Abstract
Dasatinib is an effective agent in the treatment of bcr-abl positive acute lymphoblastic leukemia and chronic myeloid leukemia cases. Various skin reactions may occur when tyrosine kinase inhibitors are used. The severity of side effects increases in phototoxic reactions with sunlight. In this case, a phototoxic rash that developed in a 78-year-old bcr-abl positive acute lymphoblastic leukemia patient is presented. This case supports that phototoxic reactions may occur with dasatinib and emphasizes that clinicians should be alert for this side effect.
Introduction
Dasatinib is an effective agent in the treatment of bcr-abl positive acute lymphoblastic leukemia (ALL) and chronic myeloid leukemia (CML) cases. 1 Tyrosine kinase inhibitors can cause edema, rash (macular and papulopustular), and phototoxic reactions. 2 In phototoxic reactions, unlike other skin reactions, the severity of the side effect increases with visible light or ultraviolet light. 3 A study reported that dasatinib may cause photosensitization through radical-mediated damage. 4 In this case, edema and phototoxic rash that developed on the periorbital area during dasatinib use are presented.
Case report
A 78-year-old patient presents with night sweats and weight loss. In the peripheral blood count leukocyte 196 × 103/µl, hemoglobin 8.5 g/dl, and platelet 63 × 103/µl were detected. In her anamnesis, she had a history of leukocytosis. Twenty-five percent of leukocytes were blast cells in the peripheral smear. It was observed that there was a 70% blast cell population in the bone marrow biopsy, and immunohistochemical findings favored pre-B ALL.
In the examination of genetic tests, bcr-abl p210 was detected positive. The value of IS-NCN (bcr-abl transcript percentage) was 62%. EWALL (European Working Group for Adult ALL) induction regimen (600 mg imatinib on days 1–28, 2 mg vincristine on days 1, 8, 15, and 22, 20 mg dexamethasone on days 1, 4, 8, 11, 15, 18, 22, and 25, intrathecal 15 mg methotrexate, 40 mg cytarabin and 12 mg dexamethasone on days 8,15, and 22 (day 1 intrathecal procedure was not performed due to thrombocytopenia)) was started. At the end of induction chemotherapy, the blast cell rate in bone marrow was 30% and the p210 IS-NCN value was 5.3%. It was planned to continue with dasatinib instead of imatinib to improve molecular response.
EWALL consolidation first cycle (100 mg dasatinib on days 1–28, 500 mg/m2 methotrexate IV on day 1, and 5000 IU/m2
EWALL consolidation second cycle (100 mg dasatinib on days 1–28, 500 mg/m2 cytarabine on days 1, 3, and 5) was given. At the end of this consolidation, the patient’s stool was slightly black. It was thought that this situation might be due to gastrointestinal bleeding associated with dasatinib. Therefore, it was planned to switch tyrosine kinase inhibitor from 100 mg dasatinib to 600 mg imatinib.
Then, EWALL consolidation third cycle (600 mg imatinib on days 1–28, 500 mg methotrexate IV on day 1, and 5000 IU/m2
EWALL consolidation fourth cycle (100 mg dasatinib on days 1–28, 500 mg/m2 cytarabine on days 1, 3, and 5) was started. On the fourth day of the protocol, swelling began in both periorbital areas, and on the fifth day, redness was added to the swelling (Figure 1).

Dasatinib-related edema and phototoxic rash.
Management and outcome
The patient was wearing a hijab. Therefore only her eyes were exposed to sunlight. Sunlight is considered to trigger the rash around the edema area. No rash was detected elsewhere on the body. After performing the NARANJO algorithm, eyelid edema and possible phototoxic rash were thought to be related to dasatinib (Table 1). 5 The patient was consulted by the dermatology department and was considered as a phototoxic drug reaction on the eyelid. Sun protection was recommended and a topical steroid cream containing mometasone furoate was given.
Naranjo adverse drug reaction probability scale.
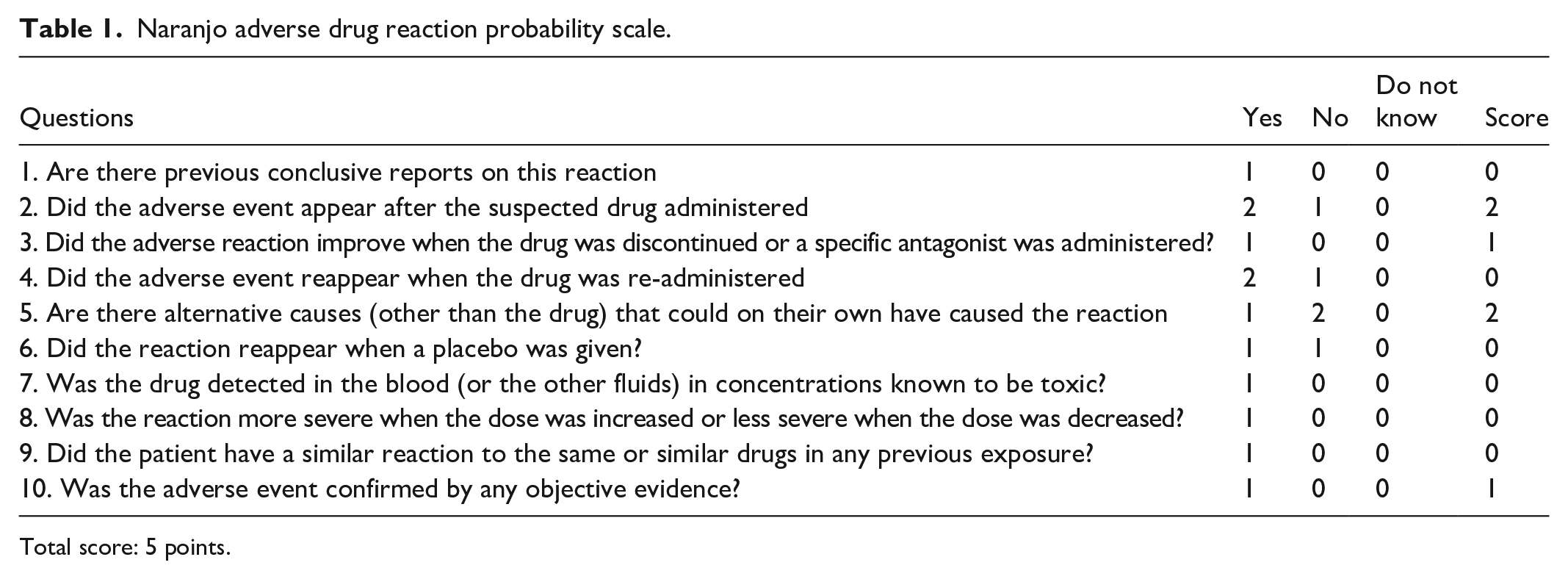
Total score: 5 points.
Following the discontinuation of the drug with sun protection, the rash significantly regressed after 8 days of medical treatment. Then topical steroid treatment was discontinued. At the end of the fourth cycle, the blast rate in the bone marrow aspiration was <5% but there was an increase in the p210 transcript level in the third cycle compared to the second cycle. Ponatinib was not available. For these reasons, the patient was started on dasatinib, but the dose was determined as 50 mg. The same side effects were not observed in the outpatient clinic follow-ups after discharge. Our patient, who was admitted to intensive care due to recurrent disease and neutropenic fever after a certain period of remission, died due to septic shock.
Discussion
Phototoxic drug reactions may occur with various tyrosine kinase inhibitors. A literature review of phototoxic drug reactions seen with tyrosine kinase inhibitors is provided in Table 2.
Review of phototoxic drug reactions with tyrosine kinase inhibitors.

TKI: tyrosine kinase inhibitor.
Phototoxic reactions reported in the literature with tyrosine kinase inhibitors are, as expected, in the body areas exposed to light. In our case, similarly to the literature, the development of a rash when the edematous eyelids were exposed to light suggested that this was a phototoxic reaction.
Here in, we have reported the first case of dasatinib-related phototoxic rash and periorbital edema. As seen in the literature review, phototoxic reactions have been reported at different times with the use of tyrosine kinase inhibitors. Cases developing over different periods of time suggest that phototoxic reactions are idiosyncratic. Our case and the occurrence of phototoxic reactions in the literature review emphasize the importance of sun protection measures in patients using tyrosine kinase inhibitors and developing skin rash. In addition, this case in which dasatinib-related periorbital edema and phototoxic rash were observed together supports that this side effect should be kept in mind for tyrosine kinase inhibitors used in the treatment of ALL and CML.
