Abstract
Thromboembolism is a significant cause of mortality and morbidity in cancer patients. While the link between cancer and venous thrombosis is well known, the recognition of arterial thrombosis as a serious complication of cancer and chemotherapeutic agents is a recent development. One of the chemotherapy agents frequently linked to acute vascular events is cisplatin. We discuss a rare case of cisplatin-related brachial arterial thrombosis in a 50-year-old man who was treated for cholangiocarcinoma with cisplatin and gemcitabine. Although rare, cisplatin-related arterial thrombosis demands careful monitoring, a high index of suspicion, and prompt management to prevent serious complications and mortality.
Introduction
Thromboembolism is a significant cause of mortality and morbidity in cancer patients. 1 Cancer patients face an increased risk of both arterial and venous thromboembolism; however, the occurrence of arterial thrombosis is extremely low compared to that of venous thromboembolism. 2 The causes of thromboembolism in cancer patients are multifactorial, related to the disease and its treatment. One potential cause of treatment-related arterial thromboembolism in cancer patients is the use of prothrombotic cancer drugs, which may include certain chemotherapy and targeted agents. 3 Cisplatin is one of the chemotherapy drugs frequently reported to be linked to arterial and venous thrombosis.3,4 It is used in combination or alone in the treatment of various malignant tumors, including head and neck, lung, hepatobiliary, and genitourinary cancer. Common toxicities related to cisplatin treatment include nausea, vomiting, myelosuppression, nephrotoxicity, neuropathy, and ototoxicity.5,6
Herein, we present an exceedingly rare case of cisplatin-related brachial arterial thrombosis and its management, aiming to contribute to the existing limited evidence of cisplatin-related arterial thrombosis and its treatment.
Case presentation
A 50-year-old male, newly diagnosed with locally advanced extrahepatic cholangiocarcinoma, presented to our oncology clinic complaining of intense pain and numbness in his left hand, persisting for 2 hours. He reported no associated fever or swelling. He had no personal or family history of diabetes, hypertension, cardiac, or other chronic medical conditions. His presentation occurred 1 week after receiving his third cycle of chemotherapy, which included cisplatin (60 mg/m2) on day 1 and gemcitabine (1000 mg/m2) on days 1 and 8 of a 21-day cycle. His treatment course had been uneventful until this presentation.
On physical examination, the patient’s vital signs were stable with an Eastern Cooperative Oncology Group performance status of 1. The pulse rate at the right radial artery was 92 beats per minute and regular. However, radial and brachial pulses were not palpable in the left upper limb, which also showed coldness, palmar pallor, and delayed capillary refill. No other pertinent findings were noted on physical examination.
He was investigated with a left upper limb arterial Duplex study that revealed an intraluminal mass in the left brachial artery with absent flow in the distal radioulnar arteries, suggesting total occlusion. Subsequent CT angiography demonstrated a critical left brachial artery occlusion with central filling defects in both proximal and distal segments (Figure 1), accompanied by hypoenhancement of the left radial and ulnar arteries. Axial images showed progressive luminal narrowing proximally, transitioning to complete occlusion distally, in contrast to the patent right brachial artery (Figure 2). Coronal reformatted views confirmed these findings, displaying a filling defect in the left brachial artery (Figure 3), suggestive of thrombotic occlusion. An electrocardiogram (ECG) was performed and the results were normal. However, echocardiography was not done. Laboratory tests, including complete blood count, bilirubin, liver enzymes, and renal function, were within normal range. D-dimer testing was not performed due to its unavailability at our hospital. The baseline CA 19-9 level was elevated at 1200 U/mL.
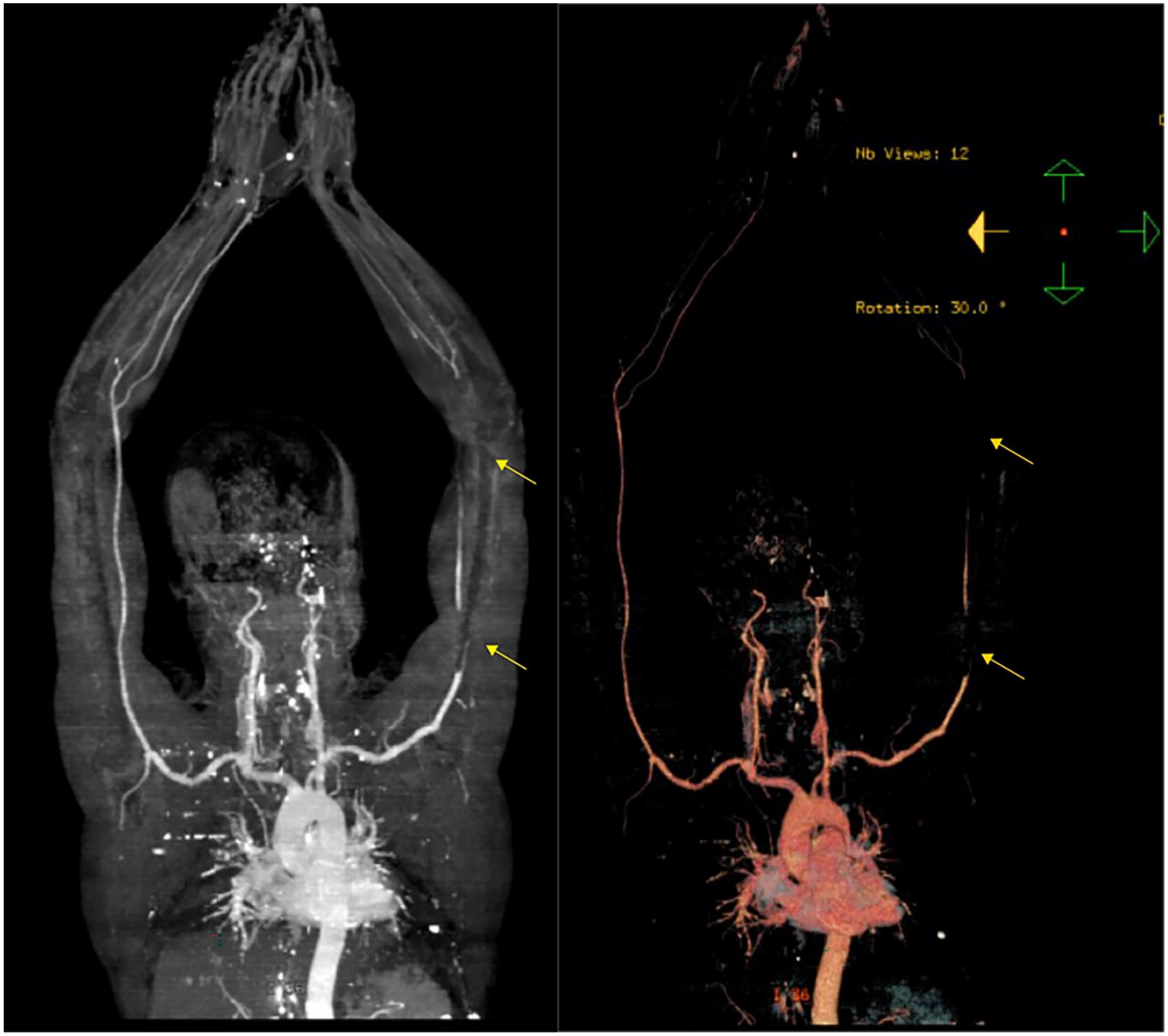
Bilateral upper extremity 3D and 3D VR (Virtual augmented reality) CT images (right and left images respectively) of the CT angiography showing filling defects in the proximal and distal left brachial artery (arrows).
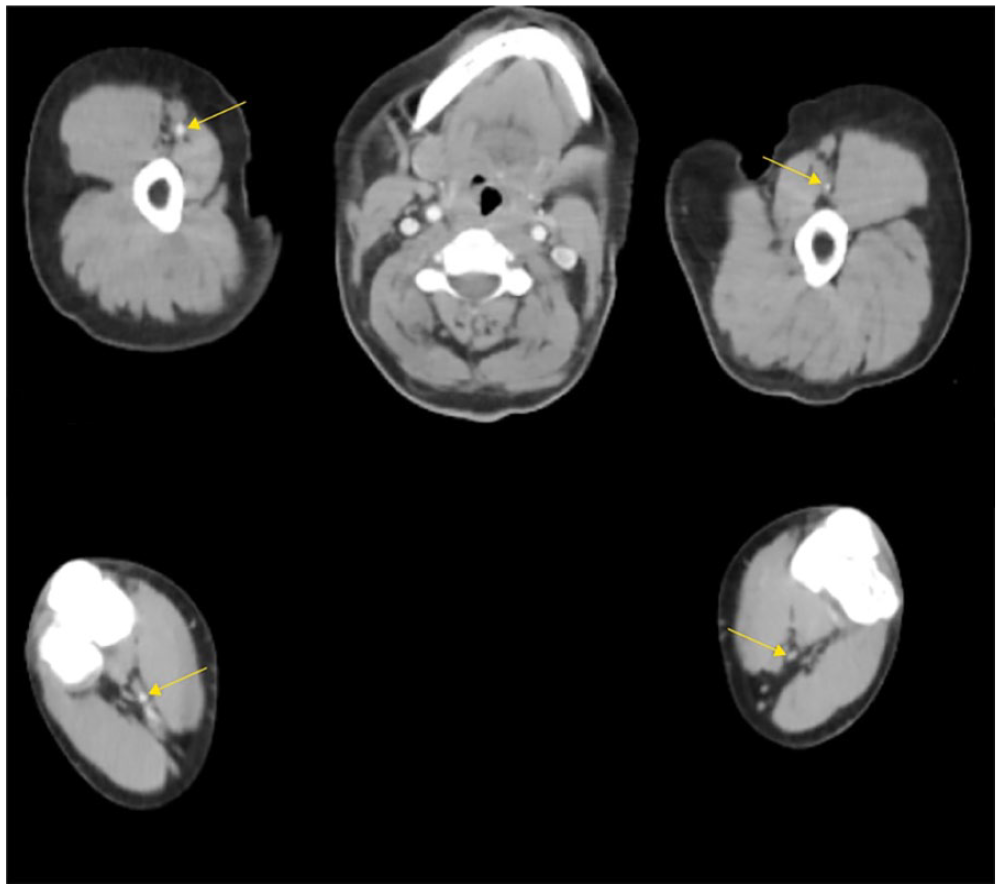
Axial bilateral CT angiographic images at the proximal (upper row) and distal brachial artery level showing left brachial artery middle level luminal partial and distal complete contrast filling defect (arrows comparing the right and left brachial artery counterparts at both levels).

Coronal reformatted images at the level of the brachial artery showing normally opacified right brachial artery (arrow in the left image) and the left brachial artery with filling defect (arrow in the right image).
A diagnosis of left upper extremity category I acute limb ischemia secondary to brachial artery thrombosis was established and the patient was started on an initial loading dose of unfractionated heparin, 5000 IU IV, followed by 17,500 IU SC BID. Subsequently, after a multidisciplinary discussion and obtaining the patient’s written consent, a definitive open thromboembolectomy was performed. Postoperatively, the patient was started on rivaroxaban (10 mg orally per day) and a loading/maintenance dose of aspirin (325/81 mg orally daily). He was scheduled for regular follow-up, and an arterial Doppler performed 6 weeks post-surgery showed no abnormalities.
Discussion
Cholangiocarcinoma stands out among high thrombotic risk cancers, alongside pancreatic, liver, lung, stomach, and brain cancers. 7 While the link between cancer and venous thrombosis is well known, the recognition of arterial thrombosis as a serious complication of cancer and certain chemotherapeutic agents is a more recent development. 3 Chemotherapy is believed to increase the risk of thromboembolism in cancer patients through drug-induced endothelial damage, increased tissue factor activity due to apoptosis-related cytokine release, and a decline in the plasma levels of anticoagulant proteins resulting from drug-induced hepatotoxicity.8,9
One of the chemotherapy agents used in cholangiocarcinoma and believed to be frequently linked to acute vascular events is cisplatin. Studies have shown that the incidence of thromboembolism is more frequent among cisplatin-based treatments compared with non-cisplatin-based treatments.7,10,11 Table 1 summarizes selected case reports of acute vascular events associated with cisplatin, including acute aortic thrombosis, ischemic stroke, myocardial ischemia, and mesenteric artery ischemia.
Arterial thrombosis events associated with cisplatin-based chemotherapy.

The exact mechanism behind cisplatin-related arterial thrombosis remains unclear. However, several hypotheses have been proposed, including elevated plasma levels of Von Willebrand factor, endothelial injury, and arterial spasms related to hypomagnesemia.22,23Additionally, studies have shown that atherosclerosis, often associated with factors like advanced age, smoking, diabetes, obesity, hypertension, and hypercholesterolemia, contributes to an increased risk of arterial thrombosis in these patients. 3 The absence of other vascular risk factors, coupled with the patient’s presentation after three cycles of cisplatin, strongly suggests cisplatin as a potential cause of arterial thrombosis. This observation is significant, as it corresponds to previous reports of cisplatin-related arterial thrombosis occurring within 90 days of starting cisplatin treatment. 24 It is worth noting that our case differs from reports of cisplatin-related aortic thrombosis, as our case presents acute limb ischemia without aortic thrombosis.
While various venous thromboembolism risk assessment tools, such as the Khorana, Protecht, and ONKOTEV scores, have been developed to evaluate the risk of venous thromboembolism (VTE), limited data exist regarding their applicability in assessing arterial thromboembolism (ATE) risk. A retrospective analysis involving 133 cholangiocarcinoma patients found that an ONKOTEV score of ⩾2 and elevated CA 19-9 levels were identified as independent predictors for thromboembolic events, with CA 19-9 being notably predictive for ATE. 7 In our case, factors such as the tumor’s location and advanced stage, along with highly elevated CA 19-9 levels, may have contributed to the increased risk of ATE development in our patient.
Apart from the scarcity of risk assessment tools, there is also a paucity of evidence concerning the management of cisplatin-related arterial thrombosis. However, acute limb ischemia, regardless of its cause, constitutes a medical emergency demanding immediate identification and intervention to prevent limb loss. 25 Prompt initiation of anticoagulation with unfractionated heparin (UFH) and careful consideration of the optimal revascularization techniques are crucial. Factors like the location, limb viability, comorbidities, and acute limb ischemia (ALI) classification should guide these decisions. 26 Our patient received immediate open thromboembolectomy followed by both antiplatelet therapy (aspirin) and a direct Factor Xa inhibitor (rivaroxaban). This treatment approach was based on extrapolation from the management of venous thromboembolism and supported by evidence from the vascular outcomes study. This study showed that combining aspirin with a low dose of rivaroxaban after limb revascularization significantly reduced the morbidity associated with acute limb ischemia, amputation, and other acute vascular events when compared to aspirin alone. 27
Conclusion
While cisplatin-related acute limb ischemia is rare, it demands vigilant monitoring, suspicion, evaluation, and prompt management to prevent severe complications in these patients. Given the rarity of the complication and the limited existing literature, we recommend conducting further multicenter institutional prospective studies to gain a better understanding of risk factors, pathophysiology, and optimal management strategies for cisplatin-related ATE in cancer patients. This study will help to establish evidence-based guidelines to enhance patient care, identify preventive measures, and refine risk assessment tools for patients taking cisplatin and other prothrombotic drugs, making a significant contribution to the medical field.
Footnotes
Acknowledgements
The willingness and cooperation of the patient and his family in our plan to publish the case for academic purposes is greatly appreciated. We are also thankful to Mr. Ermiyas Assefa, a graphic designer and engineer, for his assistance in preparing the patient’s high-quality CT pictures.
Author contributions
All Authors contributed to drafting the work. A.T. and A.A. wrote the main manuscript. A.K., E.A., and M.A. edited and revised the manuscript. A.K., A.Te., and A.A. contributed to data acquisition. All authors have reviewed and approved the final manuscript.
Availability of data and materials
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Institutional ethical approval is not required at Addis Ababa University Tikur Anbessa Comprehensive Specialized Referral and Teaching Hospital if patient consent is obtained.
Informed consent
Written Informed consent was obtained from the patient and his family for the publication of this case report and anonymized CT images.
