Abstract
Remimazolam, a short-acting benzodiazepine approved for use in general anesthesia in Japan in 2020, has cardiovascular stability superior to conventional anesthetics. Anesthesia management using remimazolam of a patient with impaired cardiac function and several complications is reported. A man in his 50s with hypertension, diabetes mellitus, and chronic kidney disease (G 5) on dialysis came to our hospital with a chief complaint of exertional dyspnea and chest tightness, and a close examination showed stenoses of three coronary arteries. The patient was managed under anesthesia using remimazolam as a sedative because anesthetics could easily induce circulatory failure. Cardiopulmonary bypass was initiated without causing any circulatory failure, and coronary artery bypass grafting and aortic valve replacement were performed. The patient was successfully weaned with a small dose of catecholamine. Remimazolam was selected for this patient because of its minimal effect on cardiac rhythm, and the anesthesia was safely administered.
Introduction
Patients undergoing cardiac surgery often have decreased cardiac function and poor vascular condition, requiring more meticulous perioperative hemodynamic management than general surgical procedures. For anesthesiologists, it is essential to closely monitor preoperative assessments, such as echocardiograms and blood test data, to comprehensively understand the patient’s overall condition. Similarly, anesthesia management demands careful selection and dosing of medications. Remimazolam, a sedative available in Japan since 2020, is currently used for general anesthesia in Japan, South Korea, and China. This drug, a short-acting benzodiazepine with a short half-life, exerts minimal impact on hemodynamics. 1 There have also been reports suggesting that remimazolam protects against intraoperative pulmonary inflammation compared with conventional anesthetics. 2 A case in which anesthesia management with this drug was successfully performed without significant complications in a patient with decreased cardiac function and concurrent COVID-19 infection is reported.
Case
The patient was a man in his 50s, standing 164 cm tall and weighing 68.2 kg. He presented to a local clinic with a chief complaint of dyspnea and was referred to our hospital with suspected acute coronary syndrome. An electrocardiogram showed ST elevation in the precordial leads (V1–V3) (Figure 1). An echocardiogram indicated a decreased left ventricular ejection fraction (LVEF) of 26% and diffuse hypokinesis, prompting coronary angiography (CAG). CAG showed triple-vessel disease (#4PD 75%, #7 90%, #8 90%, #9 75%, #11 99%, #12 99%, #13 90%), leading to planned coronary artery bypass grafting (CABG). Given the LVEF of 26% and moderate aortic regurgitation, the decision was made to proceed with aortic valve replacement (AVR) and CABG under cardiopulmonary bypass. The patient’s medical history included hypertension, diabetes mellitus, and chronic kidney disease (G 5) requiring dialysis. Two days before surgery, the patient developed a fever, cough, and sore throat and was diagnosed with COVID-19. Although postponing the surgery was considered, elevated brain natriuretic peptide levels (from 699 to 1682 pg/mL) suggested an increased risk of heart failure triggered by the infection, necessitating proceeding with the surgery. Preoperative evaluations indicated a high likelihood of hemodynamic instability during anesthesia induction. Consequently, remimazolam, which has minimal circulatory suppression, was selected for anesthesia management. On entering the operating room, the patient’s blood pressure was 104/70 mmHg, heart rate was 87 bpm, and SpO2 was 98% (O2 3 L/min, nasal cannula). Anesthesia induction was achieved with 3 mg of remimazolam, 150 µg of fentanyl, remifentanil at 0.03 µg/kg/min, and 50 mg of rocuronium. Maintenance included remimazolam at 0.5 mg/kg/h, remifentanil at 0.1 µg/kg/min, and rocuronium at 20 mg/h. Intubation was performed using an 8-mm endotracheal tube. Decreases in blood pressure were managed with 0.05 mg of phenylephrine as needed. Intraoperative monitoring included invasive arterial pressure, SpO2, central venous pressure, Patient State Index (PSI) (Sedline®, Masimo, Irvine, CA, USA), regional cerebral oxygen saturation, and transesophageal echocardiography. Continuous cardiac output monitoring was performed using the FloTrac™ Sensor (Edwards Lifesciences Japan Ltd., Tokyo, Japan), which calculates stroke volume and cardiac output (cardiac index) via arterial waveform analysis. From induction to the initiation of cardiopulmonary bypass, the cardiac index ranged between 2.4 and 3.0 L/min/m2 (Figure 2). Nicorandil, a coronary vasodilator, was started at 4 mg/h at induction. Cardiopulmonary bypass was initiated without significant hemodynamic instability, and AVR and CABG were performed using left internal thoracic artery (LITA) and great saphenous vein (SV) grafts. The anastomoses included LITA-LAD, Ao-SV-OM, and Ao-SV-4PD. The prosthesis used was the 25mm INSPIRIS aortic valve (INSPIRIS; Edwards Lifesciences Corporation, Irvine, CA, USA). On weaning from cardiopulmonary bypass, dopamine (3–5 µg/kg/min), dobutamine (3–5 µg/kg/min), and norepinephrine (0.1 µg/kg/min) were infused continuously. Post-weaning, the hemodynamics remained stable, allowing the gradual reduction of dopamine and dobutamine to 2 µg/kg/min each and the cessation of norepinephrine by the end of surgery. Remimazolam was continuously infused at 0.5 mg/kg/h post-weaning. Depth of anesthesia was monitored using the PSI, with values maintained between 20 and 40. The valve function after the prosthesis was implanted was normal. Anesthesia duration was 9 h and 52 min, surgical duration 7 h and 49 min, and cardiopulmonary bypass time 2 h and 39 min. The total fluid intake was 3050 mL, blood loss was 3226 mL, and transfusions included 12 units of packed red blood cells, 14 units of fresh frozen plasma, and 20 units of platelets. The patient was extubated on the third postoperative day in the intensive care unit. Although the patient’s condition was stable, unfortunately, 150 days after the surgery, he passed away from pneumonia unrelated to the current disease in the hospital.
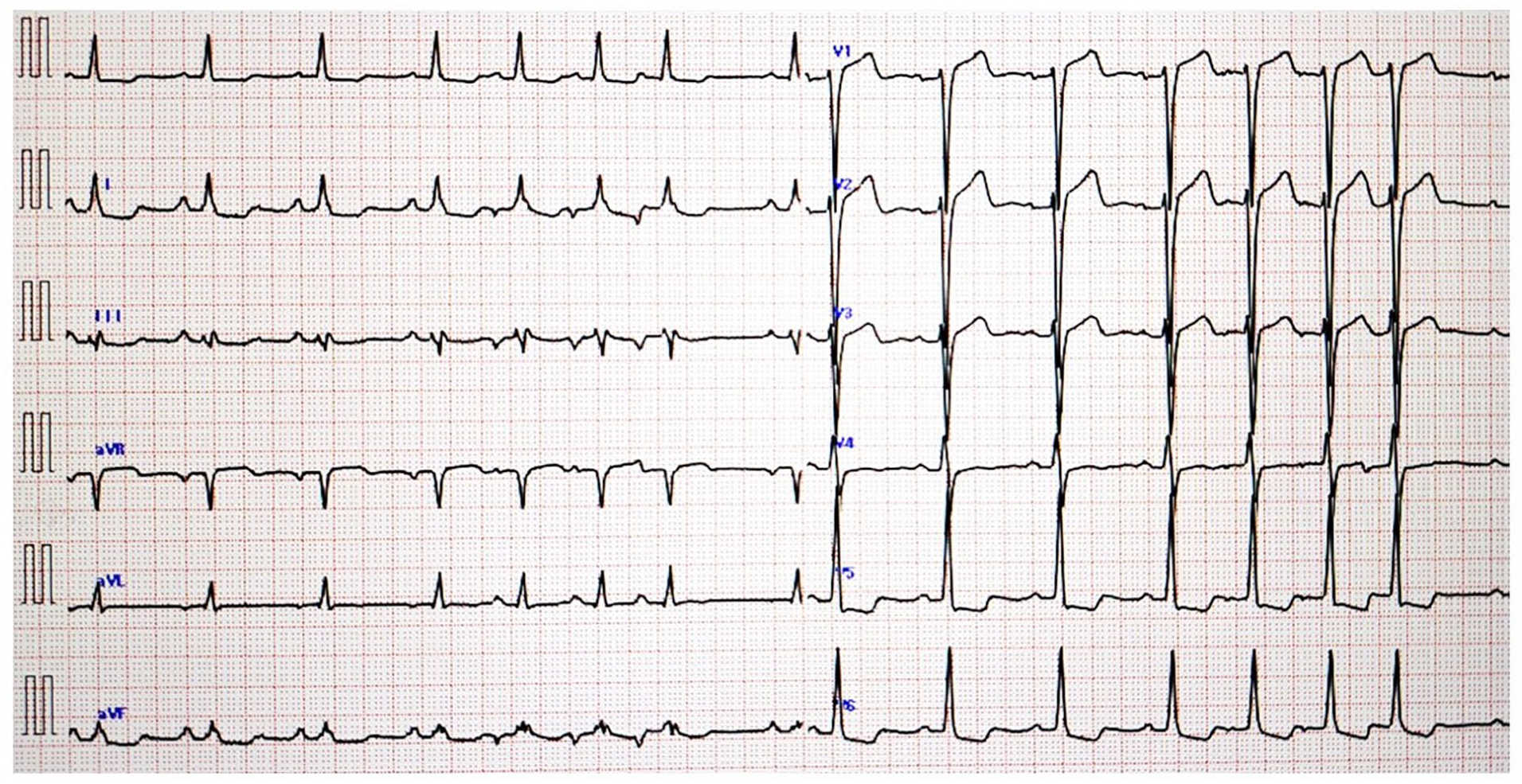
Electrocardiogram.
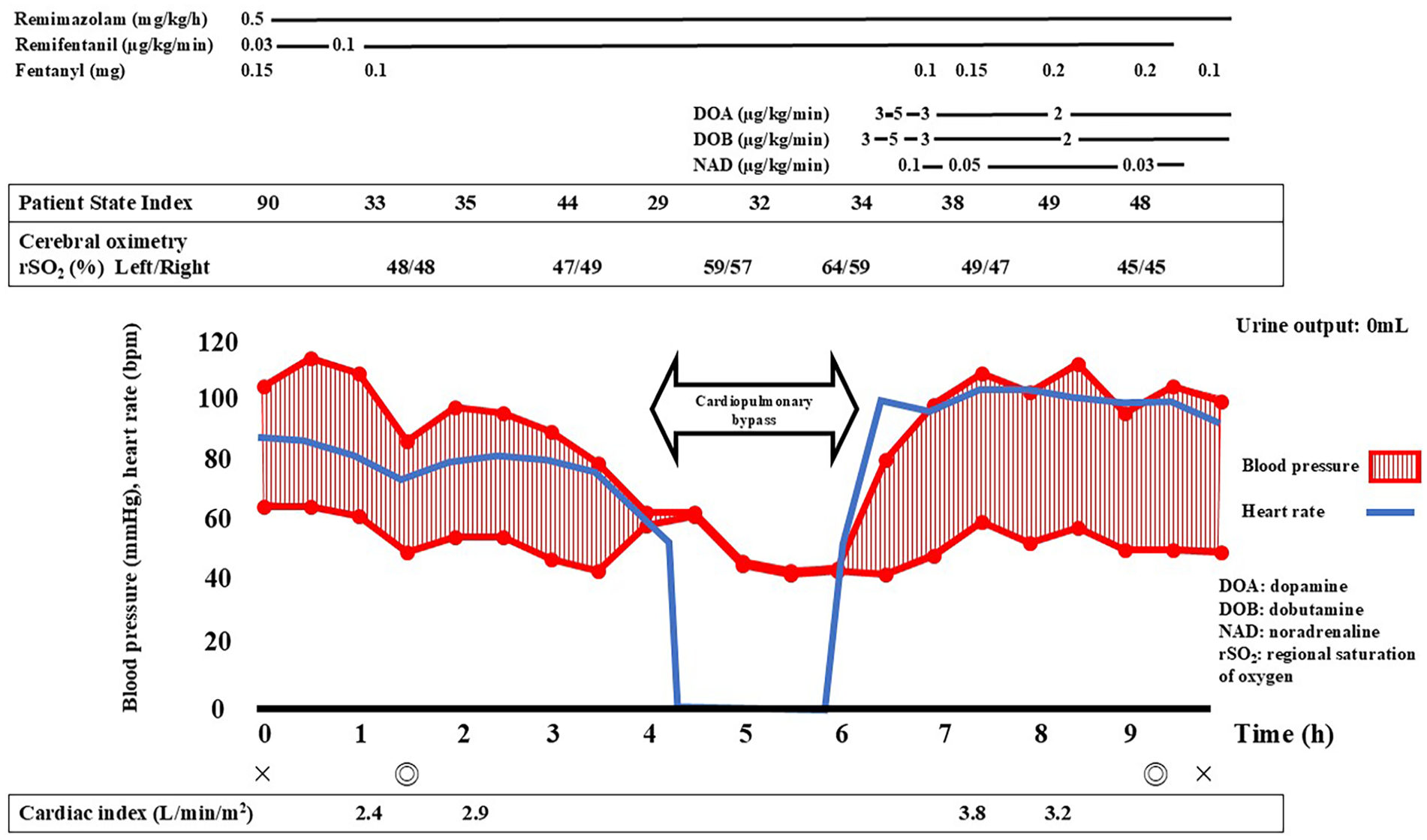
Patient’s anesthesia record.
Discussion
This case involved a patient with severe cardiac dysfunction likely due to coronary artery stenosis, making hemodynamic instability a significant concern with the use of anesthetics. Therefore, remimazolam, a sedative with minimal circulatory suppression, was chosen for anesthesia. Remimazolam has several advantageous characteristics, including minimal impact on hemodynamics, a short half-life, and the availability of a reversal agent. 1 In general, inhalational anesthetics such as sevoflurane and desflurane, commonly used for anesthesia maintenance, have strong vasodilatory effects. 3 Comparative studies between inhalational anesthetics and remimazolam for anesthesia maintenance have shown that the group using inhalational anesthetics required significantly more vasopressors. 4 When maintaining anesthesia with intravenous anesthetics, previous studies have reported that remimazolam allows for more stable hemodynamic maintenance compared to traditional intravenous anesthetics such as propofol.5,6 Furthermore, remimazolam is rapidly hydrolyzed by non-specific esterases into an inactive metabolite, CNS7054, independently of liver or kidney metabolism. 7 This results in lower residual levels and shorter accumulation time in the body than propofol. Even with prolonged and high-dose intravenous administration, remimazolam does not accumulate in the body, and the activity of its metabolites remains low, facilitating patient recovery post-surgery.8,9
Maintaining anesthesia with remimazolam can present challenges in patients with long-term benzodiazepine use due to potential tolerance, necessitating careful patient selection. 10 However, this drug is particularly useful in cases of reduced cardiac function. In the present case, an initial dose of 3 mg of remimazolam was administered, followed by maintenance at 0.5 mg/kg/h, without any critical hemodynamic deterioration. Hemodynamic parameters were monitored using the FloTrac™ system, enabling complication-free anesthesia management. Hemodynamic management using FloTrac™ is reported to improve clinical outcomes in patients undergoing major surgery, making it valuable in cases such as this one. 11 Intraoperative pain management was safely achieved with continuous remifentanil infusion. Depth of anesthesia levels, monitored using the PSI, were maintained between 20 and 40. There is a strong correlation between intraoperative depth of anesthesia with remimazolam and PSI values, making PSI a useful tool for monitoring the depth of anesthesia. 12 Though remimazolam was administered at 0.5 mg/kg/h in the present case, higher sensitivity in elderly patients than in younger patients suggests the need for careful dose adjustment based on various monitoring tools. 13 In addition, midazolam, another benzodiazepine, is associated with an increased risk of postoperative delirium, requiring cautious use. 14 In contrast, remimazolam appears less likely to cause postoperative delirium, potentially broadening its application. 15 The patient in the present case did not have delirium.
Conclusion
Remimazolam, as a sedative, offers sufficient efficacy with ultra-short-acting properties, minimizing prolonged sedation, and minimal impact on hemodynamics. Given these characteristics, it could be considered a suitable option for general anesthesia in patients with decreased cardiac function and comorbidities, such as in this case, where the patient’s overall condition is prone to deterioration.
Footnotes
Acknowledgements
The authors would like to thank Forte Science Communications, Tokyo, Japan, for English language editing.
Author contributions
T.S., Y.I., and M.H. contributed to the anesthesia management of the patient, conceptualization of the case report, and writing of the original draft. T.S. contributed to the anesthesia management of the patient. Y.I., T.S., and K.B. edited the manuscript. K.O. was the overall supervisor of this case. All authors read and approved the final manuscript.
Data availability
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient’s family for their anonymized information to be published in this article.
Consent for publication
Written, informed consent was obtained from the patient’s family for publication of this case report and the accompanying images.
