Abstract
Acute lymphoblastic leukemia is the most common childhood malignancy. Despite many advances in therapy, about 15%–20% of children with acute lymphoblastic leukemia experience a disease relapse. Isolated ocular relapse is relatively rare. A 14-year-old male with T-cell acute lymphoblastic leukemia in remission presented with sudden onset of right eye pain and visual acuity impairment. Fundoscopic examination of the eye and magnetic resonance imaging of the orbits were consistent with optic nerve infiltration. The patient was treated with salvage chemotherapy, orbital radiation and eventual bone marrow transplantation, with notable improvement in vision and regression of retinal and optic nerve findings. Optic nerve infiltration represents an ophthalmic emergency and requires urgent management. The use of radiation therapy is a helpful adjunct with systemic chemotherapy in obtaining disease remission.
Introduction
Acute lymphoblastic leukemia (ALL) is the most common childhood malignancy. 1 Despite many advances in therapy, ALL remains an important cause of morbidity and mortality in children and adults. About 15%–20% of children with ALL experience a disease relapse.2,3 The most important prognostic factors for determining post-relapse survival rate are the relapse site, duration of first complete remission (CR) and disease phenotype. 4 Relapses occur in bone marrow in 50%–60% of cases. Other sites of recurrence include central nervous system (CNS) in approximately 20%, isolated testicular relapse in approximately 5% and a combination of marrow and extramedullary disease in the remainder. A variety of ophthalmic complications may occur in patients with leukemia. These ocular manifestations can be due to a direct cancerous infiltration, or they can be indirect and linked to hematological disorders such as anemia, thrombocytopenia and coagulopathy, opportunistic infections or related to medical therapy. 5 Primary ocular infiltration can involve orbit, anterior segment, uvea and the optic nerve. 6 We describe the case of a pediatric patient with optic nerve infiltration as initial manifestation of ALL relapse.
Case report
A 14-year-old male with no significant past medical history was diagnosed with high-risk T-cell ALL in October 2018. At the time of diagnosis, complete blood count showed a leukocyte count of 533,100/µL. T-cell ALL was documented upon ensuing immunophenotypic studies (the leukemic cells were positive for CD7, CD34, CD5, CD2 and CD1a and negative for cytoplasmic MPO, CD19, CD10, CD45RA, TCRgd, TCRab, CD33, CD56, CD20, CD22 and CD66c). Brain magnetic resonance imaging (MRI) and testicular ultrasound were normal, while the abdominal ultrasound showed splenomegaly. Patient’s induction therapy started according to the modified Berlin-Frankfurt-Münster (BFM) protocol (prednisone, vincristine, daunomycin and L-asparaginase) and then, continued from November 2018 according to the Associazione Italiana di Ematologia-Oncologia Pediatrica (AIEOP) ALL 2009 protocol. 7 The bone marrow smear on day 7 of induction revealed a total of 45% of blasts. CNS prophylaxis was carried out with intrathecal methotrexate and holocranial radiotherapy (total dose: 12 Gy). The treatment was well tolerated, and bone marrow examination on day 33 revealed CR, with negative minimal residual disease (MRD). Twelve months after documentation of remission, the patient presented to the emergency department after having acute onset of unilateral diplopia, scotomas and pain of right eye.
Investigations
Physical examination of the chest, heart and abdomen was unremarkable. Ocular motility was normal. Laboratory tests showed the following results: white blood cell (WBC) count, 1320/µL; neutrophil rate, 14.4%; lymphocyte rate, 61.4%; monocyte rate, 24.2%; hemoglobin count, 9.6 g/dL and platelet count, 39,000/µL. Cerebral and orbital MRI showed a thickening of the right optic nerve with enhancement after contrast, but no other CNS involvement (Figure 1). On ophthalmologic evaluation, visual acuity was 6/20 in the right eye and 20/20 in the left eye. Fundus examination of the right eye revealed optic disk edema, tortuosity of the retinal vessels and hemorrhages (Figure 2). The rest of ocular examination was normal. Serological testing for Toxoplasma, Bartonella, HIV, parvovirus, Cytomegalovirus, Borrelia, Rickettsia and Herpes virus 1 and 2 were negative. A lumbar puncture was performed but no malignant cells were detected in the cerebrospinal fluid (CSF), while the bone marrow examination revealed 40% blast cells. Based on these findings, the patient was diagnosed with a recurrence of ALL with CNS involvement.

(a) MRI T1-weighted brain and orbits. Surrounding the right optic nerve, pronounced gadolinium uptake can be seen. (b) MRI orbit scan repeated after completion of therapy. The right optic nerve is no longer thickened.

(a) Fundus photographs at initial presentation. Right eye and left eye, respectively. The left eye was normal, while the right showed significant disk edema, tortuosity of the retinal vessels and hemorrhages. (b) Fundus photographs of the right and left eye, 3 months following bone marrow transplant. Resolution of disk edema can be noted.
Treatment and outcome
The patient was treated with chemotherapy according to the high-dose cytarabine and mitoxantrone (HAM) scheme (cytarabine 6 g/m2 on days 1–4 and mitoxantrone 10 mg/m2 on day 4) together with radiotherapy targeted to the right optic nerve (total dose: 24 Gy in 8 fractions, spread over 2 weeks). Bone marrow transplant was programmed. Evolution was marked by an improvement of visual acuity to 12/20 in the right eye, with a regression of edema at fundus examination. The treatment was well tolerated, and the patient had no ocular/orbital side effects after radiotherapy. In May 2020, the patient underwent allogeneic hematopoietic stem cell transplantation from an human leukocyte antigen-haploidentical family donor (39-year-old patient’s mother) with infusion of T-regulatory (T-reg) cells as prophylaxis of graft-versus-host disease (GvHD). The conditioning regimen was based on cyclophosphamide (15 mg/kg for 2 days), thiotepa (4 mg/kg for 2 days), fludarabine (40 mg/m2 for 4 days) and total body irradiation (total dose: 13.5 Gy). During all the follow-up, the patient remained in continuous complete morphological/molecular remission with 100% CD34+ donor chimerism.
Discussion
Relapse is the main reason for treatment failure in childhood ALL. The most common sites of extramedullary relapse are the CNS and skin in acute myeloid leukemia (AML), and CNS and testis in ALL. 8 Relapse of childhood ALL involving the eye is a rare event and represents only 2.2% of ALL relapses. 9 Although the retina is the most common site of ocular manifestations of leukemia, all parts of the eye can be involved either prior to systemic diagnosis or during the disease course. Optic nerve involvement occurs more frequently in acute compared with chronic leukemia, in children compared with adults and in ALL compared with AML. 10 The cases of ALL relapse with optic nerve infiltration in pediatric age are reported in Table 1. Leukemic infiltration of the optic nerve can manifest as blurred vision, photophobia and ocular pain, progressing to occlusion of the central retina artery and central retinal vein if not treated properly.
Published studies of ALL relapse presenting with optic nerve infiltration in pediatric patients.
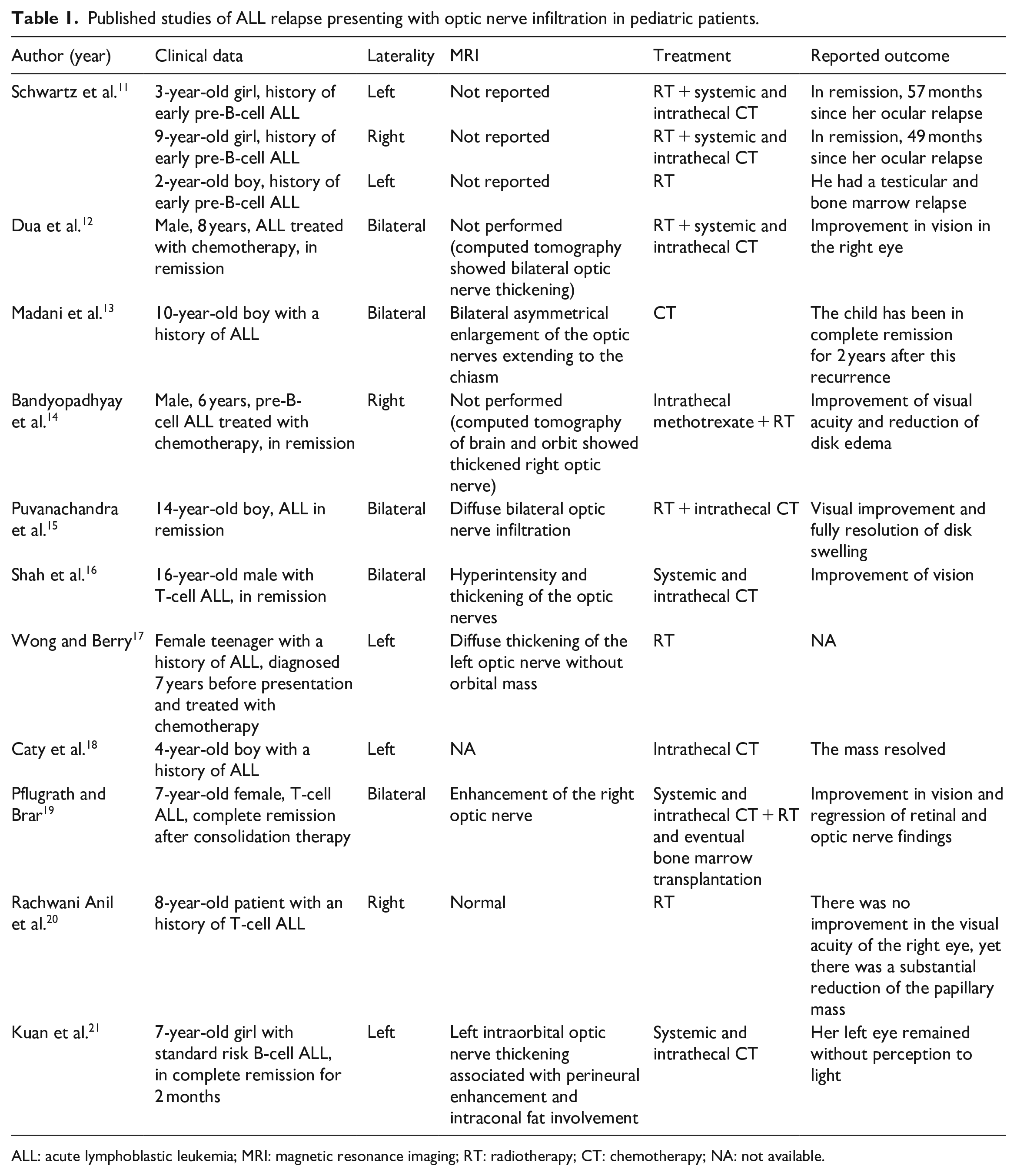
ALL: acute lymphoblastic leukemia; MRI: magnetic resonance imaging; RT: radiotherapy; CT: chemotherapy; NA: not available.
Despite survival rate of children with ALL significantly improved over the last decades, management of ocular involvement in ALL is a challenging problem; prompt diagnosis and early treatment are required to preserve the patient’s vision. Indeed, the eye is considered as a pharmacologic sanctuary, 22 which may not be treated adequately by systemically administered drugs. Given the limitations of chemotherapy, targeted radiotherapy along with systemic therapy is probably the best approach. However, radiotherapy may induce cataracts or ptosis, either of which may require future surgical intervention. 23
Optic neuropathy in the context of leukemia has a broad differential diagnosis, including infiltration, infections, compression, vasculitis, radiotherapy-induced complications or adverse effects of chemotherapeutic agents. All these possibilities should be excluded through careful investigations. A lumbar puncture must be performed in every case to aid in this differentiation. Leukemia and chemotherapy can weak the immune system and lead to opportunistic infections. Multiple viral and bacterial diseases can present as optic neuritis. Common infections include syphilis, tuberculosis, Lyme disease, Bartonella, herpetic disease, West Nile virus and HIV. 24 In leukemic infiltration of the optic nerve, fundus examination usually reveals optic disk edema and diffuse hemorrhages, while is normal in other patients. 25 Radiological and ophthalmologic evaluations are effective modalities to confirm the diagnostic suspicion. MRI may show thickening of the optic nerve or be normal. 26 Camera et al. 27 proposed ultrasound as a study method, while other authors diagnosed a leukemic relapse on the optic nerve using the fine needle aspiration biopsy (FNAB). 28 A review by Myers et al. 29 found that malignant cells are identified in the CSF in a majority of patients with leukemic infiltration of the optic nerve. Vitreous biopsy may be helpful in accurate diagnosis when CSF analysis is negative for leukemic cells. 30
In our patient, diminished visual acuity together with radiological findings, in absence of signs of cranial hypertension and negativity of immunological and infectious tests, led us to the diagnosis of leukemic optic nerve infiltration. Although in this case the patient achieved a second CR, the prognosis for patients with ocular involvement is generally poor. Routine ophthalmologic screening may antedate the diagnosis of relapse and positively impact on prognosis.
Conclusion
Although leukemic infiltration of the optic nerve is a rare event, clinicians should be aware of this possible site of relapse. Ocular symptoms should be investigated without delay in patients with a previous history of acute leukemia, and an early treatment is critical for visual recovery and prolonged survival.
Footnotes
Author contributions
All authors contributed to this manuscript. G.D., I.C., C.C., M.S.M., E.M., F.A. and K.P. wrote the abstract, introduction, case, discussion and conclusion. M.C., A.V., P.G. and F.A. performed critical edits and final revision—figures. The manuscript has been read and approved by all named authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
