Abstract
Endometriosis affects approximately 10% of premenopausal women, with urinary tract involvement in 1% of cases. Ureteric endometriosis is a rare, typically asymptomatic, cause of obstructive renal failure. We present the case of a 30-year-old woman with stage IV endometriosis and advanced human immunodeficiency virus presenting with acute, severe left flank pain and kidney injury. Imaging revealed severe, bilateral hydronephrosis due to extensive endometriosis deposits obstructing the urinary tract. Recurrent ureteric obstruction by infiltrating endometrial disease necessitated eight procedures (bilateral nephroureteric stent placements and nephrostomy insertions). Despite the severity of the disease surgery was successfully avoided. At the time of discharge and subsequent follow-up, the patient was clinically well with stable renal function (Creatinine 1.33–1.61 mg/dL). This case highlights the importance of considering endometriosis in the differential diagnosis of acute kidney injury in women. It demonstrates successful non-surgical management of severe ureteric disease to prevent irreversible renal damage.
Background/Introduction
Globally 10% of premenopausal women and 2–5% of postmenopausal women have endometriosis.1–3 Urinary tract involvement is reported in approximately 1% of cases; however, the true prevalence is probably higher given that up to 50% of women with histologically confirmed urinary tract endometriosis are asymptomatic.4–6 Despite its prevalence and associated morbidity, ureteric endometriosis has received limited attention within the renal literature. It is typically described as a clinically silent disease, confined to the left distal ureter.7,8 Rarely, deep infiltrating endometriosis leads to irreversible loss of renal function by urinary tract obstruction.9,10 Here we describe a case of urinary tract endometriosis with an atypical presentation, distinct radiological findings, and severe, diffuse atypical distribution of endometrial deposits.
Case presentation
A 30-year-old woman presented with a one-week history of severe, cyclic left lower quadrant pain not relieved by simple analgesia. Her past medical history included stage IV endometriosis 11 and advanced human immunodeficiency virus (HIV) infection. 12 She was on anti-retroviral therapy (ART), specifically a nucleoside reverse transcriptase inhibitor (NRTI), which she had discontinued for one month. She had recently arrived in Ireland from Africa seeking asylum. The patient is nulliparous and had been diagnosed with stage IV endometriosis elsewhere fourteen months prior to this admission. 11 At diagnosis, the severity of her endometriosis necessitated a right hemicolectomy due to bowel obstruction and bilateral ureteric stent insertion for obstructive uropathy. A levonorgestrel intrauterine device (IUD) was also inserted at that time and her endometriosis symptoms improved.
On presentation, the patient was afebrile and normotensive (BP 110/70 mmHg). Physical examination revealed left lower abdominal and costovertebral angle tenderness. Notably, she was not menstruating and had minimal urine output (<0.3 mL/kg/h). Laboratory testing indicated microcytic anaemia (Haemoglobin 9.7 g/dL), a white cell count (WCC) of 7200/mm3 and a C-reactive protein level of 59 mg/L. Serum urea and creatinine were markedly elevated (urea: 11.5 mmol/L and creatinine 4.64 mg/dL, Stage 3 acute kidney injury (AKI), KDIGO classification 13 ). Urinalysis demonstrated 37 RBC/uL and 36 WBC/uL with mixed growth on urine culture. Given the patient’s severe AKI, the risk of contrast-induced nephropathy (CIN) was weighed against the necessity of the computed tomography (CT) scan. The urgent need for accurate diagnostic information led to the decision to proceed, with appropriate precautions, including hydration and monitoring, taken to minimize risk. The CT scan showed bilateral ureteric stents in a good position with marked left hydronephrosis, an atrophic right kidney and innumerable retroperitoneal soft tissue densities.
Ureteroscopy and retrograde pyelogram revealed bilateral hydronephrosis and a right distal ureteric stricture. Non-metallic ureteric stents were replaced bilaterally. The patient was also restarted on antiretroviral therapy. At this time, her CD4 count was 400 cells/mm3 and her HIV viral load was detectable (17,239 copies/mL).
Investigations were completed to characterize the severity of her endometriosis and associated chronic kidney disease. A dimercaptosuccinic acid (DMSA) renal scan revealed an atrophic right kidney contributing 14% of function and the left kidney contributing 86% of function. Pelvic magnetic resonance imaging (MRI) demonstrated deep infiltrating endometriosis and uterine adenomyosis markedly distorting the urinary tract anatomy (shown in Figures 1–3). The distal right ureter coursed in an abnormal medial location and adhered to the lateral uterine wall. The distal left ureter travelled posterior and inferior to a 3.4 × 4.3 × 6 cm left adnexal cyst, closely adhering to the cyst wall. There was a loss of the normal fat plane between the uterus and bladder.

Coronal MRI image of the pelvis and abdomen showing diffuse urinary tract endometrial deposits with left ureter (arrow).

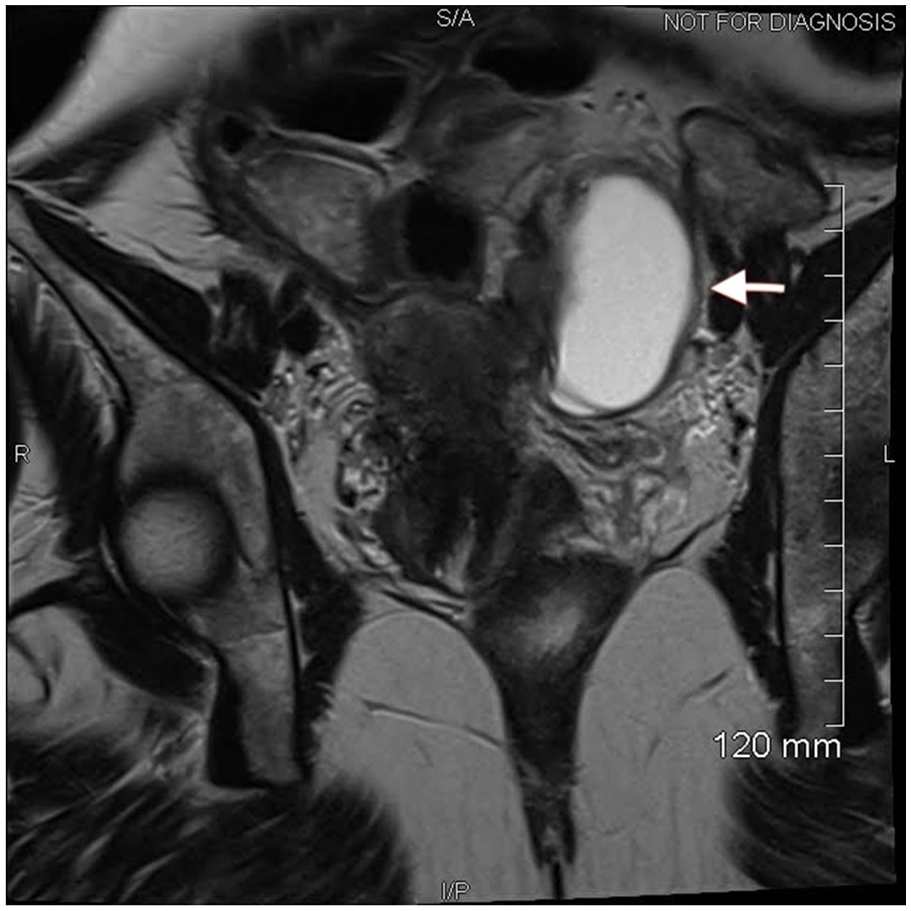
Axial pelvic MRI showing ureteric tract obstructing endometriosis (white arrows).
Axial pelvic MRI image showing T1 hyper nodule in a left endometrioma (arrow).
Given the severity of this patient’s deep infiltrating endometriosis, her case was discussed at a gynaecology multidisciplinary conference, and she was started on three monthly Decapeptyl (triptorelin pamoate) injections.
The patient’s obstructive ureteric endometriosis was severe and refractory in nature resulting in multiple recurrent episodes of AKI and urinary tract infection throughout a long hospital admission. Over an eight-week period, recurrent urinary tract obstruction by endometrial deposits resulted in four distinct episodes of AKI secondary to recurrent ureteric tract/obstruction confirmed by radiological imaging showing recurrent hydronephrosis. Recurrent obstruction with associated AKI necessitated a total of six procedures all utilizing non-metallic stents: initially a retrograde bilateral stent replacement, subsequently left nephrostomy insertion, two subsequent anterograde left stent insertions, a retrograde right stent insertion and a final left nephrostomy insertion.
In addition, ureteric obstruction resulted in two instances of complex urinary tract infection with sepsis necessitating two long courses of intravenous antibiotics and two further procedures: left retrograde stent removal and right ureteric stent replacement.
Following this complicated course of recurrent urinary tract obstruction with associated AKI/infection, the patient’s condition quiesced and she remained stable for 7 days. To decrease infection risk and nephrostomy dislodgment on discharge, it was planned to replace a left percutaneous nephrostomy with an anterograde left ureteric stent. A nephrostogram was performed via the left nephrostomy with the intention to proceed to anterograde nephroureteric stent insertion. Unexpectedly, the left collecting system was patent without ureteric stents, leading to the removal of the left nephrostomy.
At the time of discharge, the patient was clinically well with stable renal function (creatinine 1.61–2.06 mg/dL). At three months follow-up, her renal function remained stable (creatinine 1.33 mg/dL).
Discussion
This case presents a rare and unique instance of bilateral ureteric endometriosis, resulting in more severe obstruction on the right side with a right atrophic kidney. This was successfully managed with a combination of hormonal therapy and minimally invasive procedures, diverging from the typical surgical approach.
Endometriosis affects 176 million women of reproductive age globally, with approximately 1% of cases involving the urinary tract.3,14 Ureteric deposits are exceedingly rare, occurring in only 2% of cases of urinary tract endometriosis.4,10
Typically, ureteric endometriosis is asymmetrical with the distal left ureter predominately affected due to its close anatomical relationship to female reproductive organs.7,8 Our case is unique, showing extensive bilateral disease with severe, predominately right-sided obstruction and associated atrophic right kidney with minimal residual renal function (14%). 15 This demonstrates that bilateral hydronephrosis can be precipitated by endometriosis and underscores the need for nephrologists to infiltrate endometriosis in their differential diagnosis of AKI in women.
Ureteric endometriosis is difficult to diagnose due to the non-specific presentation of symptoms such as dysmenorrhoea, dyspareunia, pelvic pain, urinary urgency, frequency, subfertility/infertility or lumbar pain.7,16 In addition, many cases are asymptomatic, presenting with advanced renal failure or unilateral loss of renal function discovered incidentally.7,10 In contrast to these reports, our patient presented with acute severe, left flank pain and associated nausea.
This condition is frequently unsuspected leading to delays in diagnosis. Routine surveillance of renal function is recommended in all patients with endometriosis to identify obstructive uropathy. Nephrologists should include urinary tract endometriosis in their differential for abnormal renal function in women with suggestive symptoms and obstructive features. 10
The diagnosis of ureteric endometriosis is challenging, with a presumptive diagnosis occurring in less than 50% of cases and imaging often failing to accurately characterize the severity and extent of endometrial deposits.6,7 Timely identification requires knowledge of the variability of radiological appearances, as highlighted by this case. Ultrasonography, including both transvaginal and pelvic abdominal approaches, is the preferred initial imaging modality due to its superior sensitivity compared to CT and MRI, 17 as well as its accessibility and safety. MRI, however, is advantageous for accurately assessing morphological abnormalities and evaluating the distal ureters. 18 Our case contrasts with previous reports due to the extent of the deep infiltrating endometriosis and the resulting urinary tract obstruction by large innumerous endometrial deposits shown on imaging.
Management of ureteric endometriosis depends on disease severity, symptom burden, desire for fertility, and complications including renal impairment/recurrent infection. Treatment options include hormonal therapies (danazol or gonadotropin-releasing hormone agonists), interventional radiology (IR) procedures (ureteric stenting), or surgery (distal ureterectomy, ureteral implantation or nephrectomy). 19 Surgery is classically the treatment of choice for severe disease.5,10,19 We report a case of severe disease with obstructive uropathy and AKI where surgery was avoided.
In this case, we chose a stepwise approach and opted for stent exchange over surgery to minimise procedures given the concurrent need for retrograde removal of existing, infected, obstructed stents.
Ureteric stent placement offered several benefits, including the minimally invasive technique, a short procedural and recovery time, and good cosmetic outcomes. 20 Drawbacks include frequent lower urinary tract symptoms, risk of recurrent obstruction, stent malposition, vesicoureteral reflux and long-term complications such as recurrent urinary tract infection, stent encrustation, migration, obstruction and repeat procedures.21,22
Surgery, ureterectomy with ureteroureterostomy or ureteral reimplantation, is classically the preferred treatment for severe ureteric endometriosis.5,10,19 The fibrotic scar tissue is thought to be unresponsive to hormonal therapy and thus, surgical resection removes the obstructive tissue, reduces the need for repeat interventions, enables excision of other endometrial deposits, and allows for concurrent hysterectomy with bilateral salpingo-oophorectomy or nephrectomy, if needed. However, given surgery is invasive, with a significant recovery time and risk of complications, including stricture, hematoma, and fistula formation.23,24 We opted for non-surgical management. Despite the severity of the disease and resulting urinary tract distortion, this case was successfully managed with hormonal therapy and non-surgical procedures (ureteric stenting and temporary nephrostomy insertion) with a favourable patient outcome with the substantial recovery of renal function. This case was peculiar due to the refractory nature and rapid recurrence of urinary tract obstruction not previously described in the literature. In addition, the satisfactory response to management with a combination of hormonal therapy, antiretroviral therapy, and minimally invasive interventions is atypical for severe disease.
In addition, there is an intriguing temporal correlation in this case: the patient's endometriosis flared during a period of ARV non-adherence and subsequently quiesced upon resumption of therapy. There is evidence in the literature implicating telomerase activity in the pathogenesis of endometriosis.25–27 ARVs with anti-telomerase properties, such as NRTIs, could hypothetically influence disease activity. 28 The patient’s response raises the possibility that medications with anti-telomerase properties, such as NRTIs, may hold potential as novel therapeutic options for endometriosis, possibly warranting further investigation. However, we recognize that this association may be coincidental and warrants caution in interpretation.
Conclusion
This case highlights the importance of recognizing endometriosis as a potential, though rare, cause of obstructive uropathy in women. It underscores the value of early diagnosis and prompt intervention to prevent irreversible renal damage, particularly in atypical presentations. The favourable outcome in this case, achieved through non-surgical management and hormonal therapy, demonstrates the potential for minimally invasive approaches in severe cases of urinary tract endometriosis. Early multidisciplinary involvement remains crucial to optimizing outcomes in complex cases such as this.
Footnotes
Acknowledgements
None.
Author Contributions
C.R. and L.R. contributed to the conception, acquisition and analysis of data for the case report. M.B. contributed to the acquisition and interpretation of data for the case report. C.R. drafted the case report. L.R. and M.B. critically reviewed the case report. The final version of the case report was approved by all three authors. All authors are accountable for all aspects of the case report.
Data Availability Statement
Data of the case report is not publicly available on ethical grounds.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
