Abstract
Cardio-cerebral syndrome refers to cardiac injury following cranial brain injury, representing a critical complication of stroke associated with high morbidity and mortality. Although exercise treadmill testing is widely utilized for myocardial ischemia evaluation, its role in unmasking neurogenic syncope or cardio-cerebral interactions remains under investigation. Existing literature emphasizes that only a small proportion of post-stroke syncope cases undergo comprehensive assessment for combined cerebrovascular–cardiac etiologies, particularly evident in slow-type arrhythmia cases frequently misattributed solely to vasovagal syncope despite potential contributions from brainstem injury or autonomic dysregulation. While most reported post-exercise syncope cases attribute symptoms to vasovagal syncope or common etiologies, underlying cardiovascular/cerebrovascular pathologies are often overlooked, with limited discussion on post-exercise arrhythmia–cerebrovascular disease associations. This report details a unique case of exercise treadmill testing-induced syncope characterized by Brady arrhythmic electrocardiographic changes. Syncope evaluation revealed a newly diagnosed pontine infarction, while cardiac workup excluded structural heart disease, coronary artery stenosis, and Brugada syndrome, underscoring the differential diagnosis challenge between neurogenic and cardiogenic syncope. This case uniquely demonstrates the mechanistic overlap between vasovagal syncope and cerebrovascular injury: although bradyarrhythmia during exercise treadmill testing is typically attributed to cardioinhibitory vasovagal syncope, concurrent pontine infarction likely disrupted brainstem cardio-regulatory centers, sensitizing the patient to vagal hyperactivity. Through systematic analysis of specific arrhythmia–cerebrovascular disease relationships, the final diagnosis identified vasovagal syncope as the primary cause, with cerebral infarction serving as a potential exacerbating factor. This exercise-induced vasovagal syncope complicating stroke provides clinical insights into recognizing overlapping neurovascular–cardiovascular mechanisms during syncope evaluation.
Introduction
Cardio-cerebral syndrome (CCS) is a term used to describe various cardiac dysfunctions caused by acute brain diseases involving the hypothalamus and brainstem autonomic nervous system, which can often lead to serious cardiovascular diseases such as acute myocardial infarction (AMI), arrhythmias, myocardial ischemia, or heart failure. 1 Cerebral infarction, particularly involving the brainstem or insular cortex, disrupts sympathetic and parasympathetic pathways, leading to catecholamine surges, electrolyte imbalances, and arrhythmias. These cardiac manifestations often precede clinical deterioration, underscoring the need for early recognition. 2 Syncope refers to a temporary loss of consciousness, and post-exercise syncope can be caused by a variety of factors. These may include increased activity of the sympathetic nervous system, underlying cerebrovas-cular diseases, or cardiac issues. In addition, post-exercise syncope can be triggered by conditions such as hypoglycemia, severe anemia, or hyperventilation. The exercise treadmill test, as a graded exercise test commonly used for the diagnosis of effort-induced myocardial ischemia, can also trigger vasovagal syncope (VVS) and other conditions. Most reported cases of post-exercise syncope are considered neurally mediated syncope during recovery or other common etiologies unless there are clear cerebrovascular disease symptoms. However, due to overemphasis on common causes of syncope, potential cerebrovascular or cardiovascular system diseases are often overlooked in patients with such symptoms. Previous studies rarely discussed the association between post-exercise arrhythmias and cerebrovascular diseases. Through reporting the diagnosis and treatment process of a patient with post-exercise syncope complicated by a specific arrhythmia, our study identified a newly developed cerebral infarction in this patient, deeply explored the association between specific arrhythmias and cerebrovascular diseases and ultimately concluded that it represented VVS, while cerebral infarction might have acted as an exacerbating factor for symptom onset. This case report of exercise-induced VVS following a cerebral infarction provides insight for future clinical diagnoses.
Case description
A 47-year-old male patient was admitted to our outpatient department on October 5, 2023, due to “intermittent dizziness for 1 week.” The patient experienced dizziness without any obvious triggers 1 week prior, with no chest tightness, shortness of breath, chest pain, radiating pain, dizziness, blurred vision, syncope, nausea, or vomiting. Symptoms would alleviate after resting for a few minutes. The patient self-measured elevated blood pressure, with the highest reading being 212/114 mmHg, and did not receive any treatment. There is no history of other chronic illnesses, but the patient’s father has hypertension.
Upon admission, the patient’s vital signs were as follows: temperature 36.5°C, heart rate 78 beats/min, respiratory rate 20 breaths/min, blood pressure 195/98 mmHg, and BMI 36.57 kg/m2. Physical examination of the heart, lungs, abdomen, and nervous system was unremarkable. Laboratory tests, including blood and stool routine tests, liver and kidney function, electrolytes, coagulation function, thyroid function, glycated hemoglobin, cardiac markers, and BNP, showed no abnormalities. Urine protein was positive, and blood lipid levels were elevated: triglycerides 3.56 mmol/L, total cholesterol 5.94 mmol/L, low-density lipoprotein cholesterol 3.87 mmol/L.
The electrocardiogram showed sinus rhythm, QS waves in lead III, and T-wave changes in lead aVL. Cardiac ultrasound and chest X-ray revealed no abnormalities. The clinical provisional diagnosis is stage 3 extremely high-risk hypertension.
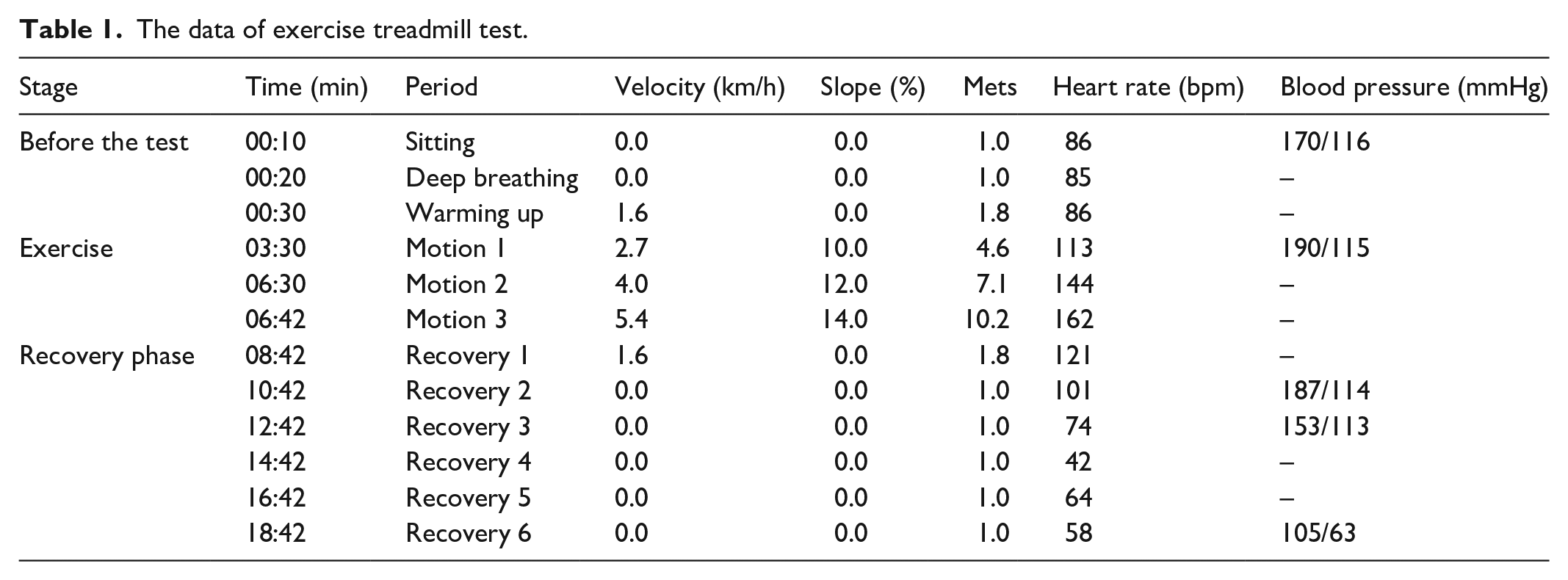
After admission, the patient was given stable antihypertensive treatment, and the dizziness symptoms improved significantly. Due to positive urine protein, we cannot rule out target organ damage from hypertension. Further inquiry into the patient’s medical history revealed occasional chest tightness after activity. The patient had comorbidities of hypertension, hyperlipidemia, and high-risk factors for coronary artery atherosclerotic heart disease. In addition, there were atypical T-wave changes on the electrocardiogram, not excluding the possibility of concomitant coronary artery atherosclerotic heart disease. It was decided to first perform exercise treadmill testing (ETT) to confirm the presence of exertional angina. During the test (using the Bruce protocol3,4), significant ST-T changes were observed when the patient reached the target heart rate (as shown in Figure 1), indicating a positive response, leading to the test being stopped. At 6 min into the recovery period, the patient suddenly developed bradycardia, with the electrocardiogram showing sinus arrest and junctional escape rhythm (as shown in Figure 2), with a minimum heart rate of 24 beats/min, accompanied by a drop in blood pressure, profuse sweating, dizziness, and loss of consciousness. Oxygen was administered, and after the patient was placed in a supine position, the rhythm and blood pressure recovered within a few minutes (as shown in Table 1). Further evaluation with coronary CT (Computed Tomography) angiography showed no significant vascular stenosis, while CT angiography of the head and neck suggested multiple narrowing of cerebral arteries, with occlusion of the left vertebral artery. A brain MRI revealed multiple lacunar infarctions, with a new infarction on the right side of the pons (as shown in Figure 3). Subsequently, a cerebral angiography was performed in the neurology department, which indicated a lack of visualization of the left vertebral artery (as shown in Figure 4), with the remaining cerebral arteries showing normal visualization. The diagnosis was left vertebral artery stenosis, and after conservative medical treatment, the patient’s symptoms improved, and they were discharged.
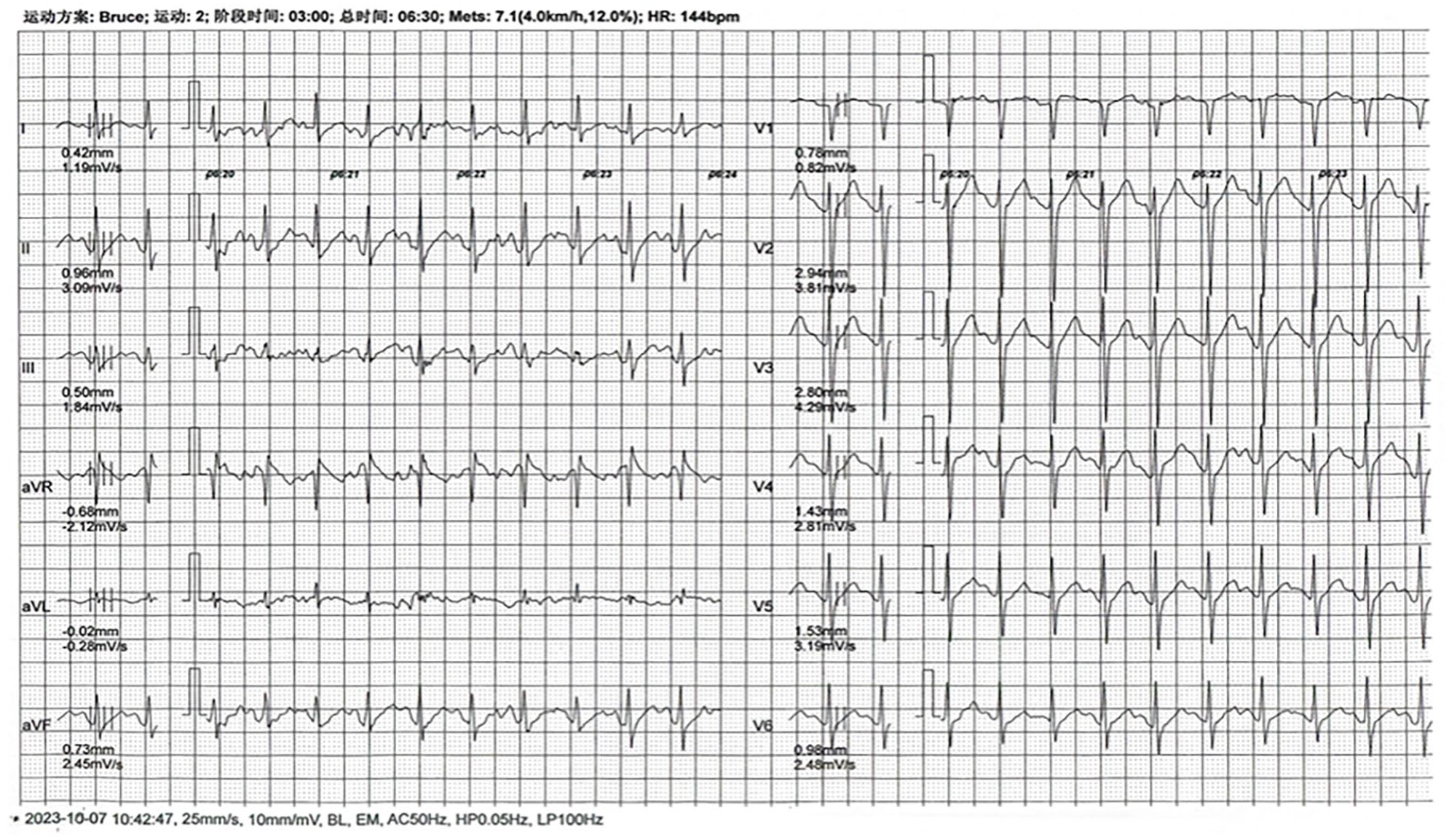
ST-T changes visible in target heart rate during exercise tablet test (indicated by lower wall lead).

A slowing heart rate can be observed, presenting with sinus arrest, and supraventricular escape rhythm during the recovery phase.
The data of exercise treadmill test.

Head MR (DWI (Diffusion weighted imaging)) shows right pontine infarction.

Whole brain angiography suggests no obvious stenosis of cerebral arteries and no enhancement of left vertebral artery. The red arrow represents the location of the lesion.
Discussion
CCS is a collective term for various cardiac dysfunctions (such as AMI, arrhythmias, myocardial ischemia, or heart failure) caused by acute brain diseases affecting the hypothalamus and brainstem autonomic nervous system. The above-mentioned lesion commonly occurs after a cerebral infarction; hence, it is also known as stroke–heart syndrome. 5 Depending on the location of the lesion, different circulatory system manifestations are exhibited. 6 When intracranial lesions stabilize or improve, cardiovascular symptoms and electrocardiographic abnormalities also improve or disappear. CCS has a high incidence and mortality rate, so it is crucial to identify high-risk patients after acute stroke, arrange appropriate cardiac monitoring, effective arrhythmia management, and prevent cardiovascular diseases to reduce morbidity and mortality rates. 7 Research has shown 8 that post-stroke cardiac dysfunction may be caused by multiple mechanisms including activation of the hypothalamic–pituitary–adrenal axis, regulation of the sympathetic and parasympathetic nervous systems, activation of catecholamines, intestinal dysbiosis, immune response, and inflammatory reactions, as well as the release of exosomes and microRNAs.
The main cardiovascular manifestations after cerebral infarction can be divided into five major categories: ischemic and non-ischemic acute myocardial injury, manifested as asymptomatic elevation of cardiac troponin; AMI after stroke; left ventricular dysfunction, heart failure, post-stroke Takotsubo syndrome; stroke-related sudden cardiac death; changes in electrocardiogram and arrhythmias after stroke, including atrial fibrillation (AF).2,9,10 This case mainly manifests in changes in arrhythmias, where any type of arrhythmia can occur in stroke patients. Researches8,11 has shown that the most commonly observed electrocardiogram changes in the early stages after stroke are repolarization changes, including prolonged QTc interval (20%–65%), ST segment changes (15%–25%), and inverted T waves with changes in amplitude and width (2%–18%). Among these, rapid arrhythmias are more common. Some studies 12 have indicated that ischemic stroke patients are eight times more likely to develop new-onset AF than non-stroke patients, and changes in post-stroke electrocardiogram are associated with patient prognosis. Regarding the timing of cardiovascular events after cerebral infarction, research results13,14 suggest that the highest incidence of arrhythmias occurs within the first 24 h, with rates significantly decreasing over time. The peak occurrence of severe cardiac adverse events such as sudden cardiac death, myocardial infarction, and heart failure appears between days 2 and 3.9, with arrhythmias occurring more frequently in patients with existing cardiac complications and established vascular risk factors (including advanced age, hypertension, and diabetes).
In this case, the patient experienced syncope during the exercise treadmill test, with the electrocardiogram showing sinus pause and supraventricular escape rhythm, accompanied by a drop in blood pressure. Further comprehensive screening of the head and cardiovascular system revealed that the patient had newly developed pontine infarction and left vertebral artery occlusion. Based on the examination results, the possibility of CCS cannot be ruled out. However, considering the electrocardiographic and blood pressure changes during the exercise treadmill test, it is highly likely that the patient experienced a VVS episode, a common neurologically mediated fainting symptom. 15 Patients with VVS may experience a decrease in heart rate and/or blood pressure when fainting or experiencing prodromal symptoms. VVS can be classified into three main types based on the characteristics of heart rate and blood pressure changes: cardioinhibitory, vasodepressor, and mixed types. In healthy individuals, intense exercise can lead to sympathetic nervous system activation. When exercise stops, the decrease in cardiac output triggers the activation of vagus nerves through volume and pressure receptors, causing vasodilation, reduced stroke volume, and heart rate to adjust the body to a resting state. In VVS patients, due to excessive vagal tone or hypersensitivity of receptors, the negative regulatory functions of blood vessels and heart are overly strong after intense exercise cessation, leading to a sudden drop in blood pressure and/or heart rate, resulting in inadequate cerebral perfusion and fainting.16,17 Some studies 18 suggest that the triggering of post-exercise fainting may also be related to changes in cerebral blood flow, with a higher likelihood of occurrence in patients with inherent narrowing of cerebral arteries. Research has shown that exercise-induced VVS often occurs in male patients, with a wide age range, particularly higher proportions in cases below 50 years of age, and can be triggered at any time after activity.
In summary, the patient experienced a mixed-type VVS during the exercise treadmill test, with cerebral infarction or vertebral artery occlusion possibly contributing to exacerbating the syncope episodes. Review of previous studies reveals that while VVS accounts for 40%–50% of syncope cases in the general population, post-stroke patients exhibit a three-fold higher risk of VVS with atypical electrocardiographic patterns. The ECG findings mentioned above also cannot be ruled out as a secondary manifestation of CCS. This case uniquely demonstrates the mechanistic overlap between VVS and cerebrovascular injury. While bradyarrhythmia observed during ETT is commonly ascribed to cardioinhibitory VVS, the concomitant pontine infarction likely impaired brainstem cardio-regulatory nuclei, thereby rendering the patient more susceptible to excessive vagal activation. In clinical practice, VVS is a common presentation during the exercise treadmill test, and some patients with post-exertional syncope may overlook the possibility of underlying cerebrovascular or cardiovascular diseases while considering VVS. This case provides mechanistic insights into how cerebrovascular injury may augment neurogenic syncope pathways. This report highlights the need for integrated neurological-cardiac assessments in stroke patients with exercise-induced syncope. The novel finding of pontine infarction exacerbating VVS-induced bradycardia challenges traditional diagnostic algorithms, advocating for routine brain imaging in syncope evaluation, particularly in high-risk populations. Future research should investigate whether brainstem lesions modulate cardiovascular reflexes, potentially identifying new therapeutic targets for preventing syncope-related morbidity in stroke.
Conclusion
This case highlights the complex interplay between cerebrovascular and cardiovascular dysfunction in a patient presenting with exercise-induced syncope. The findings suggest a mixed-type VVS triggered by intense physical activity, potentially exacerbated by underlying pontine infarction and vertebral artery occlusion. While electrocardiographic abnormalities and hemodynamic changes observed during the exercise treadmill test could reflect secondary manifestations of CCS, the clinical context strongly supports VVS as the primary mechanism. This case underscores the importance of comprehensive multidisciplinary evaluation in stroke patients with syncope, as VVS may coexist with cerebrovascular or cardiac pathologies. Clinicians should remain vigilant for overlapping etiologies to optimize management and reduce morbidity. Future studies are needed to clarify the prognostic implications of VVS in stroke populations and refine diagnostic algorithms for such complex cases.
Footnotes
Ethical considerations
Ethical approval for this study was obtained from Aerospace Center Hospital: 20231025198.
Consent to participate
Written informed consent was obtained from all subjects before the study.
Consent for publication
Written informed consent was obtained from the participant/patient(s) for the publication of this case report.
Author contributions
Zihan Zhao: conceptualization, data curation, writing (original draft). Ranran Wang: formal analysis, methodology, validation. Lihua Gao: formal analysis, methodology, validation. Meijing Zhang: conceptualization, methodology, writing (review and editing).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
