducted at Nnamdi Azikiwe University Teaching Hospital (NAUTH), Nnewi, Nigeria, from 15 February 2019 to 15 August 2019. Ethical clearance was obtained from the NAUTH Ethics Committee with the reference number NAUTH/CS/66/VOL10/169/2017/078.
Study population
The study was carried out among pregnant women with no risk factor for PPH seen in the labour ward during the period of the study and who gave written informed consent and met the inclusion criteria.
Sampling approach
A simple random sampling method was used in which eligible and consenting women with labour pains presenting at the labour ward were randomly allotted into two equal groups, viz., the IV and IM oxytocin administration arms, using randomly permuted blocks (blocks of 4 with an allocation ratio of 1:1) with software available online (http://www.randomization.com).
Inclusion criteria
These included all consented healthy women who are 18 years of age or older with term singleton normotensive pregnancy in spontaneous established labour and having successful vaginal deliveries.
Exclusion criteria
Those excluded from the study were women with preterm gestation, grand-multiparity, previous caesarean section or uterine surgery, contraindication to vaginal delivery, multiple pregnancy, foetal macrosomia, polyhydramnios, previous history of PPH, those on induction of labour, medical illness (hypertension, diabetics, hepatic disease, renal disease, pre-eclampsia), anaemia, febrile illness, hypotension, tachycardia and coagulation disorders, antepartum haemorrhage and uterine fibroids. Women who had augmentation of labour were also excluded from the study.
Sample size determination
This sample size (n) was determined using the formula: n = [(a + b)2·(p1q1 + p2q2)]/x2, Where p1 = proportion of subjects with PPH using the IV oxytocin group in a pilot study 23 = 0.24, q1 = proportion of subjects without PPH using IV oxytocin group (= 1 − p1) = 0.76, p2 = proportion of subjects with PPH using IM oxytocin group in a pilot study 24 = 0.23, q2 = proportion of subjects without PPH using IM oxytocin group (= 1 − p2) = 0.77, x = the clinical difference noted at a 20% reduction in blood loss; a = conventional multiplier for alpha = 0.05 = 1.96; b = conventional multiplier for power at 90% = 1.28. There are approximately 96 subjects in each arm. Considering an attrition rate of 20.0%, 116 participants were recruited in each arm.
Study procedure
A total of 232 participants were enrolled in this study. They were informed about the study at the antenatal clinic, and written informed consent was obtained. Eligible, unbooked participants presenting in the labour ward for the first time and who gave consent were recruited into the study. Recruited participants were randomised into two arms, viz., the IV and IM oxytocin administration arms, using a random number table. Allocation concealment was performed using serially numbered, sealed, opaque envelopes, stored and opened by independent staff (the nurse midwife) of the hospital. Each of the envelopes contained either a folded slip of paper displaying an IV oxytocin and IM placebo (Group 1), placing that patient into the group receiving the IV oxytocin and IM inactive placebo, or a paper displaying an IM oxytocin and IV inactive placebo (Group 2), placing that patient into the group receiving the IM oxytocin and IV inactive placebo. Group allocations never change after the envelope has been opened, and allotment to the arms is not done until the delivery of the foetus.
Following participant recruitment and the establishment of the active phase of labour, IV access was secured using a 16-G cannula. Blood samples were collected from each participant, delivered into an ethylenediaminetetraacetic acid bottle, and sent to the haematology laboratory to obtain the haematocrit levels using the microhaematocrit centrifuge and reader (Haematokrit 210; Hettich, Tuttlingen, Germany). Each sterile gauze, delivery mat (60 × 90 × 0.5 cm3; Divinecare®, China), and sanitary pads (25 × 6 × 2 cm3; Ladysept®, Nigeria) were weighed using a digital weighing scale (with a 1-g error range) before use. Other labour management and delivery were ensured as per the NAUTH labour ward protocol. 25 Most of the amniotic fluid was excluded from contributing to the amount of PPBL. During the second stage of labour, a clean delivery mat was placed under the maternal perineum. For participants who have had spontaneous or artificial rupture of foetal membranes (ARM), delivery of the baby was conducted, while for those with intact membranes, ARM was immediately done and delivery of the baby was conducted. At this point, ‘virtually all’ the amniotic fluid has gone out. Following this, the soaked delivery mat was immediately replaced with a pre-weighed, clean mat to absorb blood loss during the third stage of labour. The new mat was used in an attempt to exclude amniotic fluid from PPBL.
All participants received 10 IU of oxytocin (Syntocinon®; Novartis) contained in a 1 ml ampoule, either IV or IM, and 1 ml of water for injection as a placebo via a route alternate to that of administration of oxytocin within 1 min of the delivery of the baby. An independent observer prepared the oxytocin into a 2-ml syringe and labelled it ‘IV’ or ‘IM’ and the water for injection into another 2-ml syringe (labelled alternate route) in the second stage of labour and administered the oxytocin and placebo based on the instructions on the slip contained in the sealed opaque envelop. To ensure that the placebo was not given instead of oxytocin, an independent observer loaded the syringe with oxytocin and labelled it ‘IV’ or ‘IM’ only, whereas the syringe loaded with water for injection was labelled the route alternate to that of oxytocin according to the instructions on the slip in the sealed opaque envelop before both syringes are taken to the patient for administration. This labelling was done using pieces of pre-labelled masking tape marked ‘IV or IM’. This was done in the second stage room out of the sight of the patient, while the outcome assessor was asked to be in the first stage room to ensure blinding. The placebo is used to blind the patient and the outcome assessor to the route of oxytocin. In the IM group, 1 ml of oxytocin (contained in the 2 ml syringe) was administered by inserting the needle connected to the syringe perpendicularly and deep into the vastus lateralis part of the quadriceps femoris muscle and 1 ml of water for injection (contained in the 2 ml-syringe) slowly over 2 min into a peripheral vein. In the IV group, 1 ml of oxytocin (contained in the 2 ml-syringe) was administered as a bolus injection slowly over 2 min into a peripheral vein and 1 ml of water for injection (contained in the 2 ml-syringe) into the muscle. Following the administration of the oxytocin and water for injection, the outcome assessor was called into the second stage room to measure the outcomes. The maternal pulse rate and blood pressure were checked every 5 min for 30 min, then every 15 min for 1 h and 30 min by the research team members since hypotension and tachycardia are some of the outcomes assessed. The blood pressure was measured using a manual mercury sphygmomanometer (Dekamet MK3; Accoson®, UK) and a stethoscope (3M Littman® Classic IIG, USA), which were checked for functionality. The mercury meniscus (the gauge) was inspected to ensure it was at zero, and an appropriate-sized adult cuff was provided. The readings were recorded to the nearest 2 mm (rounded off upward).
Controlled cord traction was performed, and the parturient was encouraged to bear down simultaneously to deliver the placenta. The placental lobes were inspected for completeness under a good light source and running water. The placenta was manually removed if it was not delivered within 30 min of delivery. Palpation of the tone of the uterus and uterine massage were done by a research assistant every 15 min for 2 h to assess for and prevent uterine atony. The perineum, vagina, and cervix were inspected for lacerations. Lacerations or episiotomy wounds, if present, were repaired immediately under local anaesthesia (lidocaine).
A sanitary pad was placed over the perineum. If there was significant vaginal bleeding within 10 min of the oxytocin injection adjudged to be only due to uterine atony, the uterus was massaged, the urinary bladder emptied, and additional uterotonics were administered at the discretion of the doctor on duty. The patient was monitored for vaginal bleeding for the next 24 h, as well as their vital signs. The amount of vaginal blood loss and history of side effects such as shivering, pyrexia, and nausea/vomiting 24 h postpartum were recorded by the research team. The weights of the blood-soaked delivery mat and perianal pads were measured using the same digital weighing scale. The total volume of blood loss was calculated using the method described by Gai et al. 26 as follows: Quantity of blood (ml) = [(weight of used materials + weight of unused materials) weight of all the materials before use] ÷ 1.05. Blood samples were drawn from the parturient for haematocrit measurement at 48 h postpartum. In this study, primary atonic PPH was defined as vaginal blood loss of 500 ml or more only due to uterine atony within the first 24 h postpartum.
Outcome measures
The primary outcome measures were the mean PPBL following vaginal delivery and haematocrit change. The secondary outcome measures included use of additional uterotonics, need for blood transfusion, need for surgical intervention, postpartum maternal anaemia and adverse effects (nausea, vomiting, hypotension, etc.).
Blood samples collected for both prepartum and postpartum packed cell volume estimations were analysed by senior laboratory scientists in the haematology laboratory of the same institution.
Data and statistical analysis
The data after collection was checked for completeness and tabulated. Then the randomisation code was decoded. Analysis was done using SPSS version 26 (IBM Corp., Chicago, IL, USA). Continuous variables were represented as mean ± standard deviation (X ± SD) (if normally distributed) or median and interquartile range (if not normally distributed). Initially, the Shapiro-Wilk test was used to verify the normality of the data distribution. Categorical variables were analysed using Chi-square tests where appropriate; continuous data were analysed using the T-test if parametric or the Mann-Whitney T-test if non-parametric. 27 The statistical significance was inferred at a p-value <0.05.
Results
Overall, during the study period, 397 women were screened for eligibility, of which 316 were enrolled in the study. Of the 316 women enrolled, 159 were randomly assigned to receive oxytocin intravenously, and 157 were randomly assigned to receive oxytocin intramuscularly. Following randomisation, a total of 84 women were excluded from the IV and IM oxytocin groups due to genital lacerations, an emergency Caesarean section from failed vacuum delivery, and their insistence on going home within 24 h postpartum without allowing adequate time for haematocrit estimation. At the end, 115 women were available for analysis in the IV arm and 117 in the IM arm of the study.
The demographic and obstetric characteristics were similar between the IV and IM groups. There was no statistically significant difference between the age of participants, parity, booking status, mean gestational age at delivery, neonatal birth weight, and duration of the third stage of labour (p < 0.05) in both groups. The mean age of participants in the IV group was 30.35 years (±4.92) and 30.16 years (±5.18) for the IM group. The majority of participants were multiparous (89.6% versus 86.3%) when compared to nulliparous women (11.3% versus 12.8%) in both groups, respectively. The gestational age in days at delivery was 279.9 (±54.89) for the IV group and 278.0 (±54.72) for the IM group. The mean foetal birth weight was 3.10 ± 0.34 kg, when compared to 3.63 ± 0.32 kg in the IM group. This is shown in Table 1.
Demographic and obstetric characteristics of the two study groups.

T-test.
Table 2 shows PPBL in the two groups. There was no statistically significant difference in the mean PPBL between the IV and IM groups (254.17 ± 34.85 ml versus 249.4 ± 39.88 ml; p = 0.210). Additionally, there was no significant difference in the proportion of women who had atonic primary PPH between the IV and IM groups (14.8% versus 10.3%; p = 0.110).
PPBL in the two study groups.
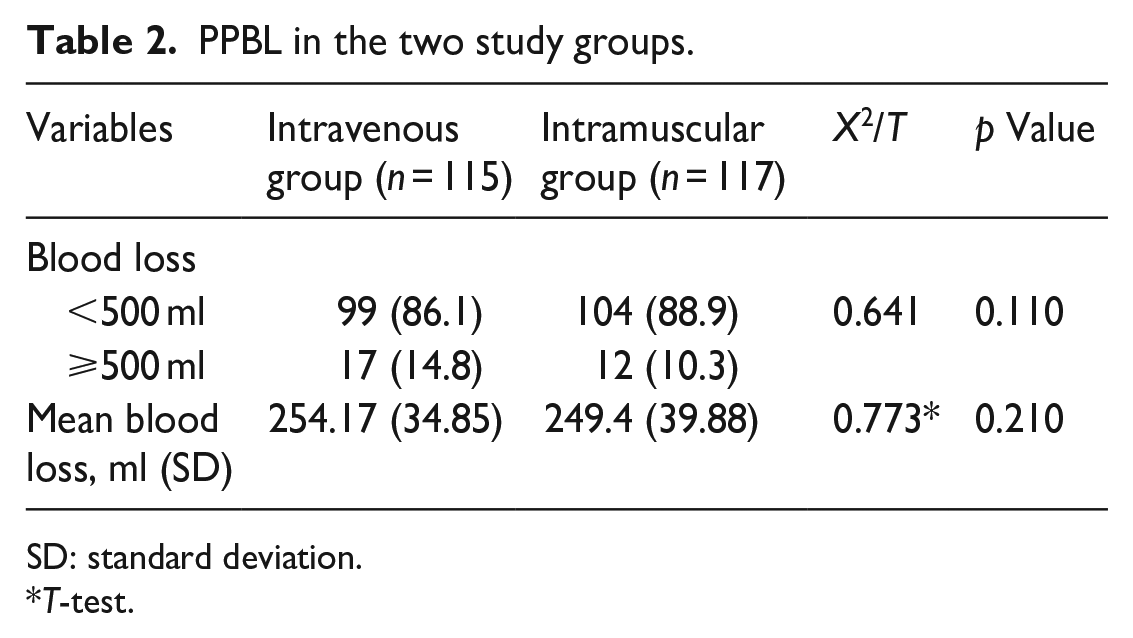
SD: standard deviation.
T-test.
The mean percentage of prepartum (34.32 ± 3.43% versus 35.30 ± 3.37%; p = 0.347) and postpartum (31.92 ± 2.57% versus 33.2 ± 2.21%; p = 0.215) haematocrit, respectively, in the IV and IM groups were similar in both groups. There was no significant difference in the mean haematocrit change between the two study groups. (2.4 ± 0.8% versus 2.1 ± 0.6%; p = 0.412). This is shown in Table 3.
Haematocrit levels in the two study groups.

These maternal vital signs prior to oxytocin and placebo administration between the two study groups are shown in Table 4. There was no significant difference in maternal pulse, temperature or systolic and diastolic blood pressure between the IV and IM groups (p > 0.05). In addition, there were also no significant differences between maternal vital signs following oxytocin and placebo administration between the two groups (p > 0.05). This is shown in Table 5.
Maternal vital signs prior to oxytocin or placebo administration.
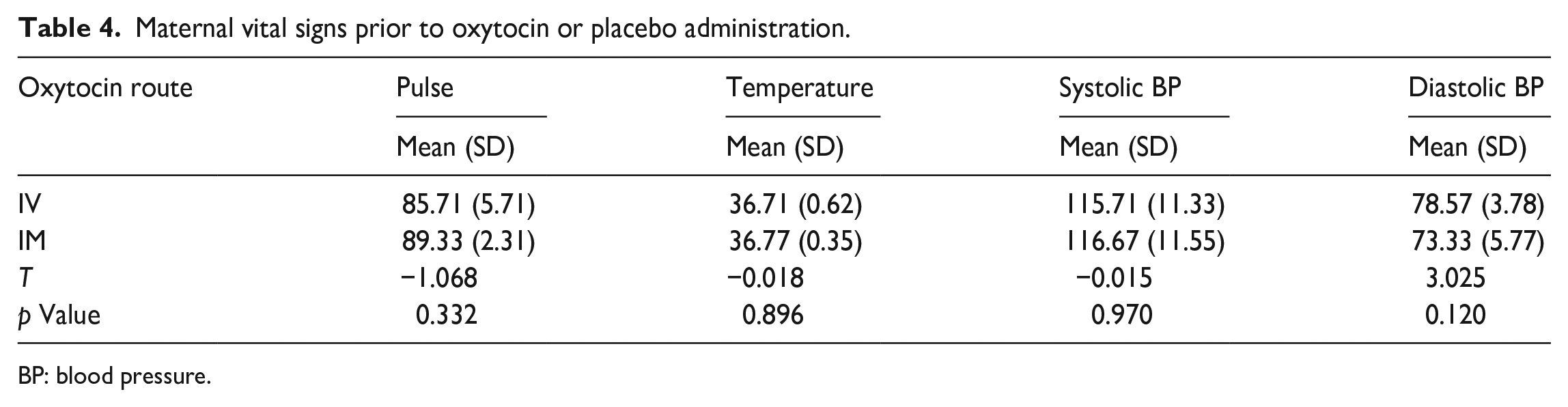
BP: blood pressure.
Maternal vital signs following oxytocin and placebo administration.
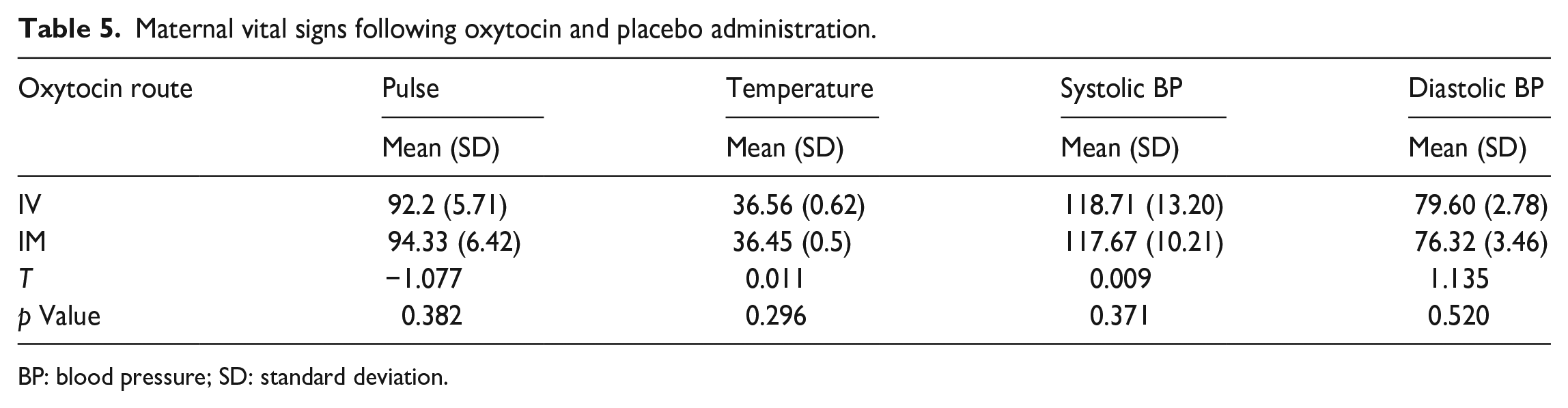
BP: blood pressure; SD: standard deviation.
Table 6 shows the adverse maternal effects of oxytocin in the IV and IM groups. There was no significant difference in the incidence of nausea, vomiting, headache, shivering, pyrexia or tachycardia in both groups, respectively (p > 0.05). No incidence of hypotension was recorded in both groups.
Adverse effects in the two study groups.

Interestingly, the proportion of women who required additional uterotonic for primary PPH prophylaxis in participants who developed uterine atony was significantly higher in the IV group than in the IM group (21.73% versus 14.53%; p = 0.032). However, there was no significant difference in the need for blood transfusion (9.57% versus 5.13%; p = 0.071) or postpartum maternal anaemia (13.04% versus 11.10%; p = 0.853) between the two groups, respectively. The need for surgical interventions for controlling atonic primary PPH was similar in both groups. This is shown in Table 7.
Secondary outcomes of the study.
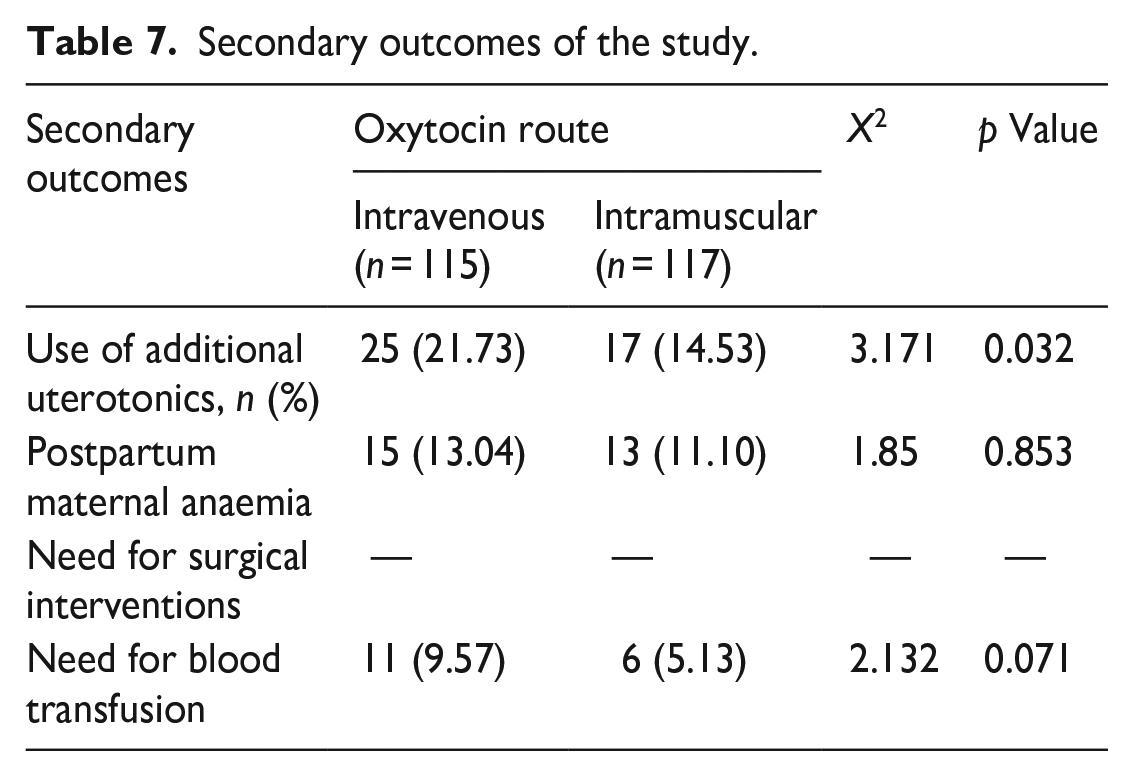
Table 8 shows the surgical interventions performed in this study. The most commonly performed surgical intervention was manual removal of the placenta (ROP). This procedure was performed in 4.31% of participants in the IV group, compared to 2.59% in the IM group. Although the need for manual ROP was slightly higher in the IV group than in the IM group, the difference was not statistically significant (p = 0.211). Additionally, 4.3% of the participants in the IV group had evacuation of retained product of conception (ERPOC) as opposed to 2.59% in the IM group (p = 0.211).
Surgical interventions in the two study groups.
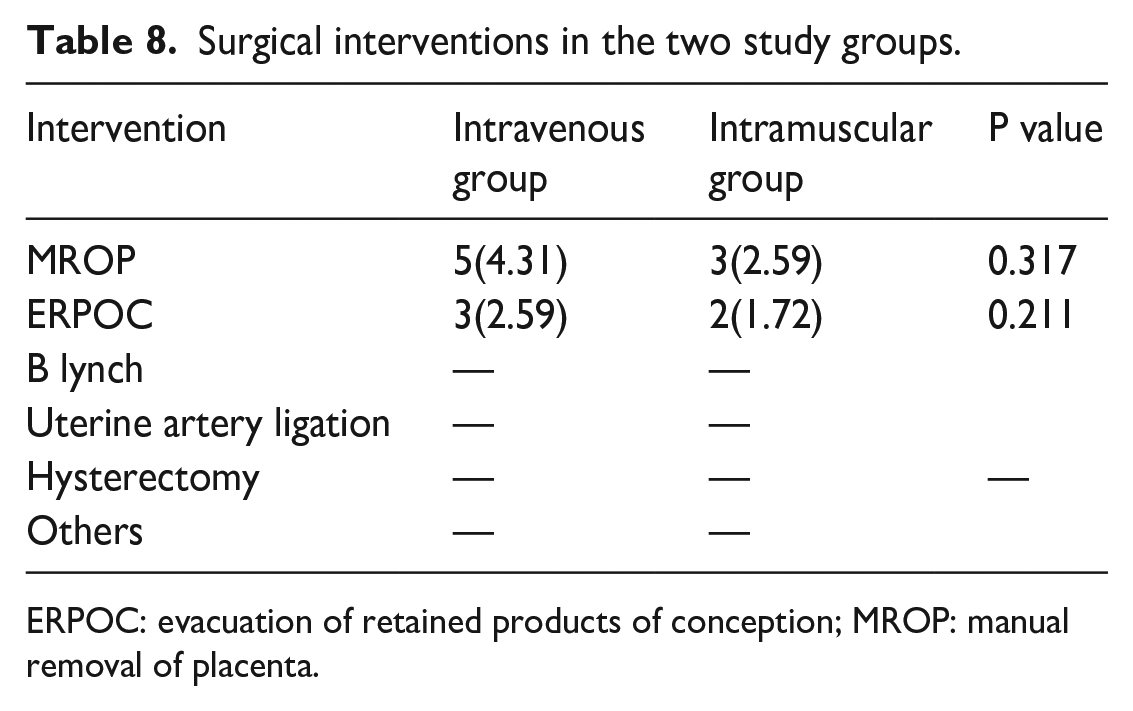
ERPOC: evacuation of retained products of conception; MROP: manual removal of placenta.
Discussion
The motivation behind this study is that primary PPH still remains one of the leading causes of maternal mortality, especially in developing countries like Nigeria. This present study demonstrated that either the IV or IM route of oxytocin administration did not significantly influence the mean PPBL. This finding is in conformity with the finding in a similar study in Turkey by Orhan et al. 28 (226.6 ± 172.9 ml for the IV group versus 253.5 ± 176.5 ml for the IM group; p = 0.134). Also, with another similar Turkish study by Dagdeviren et al., 29 (32.04 ±19.03 ml for the IV group versus 31.65 ± 21.43 ml; p = 0.572), Sheldon et al., following their assessment of the independent and combined effectiveness of all three AMTSL interventions and the effect of the route of oxytocin administration on mean PPBL using secondary data from two robust multi-centred double-blinded randomised control trials30,31 reported that when oxytocin was combined with the other two AMTSL interventions, the route of administration had no effect. 32 In contrast to this finding, Adnan et al., 33 in Ireland, in their Labour Oxytocin Route (LabOR) trial, found a significant reduction in mean PPBL (385 ml for IV group versus 445 ml for IM group; odds ratio = 60 at 95% CI; p = 0.01). 33
This current study also demonstrated that no significant difference in the haematocrit change was found between the two study groups. This finding is in agreement with the findings in two previous studies.28,33 Orhan et al. 28 noted that the haematocrit change was similar when oxytocin was administered in the context of the AMTSL in both study groups (3.9 ± 2.9% for the IV group versus 3.5 ± 3.1% for the IM group) (p > 0.05). 28 Similarly, Adnan et al., 33 in their RCT trial, found that the haematocrit change was similar for both study groups (0.047 ± 0.042% for the IV group; 0.050 ± 0.045% for the IM group; p = 0.29). The findings of similar haematocrit change noted in the current and previous studies are probably a reflection of the statistically similar mean PPBL as well as similar mean pre- and postpartum haematocrit levels in both study groups noted in these studies. The finding of similar haematocrit change or decrease in this current study suggests that there is no significant difference in the occurrence of postpartum maternal anaemia when oxytocin is administered intravenously or intramuscularly in the AMTSL.
Nevertheless, in this current study, the use of additional uterotonic drugs was significantly higher in the IV group than in the IM group. This is in conformity with the findings by Dagdeviren et al. 29 but in disagreement with two other previous studies. Dagdeviren et al. and Orhan et al. noted no significant difference in the use of additional uterotonics in the two study groups. 28 Adnan et al., however, corroborated the finding by Dagdeviren. One possible reason for this finding is that following IV administration of oxytocin, the onset of action/myometrial contractility is almost immediate, with a plateau concentration after 30 min, whereas its onset of action is between 3 and 7 min, with its clinical effect lasting up to 1 h when administered intramuscularly. 1 The IM oxytocin may therefore be regarded as a ‘sustained/continuous release’ oxytocin, causing sustained uterine contraction and therefore accounting for less use of uterotonics when compared to the IV oxytocin.
In the current study, the occurrence of side effects following IV oxytocin did not differ significantly from that of IM oxytocin. This finding is similar to a study by Dagdeviren et al. 29 but differs from case reports that showed bolus IV oxytocin is associated with serious maternal haemodynamic consequences.34,35 However, earlier obstetric textbooks cautioned that oxytocin should be administered via the IM route or as a dilute infusion.36,37 Orhan et al. 28 observed that there were no adverse effects except for uvular oedema in only one woman in the group that received IM oxytocin following the delivery of the baby. Adnan et al. 33 also noted that the number of side effects was lower in women who received IV oxytocin compared to those who received IM oxytocin.
The proportion of women who had postpartum anaemia and the need for blood transfusion were similar in the two study groups, and this is similar to Dagdeviren et al.’s 29 findings. Orhan et al. 28 reported no postpartum anaemia or blood transfusion for both groups.
The findings from this present study have some important clinical and economic implications, especially in our settings. In our setting, primary health centres and delivery homes far outnumber centres, with the maternity services occupied by health workers who may lack the requisite skills for IV drug administration. As a result, IM oxytocin may be recommended as the preferred choice in the AMTSL since it requires fewer skills to administer and is as effective and safe as IV oxytocin.
This work is a randomised study, and its methodology was designed to significantly reduce the bias introduced by liquor amnii in measuring PPBL in some previous randomised studies. Unlike in previous randomised studies, this present study assessed only the amount of blood loss from uterine atony in the third stage of labour by excluding those with PPH from genital lacerations (a complication of the second stage of labour), which could be a confounder when included. Also, this study is unique in that it did not include people with labour augmentation or induction using oxytocin. This research design phenomenon could be a key variable in interpreting other works that did find the IV route to be a more efficacious route for prophylaxis. Some limitations were difficulty in the complete elimination of liquor amnii in the measured blood loss, and the use of an electrocardiographic machine in the second stage room to study the hemodynamic effects of oxytocin was not feasible. Another limitation is that the effects of the bias from incomplete blinding of the research team members and those of the performance bias from dissimilar packaging of the oxytocin and placebo, though present, were reduced by complete blinding of the outcome assessment. It was not feasible to manually measure all the postpartum maternal vital signs before oxytocin-placebo administration and to administer oxytocin as well as placebo within one minute following the delivery of the baby (in the context of AMTSL). Additionally, this study could not report some important but not predetermined outcome data. For example, we do not have the data for maternal weight, body mass index, or length of first or second-stage labour. Also, we do not have data on the patterns of treatment for those who received or needed more uterotonics. These will be evaluated in future studies on the topic. The study adhered to the CONSORT guideline. 38
Conclusion
Although intravenously and intramuscularly administered oxytocin in the context of AMTSL show similar efficacy in terms of preventing mean blood loss from uterine atony of at least 500 ml in the first 24 h postpartum, patients who received IV oxytocin are more likely to require additional uterotonics to reduce their likelihood of having atonic primary PPH. Oxytocin administered via the two routes shows a similar side effect profile.
Since the administration of IM oxytocin is easier and requires fewer skills than IV oxytocin and can be administered by all maternity staff and centres, the findings from this present study suggest that the IM route may be considered the preferred route of prophylactic oxytocin in the AMTSL. This is imperative in order to significantly reduce the occurrence of atonic primary PPH and its associated maternal morbidity and mortality, especially in our resource-poor settings. A well-designed and robust multi-centre double-blind placebo-controlled trial is needed to increase the body of evidence and enhance the generalisability of the findings from this present study.
Footnotes
Acknowledgements
The authors are grateful to the recruited study participants, the NAUTH hospital staff and management, and everyone who took part in the study.
Author contributions
EIO, the principal investigator, was involved in conceptualisation and design, manuscript writing and revision, and data collection. GUE, JII and COE contributed to supervision, manuscript writing and revision. CGO, EPI, EOU, BCO, IIM, OSU, CAO, DNO, CBO, CCO, CLO, DCI and IJO were involved in manuscript writing and revision, data collection and analysis.
Consent for publication and consort statement
Not applicable in this study. The study adhered to the CONSORT guideline.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The cost of the research was entirely borne by the researcher.
Ethical considerations and consent for the study
Ethical approval for the conduct of the study was obtained from the NAUTH Ethics Committee with the reference number NAUTH/CS/66/VOL10/169/2017/078. The study was conducted based on the ethical principles for conducting human research according to the Helsinki Declaration. A written informed consent was obtained from the participants prior to the study.
Informed consent
A written informed consent was obtained from the participants before the study.
Trial registration
The trial was registered with the Pan African Clinical Trial Registry (PACTR) with approval registration number PACTR201902721929705.
