Abstract
Five cases of non-neuronal granular cell tumours of the oral cavity are documented in the literature. Additionally, one case of a non-neuronal granular cell tumour with features of malignancy was described. A malignant granular cell tumour is a rare neoplasm and counterpart of a benign granular cell tumour. The cell of origin of the granular cell tumour was reported from the Schwann cell. Some granular cells originated from non-neural components and were negative for immunohistochemistry S100. Immunohistochemistry is required to confirm further and categorize ulcero-proliferative and erythematous polypoidal oral cavity lesions. These lesions can mimic squamous cell carcinoma, mucoepidermoid carcinoma and pyogenic granuloma in morphology. We are presenting a rare case of malignant granular cell tumour of non-neuronal origin on the floor of the mouth. To our knowledge, it is the first case of a malignant non-neuronal granular cell tumour.
Background
A malignant granular cell tumour is a rare neoplasm and counterpart of a benign granular cell tumour. 1 The cell of origin of the granular cell tumour was reported from the Schwann cell. 2 But some granular cells were originated from non-neural components and negative for IHC (immunohistochemistry) S100. 3 Initially, these tumours (non-neuronal/negative for S-100 were called ‘Primitive polypoid granular cell tumours’. 4
Granular cell (GC) tumours can be present anywhere in the body, but 50% are present in the head and neck region, with the most common sites being the tongue (80%) in the oral cavity, skin and soft tissue.2,5,6 The classical presentation of GC tumour is solitary, painless and 3–4 cm in size in middle-aged females. 5 The literature reported five cases of non-neuronal (S-100 negative) granular cell tumours in the oral cavity.7–10 Malignant GC tumour is a rare entity, reported in approximately 1%–2% of cases and having a bad prognosis. 11 We are writing about the sixth case of a non-neuronal granular cell tumour with its malignant counterpart. The indexed study aimed to present a rare case of malignant granular cell tumour of non-neuronal origin on the floor of the mouth.
Case report
A 57-year-old male presented with ulcero-proliferative growth on the floor of the mouth for three years and came in ENT OPD. A biopsy was taken with patients’ written informed consent and sent to the histopathology department. Grossly, we received multiple soft tissue pieces, altogether measuring 1.8 × 1.2 × 0.5. A microscopic examination revealed a fragmented biopsy; most fragments showed necrosis, and a few viable fragments showed a tumour arranged in solid sheets and focal short fascicles. The tumour cells are large polygonal with round-to-oval nuclei, vesicular nuclear chromatin, mild nuclear pleomorphism and prominent nucleoli with moderate-to-abundant amounts of fine granular cytoplasm (Figure 1(a) and (b)). Few areas showed spindle cell differentiation with elongated hyperchromatic nuclei, irregular membranes, moderate nuclear pleomorphism and a scant-to-moderate amount of pale cytoplasm (Figure 1(c)). In addition, large areas of necrosis (Figure 1(d)), a few atypical (0–4)/10 HPF mitosis and apoptotic bodies were also noted. On histomorphology, the diagnosis suggested a malignant granular cell tumour. On IHC examination, the tumour cells were positive for vimentin (strongly, diffuse membranous and cytoplasmic), NSE (nonspecific enolase, diffuse cytoplasmic) and CD10 (cytoplasmic granular) (Figure 2(a)–(c)). The tumour cells were negative for S100 (Figure 2(d)), P40, CK5/6, and Pan-CK. The diagnosis confirmed a malignant granular cell tumour of non-neuronal origin.
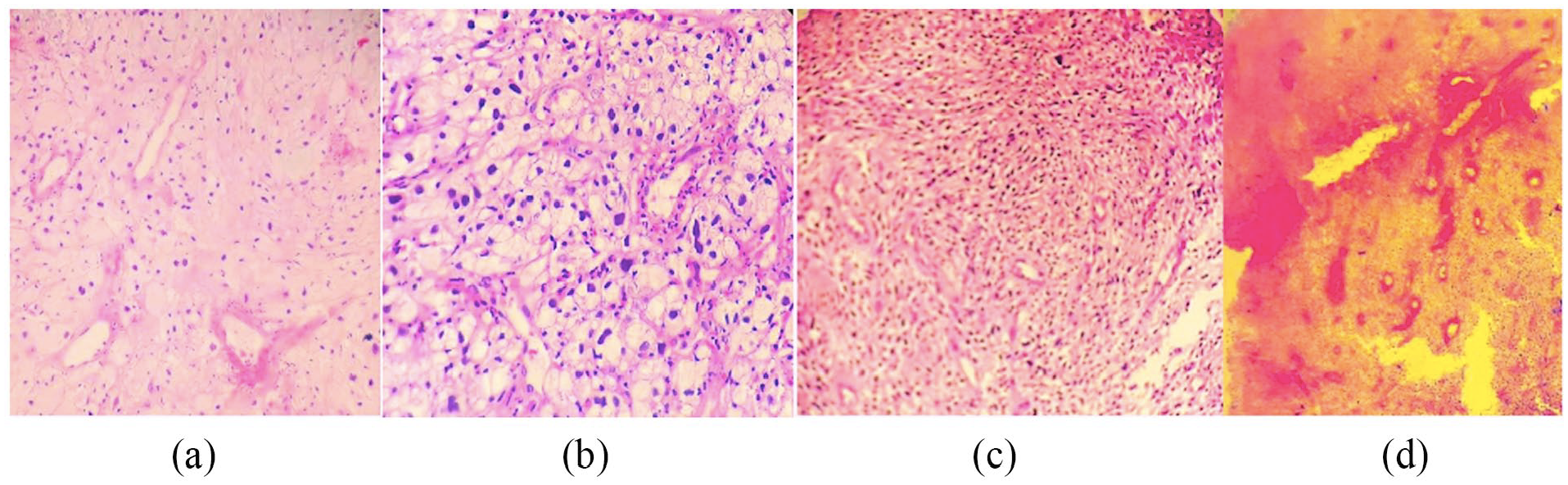
A panel of microphotographs of malignant granular cell tumour: (a) Solid sheets of tumour cells, polygonal with abundant granular cytoplasm (H&E, ×100); (b) Tumour cells showed mild-to-moderate nuclear pleomorphism, vesicular nuclei and prominent nucleoli (H&E, ×400); (c) Spindle cell differentiation, short fascicles and moderate nuclear pleomorphism (H&E, ×200); (d) Large areas of necrosis (H&E, ×40).

Immunohistochemistry of non-neuronal malignant granular cell tumour (a) Vimentin strongly (×200), (b) NSE (non-specific enolase) diffuse granular cytoplasmic positivity (×40), (c) CD10 cytoplasmic granular positivity (×200) and (d) S100 negative (×200).
Discussion
In this case, we found an abnormal location of granular cell tumour on the floor of the mouth and a lack of expression of S-100 on IHC. There were five cases of non-neuronal granular cell tumours in the literature compared with our case in Tables 1 and 2 for clinical, histomorphology and IHC.7–10
Clinical and histomorphology of six cases of non-neuronal granular cell tumour.

NA: not available; Occ.: occasional; HPF: high-power field.
Immunohistochemical features of six cases of non-neuronal granular cell tumour.
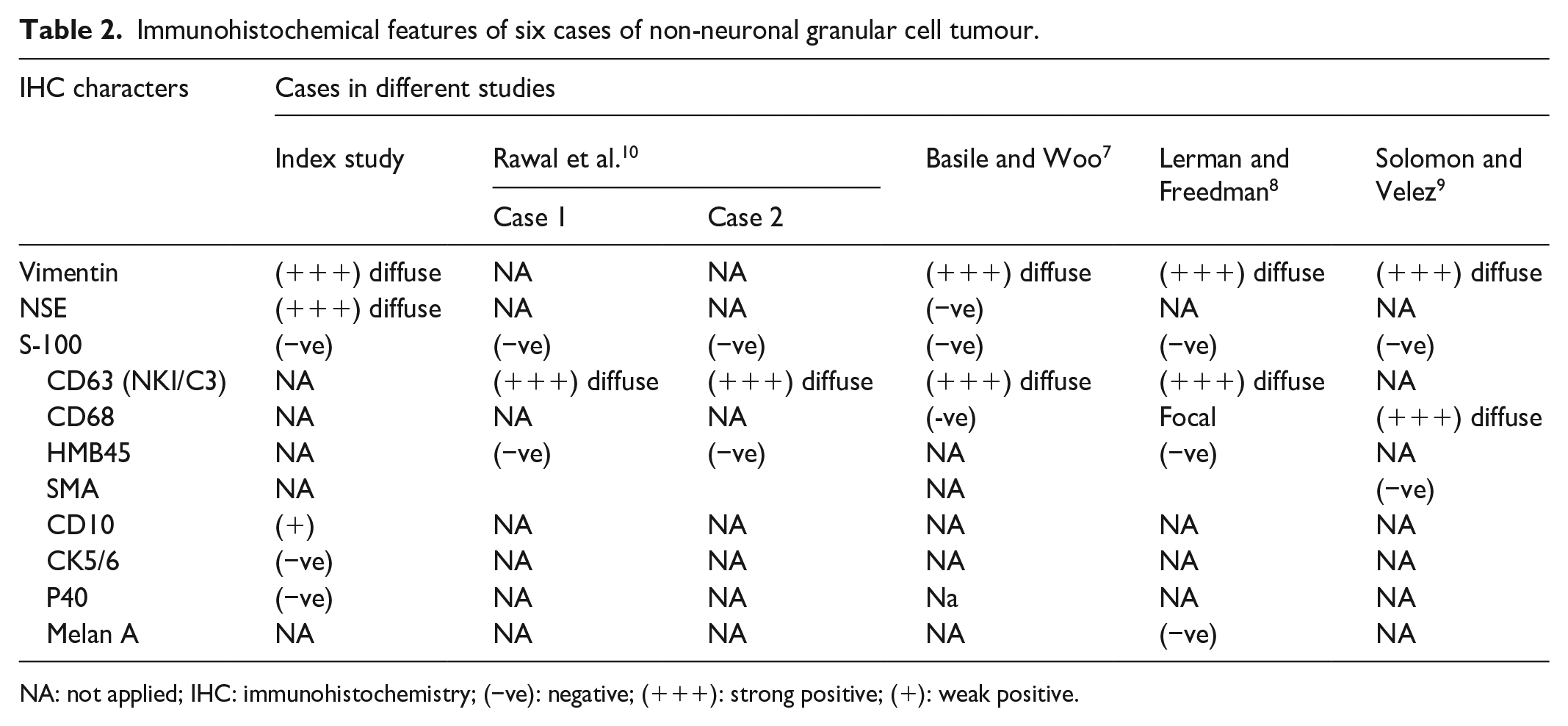
NA: not applied; IHC: immunohistochemistry; (−ve): negative; (+++): strong positive; (+): weak positive.
Histomorphologically, that tumour showed high cellularity, spindle cell differentiation, hyperchromasia, moderate nuclear pleomorphism, vesicular nuclear chromatin, prominent nucleoli and a large area of necrosis, and atypical mitotic figures (0–4/10 HPF) favouring malignant granular cell tumour; similar histomorphology has been described in one study. 12 Our case findings corroborated this study, which was reported as a malignant granular cell tumour like the Fanburg-Smith criteria that assessed the following six histologic criteria: necrosis, spindling, vesicular nuclei with prominent nucleoli, increased mitotic activity (>2 mitoses/10 high-power fields at 200× magnification), high nuclear to cytoplasmic (N:C) ratio and pleomorphism. 13 Most of the granular cell tumours originate from Schwann cells of Nevers. 2 On IHC, these GC tumours were positive for S-100 and NSE. 2 But some studies demonstrated their origin from a non-neuronal component. Cohen et al. reported a non-neural granular cell tumour (NNGCT) ( also known as primitive polypoid granular cell tumour) different from a conventional GC tumour by expressing more nuclear atypia, relatively high mitosis (0–10/10 HPF) and negative for S-100 IHC. 13 Conventional GC tumour was positive for vimentin, S-100, NSE, CD68, CD56, CD57, calretinin and inhibin. 2 That was different from NNGCT, which was positive for vimentin, NSE, CD68 and CD10, and negative for S-100, with similar positivity of IHC found in our case.13,14 Some studies reported NNGCT-associated fusion of the ALK gene and positive for Anaplastic Lymphoma Kinase (ALK) on IHC. 3 Due to our department’s lack of ALK IHC, we could not do this in this case. The patients of NNGCT were managed with complete surgical excision along with chemotherapy and radiotherapy.1,7,10 Index case was lost to follow-up after complete surgery.
There were a few case series of cutaneous granular cell tumours and polypoidal features. It was negative for S-100 IHC, first described by LeBoit et al. in 1991.4,3,15 Differential diagnoses of malignant granular cell tumour based on histomorphology are alveolar soft parts sarcoma, angiosarcoma, atypical fibroxanthoma, congenital granular cell epulis, dermatofibrosarcoma protuberans, epithelioid histiocytoma, fibroxanthoma, granular cell dermatofibroma, hibernoma, leiomyosarcoma, malignant fibrous histiocytoma, malignant peripheral nerve sheath tumour, melanoma, neurofibroma, granular cell tumour, reactive granular cell change, reticulohistiocytoma, rhabdomyosarcoma, schwannoma, squamous cell carcinoma, trichoblastoma and xanthoma. 16 Long-standing polypoidal growth with minimum symptoms indicated NNGCT. Until now, six cases of non-neuronal granular cell tumours of the oral cavity have been described; our case is the first case of malignant granular cell tumour.
Limitations
We did not follow up our patient.
Conclusion
Through this case, we presented a rare case of a non-neuronal malignant granular cell tumour at an unusual location (floor of mouth) and confirmed with IHC. To our knowledge, it is the first case of a malignant non-neuronal granular cell tumour.
Footnotes
Acknowledgements
None.
Authors’ note
This paper was presented as poster in PATHCON & Lab EXPO conference, December 2023, New Delhi, and obtained third prize.
Author contributions
A.K. prepared the manuscript and figure. N.T. provided suggestions for and edited the manuscript.
Data availability statement
Data are available upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
As per our institution, IEC does not require ethics approval to report individual cases.
Informed consent
The informed and written consent was obtained from the patient(s) for their anonymized information to be published in this article.
