Abstract
We present two cases of epilepsy associated with Graves’ disease. Case 1 is a 22-year-old woman. She had three epileptic seizures and was diagnosed with idiopathic generalized epilepsy. She was treated with valproic acid (VPA). She was later diagnosed with Graves’ disease, and treated with antithyroid medication (thiamazole). We added a thyroid medication (levothyroxine) because of a decrease in free thyroxine observed with antithyroid medication. Case 2 is an 18-year-old woman. She had three epileptic seizures and was diagnosed with juvenile myoclonic epilepsy and treated with VPA. Then, she was diagnosed with Graves’ disease and was treated with thiamazole. Levothyroxine was added due to low fT4 induced by thiamazole. Due to poor compliance with antithyroid medication, the thyroid functional status was not stable. Both patients became seizure-free and euthyroid after VPA and thiamazole treatments.
Introduction
Thyrotoxicosis is, on occasion, induced by central nervous system dysfunctions such as hyperexcitation, irritability, and seizures. 1 Furthermore, seizures have been reported as a complication of Graves’ disease.1–3 However, epilepsy associated with hyperthyroidism is rare.4–6 Thyroid hormone has been reported to lower the seizure threshold,4,5 and thyroxine can reportedly induce seizures. 7 On the other hand, several antiepileptic drugs such as valproic acid and carbamazepine are known to increase thyroid hormone metabolism and lead to hypothyroidism. 8 Thyroid function monitoring may be needed for epileptic patients managed with antiepileptic drugs.
Case report
Case 1: A 22-year-old woman presented with chief complaints of generalized tonic-clonic seizures and goiter. Her past history was unremarkable, and she had no family history of epilepsy or thyroid disease. The generalized tonic-clonic seizures, lasting several minutes, had first occurred at 10 years and 11 months of age (first episode). At the time, a diffuse spike and wave complex was observed on an electroencephalogram (EEG). She was brought to our hospital at age 12 years and 7 months. Her EEG and brain magnetic resonance imaging (MRI) findings were unremarkable, and no goiter was detected. Then, similar seizures occurred at age 15 years and 6 months (second episode) and again at age 15 years and 8 months (third episode). A diffuse spike and wave complex was seen. Her psychomotor development was normal. She was diagnosed as having idiopathic generalized epilepsy (IGE), and treatment with valproic acid (VPA) (400–600 mg/day dose) was started. We recognized a diffuse goiter at age 15 years and 11 months. Her laboratory results were as follows: thyroid stimulating hormone (TSH) <0.008 μU/mL (reference range: 0.53–4.43 μU/mL); free triiodothyronine (fT3) 7.98 pg/mL (reference range: 1.95–3.55 pg/mL); free thyroxine (fT4) 2.87 ng/dL (reference range: 0.94–1.60 ng/dL); TSH receptor antibody (TRAb) 60.4% (reference range: –10% to +10%); thyroid-stimulating antibody 340% (reference range: <180%). Thyroid swelling and accumulation were identified employing technetium (Tc)-99m pertechnetate scintigraphy. Graves’ disease was diagnosed, and antithyroid medication (thiamazole) (20 mg/day dose) was started. She exhibited no exophthalmos, finger tremor, or emaciation. Because the level of fT4 was reduced while under treatment with thiamazole, we added a thyroid replacement agent (levothyroxine) (25–50 μg/day dose), and fT4 values improved. No epileptic seizures were observed with VPA administration. Improvement in EEG was observed. However, thiamazole (10 mg/day dose) was restarted due to levels of TSH (0.11 μU/mL), fT4 (1.96 ng/dL), fT3 (5.05 pg/mL), and TRAb (48.5%). The disease course is shown in Figure 1(a), and EEG results in Figure 2(a).
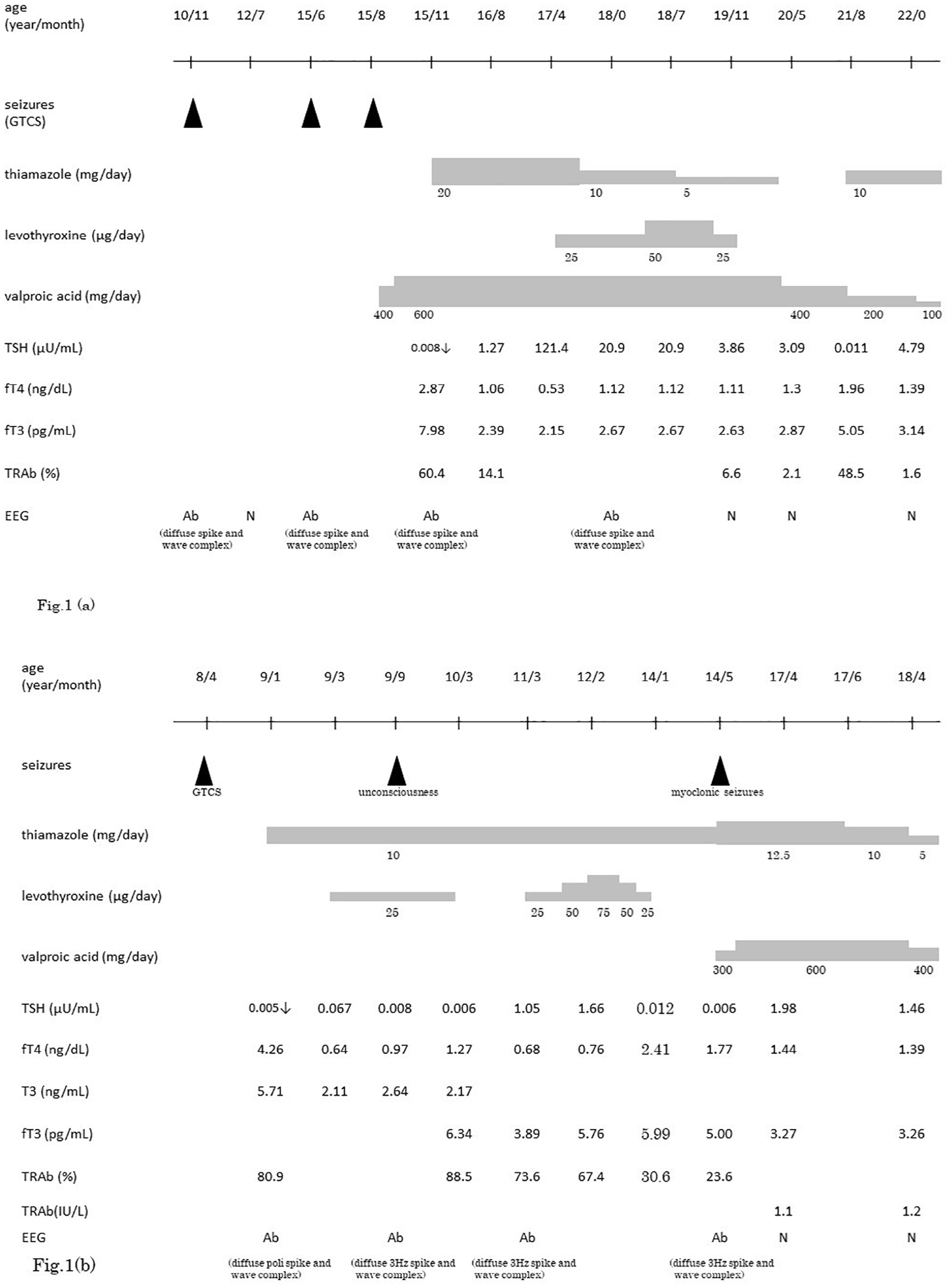
Clinical course. Upper: (a) Clinical course of case 1. Bottom: (b) Clinical course of case 2.

EEG. Upper: (a) EEG of Case 1 at age 15 years and 11 months, in the awake state. Diffuse spike and wave complexes were seen. Calibration: horizontal scale, 1 s; vertical scale, 100 μV. (b) EEG of Case 2 at age 9 years and 9 months, in the awake state. Diffuse 3 Hz spike and wave complex with short bursts predominantly in the frontal region calibration: horizontal scale, 1 s; vertical scale, 100 μV.
Case 2: An 18-year-old woman presented with chief complaint of seizures and goiter. Her past history included febrile seizures at 1 year of age. She had no family history of epilepsy or thyroid disease. Her first generalized tonic-clonic seizures lasting several minutes had occurred at 8 years and 4 months of age (first episode). Diffuse polyspike and wave complexes were observed on EEG. Goiter, weight loss, and increased sweating without exophthalmos or finger tremors were observed starting at age 8 years and 10 months. She was brought to our hospital at 9 years and 1 month of age. Her laboratory results were as follows: TSH <0.005 μU/mL; triiodothyronine (T3) 5.71 ng/mL (reference range: 0.94–1.70 ng/mL); fT4 4.26 ng/dL; TRAb 80.9%. Increased thyroid gland uptake was observed on Tc-99m pertechnetate scintigraphy. Graves’ disease was diagnosed, and thiamazole (10 mg/day dose) was started. Due to the low level of fT4 (0.68 ng/dL) under thiamazole (10 mg/day dose) treatment, we added levothyroxine (25–75 μg/day). Low compliance with thiamazole treatment resulted in thyroid toxicosis and loss of consciousness was observed several times at age 9 years (second episode), followed by several myoclonic seizure episodes with upper extremity involvement at age 14 years (third episode). Her brain MRI was unremarkable, showing a diffuse 3 Hz spike and wave complex, predominantly in the frontal region. EEG revealed no abnormalities induced by either photo-stimulation or hyperventilation. Her psychomotor development was normal. She was diagnosed with juvenile myoclonic epilepsy (JME). After the third episode, she was treated with VPA (300–600 mg/day dose). Her epileptic seizures have since been controlled, and her thyroid function remains in a euthyroid state. The disease course is shown in Figure 1(b), and EEG records in Figure 2(b).
Discussion
The prevalence of childhood epilepsy is reportedly 5.3 per 1000 in Japan. 9 On the other hand, the prevalence of Graves’ disease in childhood is very low at 0.02%, accounting for less than 5% of all cases of Graves’ disease. The male–female ratio is 1:5. Most childhood cases manifest around puberty, at 11–15 years of age. 10 These figures suggest that epilepsy and Graves’ disease are rarely associated.4–6
Treatment for Graves’ disease includes antithyroid medication, radioactive iodine ablation, and surgery. 11 In addition, in a previous study, l-thyroxine can be included when hypothyroidism occurs during antithyroid administration. 12 The administration of thyroxine during antithyroid drug treatment decreases both the production of antibodies to the TSH receptor and the frequency of recurrence of hyperthyroidism. 12 Recently, block and replace regimen therapy with antithyroid and thyroid medication has been discussed. 11
Hyperthyroidism is on occasion accompanied by central nervous system dysfunctions such as seizures.1–3,6 Notably, there is one report of seizures induced by a thyroid medication. 7 When hyperthyroidism is accompanied by seizures, antithyroid and antiepileptic drugs such as VPA and carbamazepine (CBZ) are used. However, these agents are known to affect thyroid hormones. 8 Furthermore, antiepileptic drugs reportedly increase TSH while mildly decreasing fT3 and fT4. 8 As yet, the physiopathology of seizures associated with hyperthyroidism is not well understood.6,13
Both of our cases were diagnosed with epilepsy and subsequently with Graves’ disease. In case 1, the third seizure occurred at age 15 years and 8 months, and VPA was started. Graves’ disease was diagnosed just 3 months later. When the third episode occurred, thyroid hormones were not measured, but she may have had hyperthyroidism at the time. Also, her EEG was abnormal, which may have caused the seizure. Subsequently, seizures were well controlled by VPA. Case 2 presented with hyperthyroidism 9 months after the first seizure. Treatment with an antithyroid drug was started, but seizures recurred again at 9 years and again at 14 years of age due to poor compliance with thiamazole treatment. At the time of the third seizure episode, both hyperthyroidism and abnormal EEG findings were observed. Her EEG with 3Hz diffuse spike and wave bursts was similar to that described in the report of Broutian et al. 6 Su et al. reported that high thyroid hormone levels due to low compliance with antithyroid drug treatment may have been a factor in precipitating the onset of JME. 4 No seizures were observed after the addition of an anticonvulsant. Maeda et al. reported that IGE might tend to be induced by hyperthyroidism, and then possibly improved without an anticonvulsant if the thyroid function and EEG findings were within the normal ranges. 5
Epilepsy is generally regarded as being more common in men, while IGE is more likely in women. 14 Graves’ disease is, however, more likely to affect more women, and seven of the eight cases of Graves’ disease with IGE were female.3–7 Thus, sex hormones may well be an important factor in the susceptibility of the brain to seizures.
Conclusion
We have presented two cases developing IGE in association with Graves’ disease. Hyperthyroidism and epilepsy are rarely associated. In our cases, normalization of thyroid function and administration of antiepileptic medication exerted a suppressive effect on the seizures. Importantly, the mechanism by which hyperthyroidism causes epileptic seizures has yet to be elucidated.
Footnotes
Acknowledgements
We are grateful to the patients and their parents for their commitment and permission to publish this case report.
Author contributions
S.N., M.O., and S.N. drafted the manuscript. S.N., M.O., and S.N. evaluated the patients clinically. S.N. wrote the manuscript. M.O., and S.N. provided conceptual advice. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Informed consent was given in the presence of patient and their parents. Written informed consent was obtained from the patient and their parents for their anonymized information to be published in this article.
The mentality of two patients was fine.
