Abstract
Herein, we present the case of a 17-month-old boy with asplenia, bilaterally absent pulmonary arteries, and bilateral patent ductus arteriosus, who underwent a successful Fontan operation. The central pulmonary artery was created using a pericardial roll, which was initially oversized due to the elevated pressure from the systemic-to-pulmonary shunt. The size of the roll was reduced through the process of pressure reduction by bidirectional Glenn and Fontan operations. This case provides an example of blood source-associated size transition of the pericardial roll used in the pulmonary position. Recognizing this phenomenon is vital for the successful management of this patient group.
Introduction
In cases of duct-dependent pulmonary arteries, severe lung infection, hemoptysis, and pulmonary hypertension may develop if left untreated, 1 especially when associated with an absent central pulmonary artery with bilateral ductus. Therefore, early reconstruction of the pulmonary artery is important, 2 especially in patients with a functional single ventricle. Reconstruction may be achieved by direct anastomosis, 3 but this sometimes involves an interposing material to bridge the bilateral pulmonary arteries. The distensibility of this material may dramatically differ from that of the native pulmonary artery, and its size may be prone to change by different blood sources. This knowledge is especially important in managing these patients.
Patients
A male patient, born at a gestational age of 40 weeks and weighing 3.5 kg, appeared cyanotic upon delivery and was transferred to our hospital when he was 17 days old. He was diagnosed with asplenia, right-sided single ventricle, bilaterally absent pulmonary arteries, and atrial septal defect. Prostaglandin-E1 infusion was started immediately. A computed tomography (CT) scan revealed that the right ductus arteriosus branched off the brachiocephalic artery, and the left ductus arteriosus off the aortic arch in the normal position. A cardiac catheterization performed at 22 days (Figure 1) revealed a pulmonary vascular resistance of 1.6 U/m2 and right pulmonary wedge pressure of 13/10 mmHg. The percutaneous oxygen saturation was 82%, and the pulmonary artery diameters were both 5 mm. At 1 month of age, central pulmonary artery creation using an autologous pericardial roll, a central shunt with a 4 mm artificial graft, and atrial septal defect enlargement were performed. Both ductus arteriosus were ligated (Figure 2). Although direct anastomosis of the left and right pulmonary arteries was preferable considering the patient’s growth, the distance between these two pulmonary arteries deemed too large to ensure a tension-free anastomosis. Referring to a previous report, 4 we created a fresh autologous pericardial roll wrapped around an 8-mm sizer, switching the stitches at 5-mm intervals. While anastomosing the pulmonary artery, the stitches were also switched four times to avoid a purse-string effect. Postoperative CT revealed significant size discrepancies between the pericardial roll and the native pulmonary arteries. Predischarge percutaneous oxygen saturation was approximately 80%. Thereafter, his developmental milestones and body weight gain were within the normal range, and he underwent repeat catheterization at 5 months of age (Figures 3 and 4). The study revealed a pulmonary vascular resistance of 2.2 U/m2, pulmonary artery pressure of 22/18 mmHg, and well-developed pulmonary arteries with a pulmonary artery index of 234 mm2/m2. Pursuant to this study, bidirectional Glenn was performed at 6 months. After bidirectional Glenn, the enlarged autologous pericardium was reduced in size to a diameter similar to that of the native pulmonary arteries. The pulmonary vascular bed continued to grow steadily with a pulmonary artery index of 187 mm2/m2, pulmonary vascular resistance of 1.4 U/m2, and superior vena cava pressure of 10/9 mmHg according to the catheterization at 19 months of age. Subsequently, total cava pulmonary connection was performed at 31 months of age (Figure 5). Presently, the caliber of the autologous pericardium seems unchanged by serial echocardiographic studies.

Catheterization performed at 22 days. White arrows indicate bilateral ductus arteriosus.

(a) This preoperative drawing shows that the ductus arteriosus on both sides were connected to the respective left and right pulmonary arteries. The superior vena cava was located on the left side. (b) This drawing shows the condition after the initial operation (reconstructed main pulmonary artery and central shunt). (c) This post-Glenn procedure drawing shows that the central shunt was ligated, and the superior vena cava was anastomosed to an autologous pericardial roll. (d) This detailed drawing of Figure 2(b) shows that the ductus arteriosus on both sides were ligated, severed, and excised down to the ductal tissue, cutting into the upper lobe and middle-lower lobe branches. They were then anastomosed to an autologous pericardial roll.

Catheterized at 5 months of age. An enlarged autologous pericardial portion is depicted.
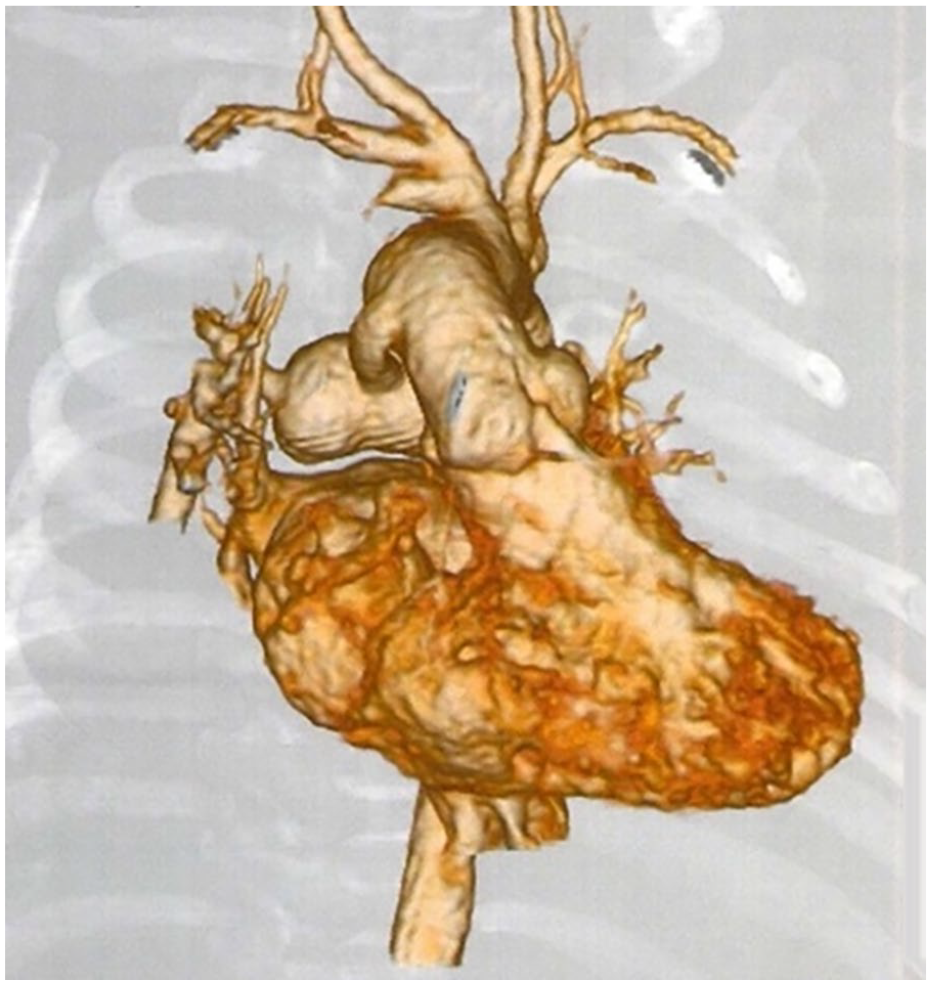
CT scan at 5 months of age. An enlarged autologous pericardial portion is depicted.
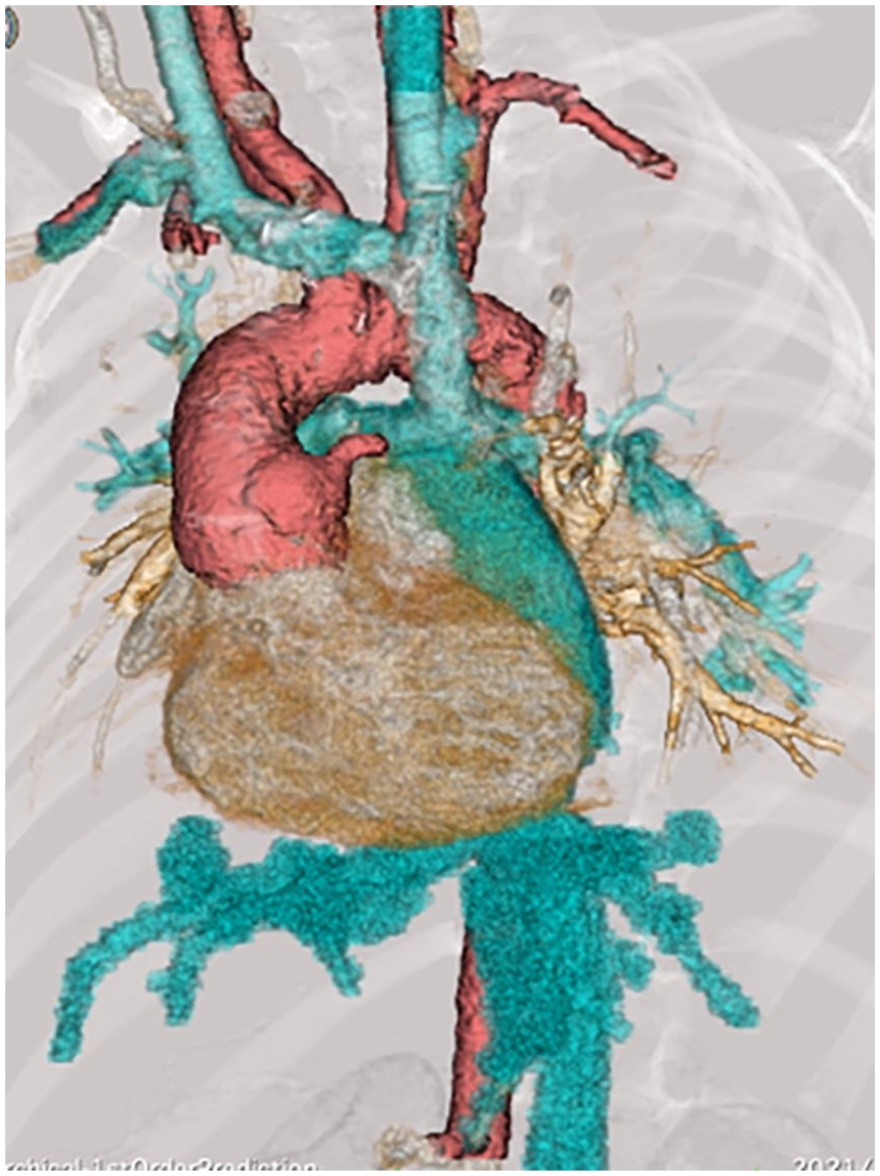
CT scan with three-dimensional volume-rendered reconstruction after total cavo pulmonary connection.
Discussion
Unilateral pulmonary atresia can be associated with congenital heart disease.3,5–7 It is purportedly caused by an abnormality in the development of the cardiac neural crest cells in the sixth branchial artery, 1 which occurs at approximately 30 days of gestation. Less frequently, the bilateral pulmonary arteries may originate from the patent ductus arteriosus separately. In any of these situations, maintenance of the ductus arteriosus is essential, as narrowing is life-threatening. In the latter instance, 5 a left-to-right pulmonary blood flow discrepancy may exist. In this case, the temporary administration of nitrogen mitigated this flow discrepancy and prevented unilateral pulmonary hypertension. In order to establish a direct continuity of bilateral pulmonary arteries, an interposing material may be necessary. 8 There have been numerous reports on unilateral pulmonary artery absence from childhood to old age.3,5–7 Reconstruction methods include direct anastomosis, autologous blood vessels, 4 autologous pericardial rolls, 4 and prosthetic grafts. 8 In cases of severe pulmonary hypertension, there have been reports 8 of using prosthetic graft due to concerns about the dilatation of the autologous pericardium. In this case, because the diameter of the reconstructed pulmonary artery was 5 mm, we chose to reconstruct with autologous pericardium, considering the ease of correcting the diameter difference and the potential for dilatation. This case shows the process of size reduction of the pericardial roll by switching the blood source from the systemic-to-pulmonary shunt to the Glenn anastomosis. While pulmonary arterial pressure is maintained via a shunt, the pericardium can become overtly dilated. However, its caliber normalizes when the blood source is switched to the Glenn anastomosis. This unique extensibility property of the pericardium must be recognized in achieving an appropriate size of the pulmonary artery at the time of Fontan completion. Future issues of this patient include potential catheter interventions to expand the pericardial roll, because of the lack of growth potention. Another consideration is that the diameter of the Glenn anastomosis is also not supposed to increase in size. Therefore, whether to leave an azygous connection to alleviate future venous congestion is another point of discussion in this context.
Conclusion
In this case, we experienced a patient with bilaterally absent pulmonary arteries with asplenia, single ventricle, and bilateral ductus arteriosus, in which we used an autologous pericardial roll for the reconstruction of the central pulmonary artery, and the Fontan procedure was achieved in stages. The size of the autologous pericardial roll changed in relation to the blood supply source. Recognizing this phenomenon is important in the management of this patient group.
Footnotes
Author contributions
Analyzed the data: T.N. Contributed to the writing of the manuscript: T.N. and M.A. Agree with manuscript results and conclusions: G.S. Jointly developed the structure and arguments for the article: T.N., M.A., and G.S. All authors reviewed and approved of the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval to report this case series was obtained from Research Ethics Committee of Kanazawa Medical University (APPROVAL NUMBER/C119).
Informed consent
Written informed consent was obtained from the legally authorized representative of the subject prior to study initiation since subject is minor.
