Abstract
Anti-phospholipid syndrome is an autoimmune disorder characterized by episodes of arterial and/or venous thrombosis and/or pregnancy morbidity in the presence of anti-phospholipid antibodies. Catastrophic anti-phospholipid syndrome is an accelerated form of the disease with rapid involvement of multiple organ systems often posing a diagnostic challenge. There is a paucity of literature on the myriad presentations of catastrophic anti-phospholipid syndrome owing to the orphan nature of the disease. We present three cases of catastrophic anti-phospholipid syndrome in patients with systemic lupus erythematosus that presented with episodes of thrombosis involving both arterial and venous systems and multisystem organ failure. Timely diagnoses were made based on a high index of suspicion and were managed with a combination of systemic glucocorticoids, cyclophosphamide, plasmapheresis, intravenous immunoglobulin and other supportive measures. However, despite providing the standard of care, we encountered a poor outcome in two of these patients, highlighting the high mortality associated with catastrophic anti-phospholipid syndrome.
Keywords
Introduction
Anti-phospholipid syndrome (APS) is an autoimmune disorder characterized by one or more episodes of arterial, venous or small vessel thrombosis in any tissue or organ with/without associated pregnancy morbidity in the form of foetal deaths or unexplained consecutive abortions, in the serological presence of anti-phospholipid antibodies – lupus anticoagulant, anti-cardiolipin antibody IgM/IgG or anti-β2-glycoprotein-I antibody IgM/IgG persisting for at least 12 weeks.1–3 APS may manifest as primary APS or in the background of a connective tissue disease like systemic lupus erythematosus (SLE), when it is called secondary APS.
A subset of patients with APS present with catastrophic antiphospholipid antibody syndrome (CAPS), an accelerated form of disease, with multisystem organ failure, often posing a diagnostic challenge, and associated with high mortality. 4 In European countries, CAPS accounts for up to 5.4% of mortality among APS patients. 5
We present three cases of CAPS in the background of SLE, who presented with a rapid progressive involvement of multiple organ systems and had varied outcomes despite the timely diagnosis and emergent management.
Case presentation
Case 1
A 22-year-old lady diagnosed with SLE 4 years back (basis – acute cutaneous lupus rash, proteinuria, positive anti-nuclear antibody and anti-Smith antibody, and reduced complement levels) had been managed with hydroxychloroquine, prednisolone, mycophenolate mofetil and diuretics for a year, after which she had felt better and discontinued treatment. She presented to a private hospital with anasarca for 1 month and withdrawn behaviour for 10 days duration. She had leucocytosis 34,800/μL, elevated serum procalcitonin 0.7 ng/mL (normal < 0.05), prolonged activated partial thromboplastin time (48.1 s), proteinuria and bilateral moderate pleural effusion. A kidney biopsy showed IgA nephropathy. She was managed as a probable case of sepsis with septic encephalopathy. However, with no improvement in her general condition, she was referred to our hospital for further management.
On examination, she was ill-looking and had tachycardia, tachypnoea, hypotension, pedal oedema, ascites, bilateral pleural effusion and withdrawn behaviour. She had anaemia, haemoglobin 10 g/dL, leucocytosis 32,400/μL, thrombocytopenia 61,000/μL, positive direct Coombs’s test, raised D-dimer 4070 ng/mL (normal 200–250 ng/mL) and raised serum creatinine. Fundoscopy, plain computed tomography brain and cerebrospinal fluid analysis were normal. She had anti-nuclear antibody 4 + speckled pattern on immunofluorescence with a positive anti-Smith and antinuclear ribonucleoprotein antibody on extractable nuclear antigen profile. Further evaluation with a computed tomography pulmonary angiography showed a filling defect in the right segmental branches of the right middle lobe and left lower lobe (Figure 1(a)) with peripheral wedge-shaped consolidations in the right middle lobe and left lower lobes (Figure 1(b)) suggestive of pulmonary thromboembolism, and an incidental finding of splenic infarct on the extended field of view (Figure 1(c)).
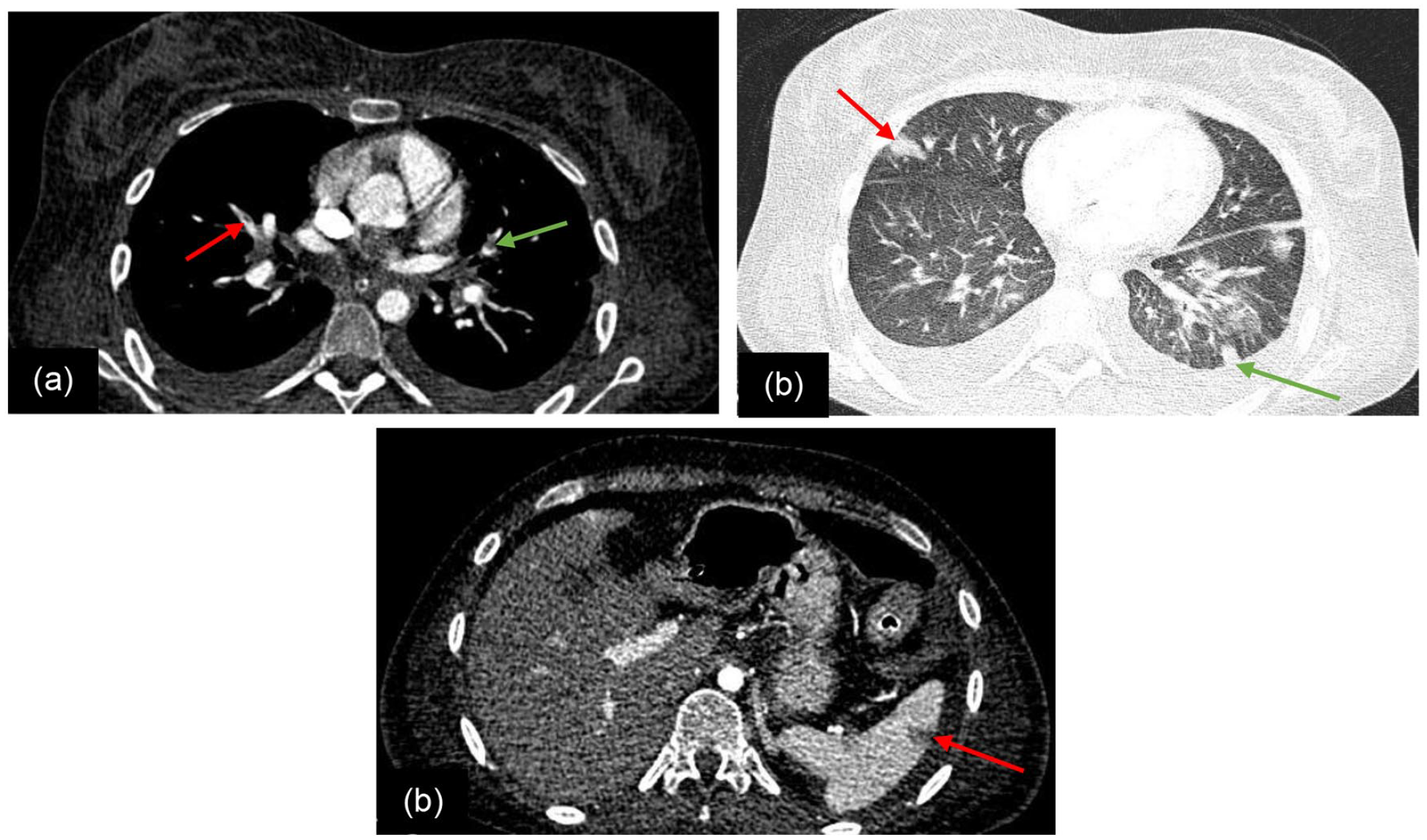
Computed tomography pulmonary angiogram of a 22-year-old female with catastrophic antiphospholipid syndrome and systemic lupus erythematosus. (a) A filling defect in the right middle lobe (red arrow) and left lower lobe segmental branches (green arrow) suggestive of thrombosis. (b) Corresponding wedge-shaped sub-pleural consolidations in the right middle lobe (red arrow) and the left lower lobe (green arrow). (c) Extended field of view showing splenic infarct (arrow).
She was managed as a case of SLE with pulmonary thromboembolism, probable aetiology – APS, with fondaparinux (heparin withheld in view of thrombocytopenia), intravenous immunoglobulin, broad-spectrum antibiotics and transfusion of packed red blood cells and platelets. On the following day while on treatment, she developed right hemiparesis with aphasia; a magnetic resonance imaging brain showed hyperacute infarct in the left middle cerebral artery territory (Figure 2(a) and (b)) and in the left posterior inferior cerebellar artery territory (Figure 2(c) and (d)). Magnetic resonance angiography showed thrombosis of the left middle cerebral artery (M1 and M2 segments) (Figure 2(e)) and magnetic resonance venography showed thrombosis of right transverse and sigmoid sinus (Figure 2(f)). The APS panel sent on the day of admission was positive for lupus anticoagulant. Based on the findings of pulmonary thromboembolism, stroke, cerebral venous sinus thrombosis, splenic infarct and lupus anticoagulant, she was diagnosed as a case of ‘probable CAPS’ based on the 2003 international consensus statement on CAPS. 4 Intravenous immunoglobulin and anticoagulation were continued with the addition of pulse methylprednisolone, followed by cyclophosphamide 750 mg. However, she had a refractory course and developed features of cytokine storm syndrome, followed by neutropenic sepsis and succumbed to her illness.

Magnetic resonance imaging and magnetic resonance angiography of a 22-year-old female with catastrophic antiphospholipid syndrome and systemic lupus erythematosus. (a) and (b) Diffusion-weighted imaging and corresponding apparent diffusion coefficient map show restriction of diffusion in the left middle cerebral artery territory suggestive of hyperacute infarct. (c) and (d) Diffusion-weighted imaging and corresponding apparent diffusion coefficient map show restriction of diffusion in the left posterior inferior cerebellar artery territory (arrows). (e) Magnetic resonance angiography image shows loss of flow-related enhancement of left middle cerebral artery (M1 and M2 segments) and its branches suggestive of thrombosis (arrow). (f) Magnetic resonance venography shows loss of flow-related enhancement of right transverse and sigmoid sinuses suggestive of thrombosis (arrow).
Case 2
A 23-year-old lady presented to a government hospital with a history of oral ulcers, alopecia, photosensitive malar rash, polyarthritis of small joints of hands, Raynaud’s phenomenon, intermittent fever of 1-month duration along with headache and gangrene of the right thumb of 5 days duration (Figure 3(a)). Initial evaluation had revealed normal haemoglobin and cell counts, anti-nuclear antibody by immunofluorescence 3 + speckled, raised anti-double–stranded deoxyribonucleic antibodies 52.7 IU/mL (normal < 30 IU/mL). She was managed as a case of SLE with hydroxychloroquine and prednisolone, and then discharged. Four days later, she suffered eight episodes of generalized tonic-clonic seizures. On examination, she had bilateral papilloedema but no focal neurological deficit. Magnetic resonance imaging of the brain showed increased white matter intensities and magnetic resonance venography showed a thrombus in the middle one-third of the superior sagittal sinus with complete obliteration of straight sinus (Figure 3(b)).

A 23-year-old female with catastrophic antiphospholipid syndrome and systematic lupus erythematosus. (a) Dry gangrene involving the distal phalanx of the right thumb. (b) Sagittal reformatted magnetic resonance venogram (MRV) of brain shows loss of flow-related enhancement in superior sagittal (red arrows) and the straight (green arrow) sinuses suggestive of thrombosis. (c) Dry gangrene involving all toes.
She was managed as a case of cerebral venous thrombosis, anti-coagulated with low molecular weight heparin and was referred to our centre for further management. At our centre, examination revealed progression of digital gangrene to involve all toes (Figure 3(c)). She had anaemia (haemoglobin 8.8 g/dL), raised erythrocyte sedimentation rate 60 mm/first hour, raised C-reactive protein 14.2 mg/dL, with APS screen positive for lupus anti-coagulant. Based on cerebral venous thrombosis, digital gangrene and lupus anticoagulant, she was managed as a case of SLE with ‘probable CAPS’ with pulse methylprednisolone, low molecular weight heparin bridged with warfarin, anti-epileptic drugs, cyclophosphamide 750 mg and hydroxychloroquine. While in hospital, she developed tingling and numbness in the thumb, index and middle fingers of both hands with decreased sensations, and a nerve conduction study confirmed mononeuritis multiplex which was managed with intravenous immunoglobulin. She was discharged after achieving full recovery of sensory function.
Case 3
A 47-year-old lady was a case of SLE with lupus nephritis class IV diagnosed by renal biopsy (basis of diagnosis – anasarca, dyspnoea, antinuclear antibody by immunofluorescence 4 + speckled pattern, reduced complement levels, direct Coomb’s test 2+). She had already received treatment with three doses of pulse methylprednisolone (1 g/dose) followed by two doses of cyclophosphamide (500 mg/dose) as per the Euro-Lupus regime. Prior to her third dose of cyclophosphamide, she presented with paraparesis of 5 days duration along with paraesthesia and cold sensation in lower limbs of 3 days duration. On examination, she had tachycardia and hypotension. Her left lower limb was cold and clammy with mottled skin and absent femoral, popliteal, anterior tibial and dorsalis pedis pulses. The muscle power in the upper limbs was grade 4 at shoulder joints. The muscle power in the lower limbs was grade 1. Touch, proprioception and vibration sensations were preserved in lower limbs with impaired pain and temperature. She had haemoglobin 9.4 g/dL, leucocytes 17,100/μL, platelets 199,000/μL, prothrombin time 15 s, international normalized ratio 1.15, activated partial thromboplastin time 30.6 s, urea 63 mg/dL and creatinine 1.2 mg/dL with raised serum procalcitonin and creatine phosphokinase levels (813 IU/L). Screening for APS was positive for high titre anti-β2-glycoprotein-I IgM antibody. 2D echocardiogram showed a left ventricular ejection fraction of 60%, large pericardial effusion anterior to right ventricular apex (max diameter 17 mm) with no evidence of cardiac tamponade, along with bilateral pleural effusion. Computed tomography angiogram of the abdominal aorta and bilateral lower limb arteries showed thrombosis infra-renal abdominal aorta (Figure 4(a)) and in the left popliteal artery (Figure 4(b)).

A 47-year-old lady with systemic lupus erythematosus and lupus nephritis class IV presenting with arterial thrombosis. Computed tomography angiogram of the abdominal aorta and bilateral lower limbs show (a) hypodense thrombus within the distal abdominal aorta (infra-renal), and (b) in left popliteal artery (arrow).
A clinical diagnosis of anterior spinal artery thrombosis was considered and a magnetic resonance angiography was planned (however could not be done due to the poor general condition of the patient). Based on clinical anterior spinal artery thrombosis, bilateral lower limb arterial occlusion, anti-β2-glycoprotein-I IgM antibody, she was managed as a case of SLE with ‘probable CAPS’ with pulse methylprednisolone (1 g/dose for 3 days), low molecular weight heparin, broad-spectrum antibiotics and plasmapheresis. She was planned for amputation of the left lower limb. However, the patient developed sudden haemodynamic deterioration after the first cycle of plasmapheresis requiring vasopressor support and mechanical ventilation. Plasmapheresis was stopped and intravenous immunoglobulin was started. However, on the second day of intravenous immunoglobulin, she developed sudden bradycardia and cardiac arrest and died.
Discussion
CAPS defines an accelerated form of disease with widespread thrombosis with multiorgan failure. The diagnosis of definite or probable CAPS is based upon the 2003 consensus statement on the classification criteria and statement guidelines for the CAPS Registry Project Group.2,4 It has a sensitivity of 90.3% and specificity of 99.4% in the diagnosis of CAPS. 1
CAPS can involve multiple organ systems with thrombosis in vascular beds of all sizes. The proposed mechanisms of thrombosis are: cellular activation of endothelial cells, monocytes, neutrophils and platelets; inhibition of natural anticoagulant mechanisms, inhibition of fibrinolysis and enhanced conversion of factor XI to XIa; activation of the complement system; trophoblast dysfunction and mammalian/mechanistic target of rapamycin pathway activation. However, the extent to which various mechanisms predominate or the common central pathway of thrombosis remains unresolved. 6
While the majority of cases of CAPS reported are in females, a recent review of incident cases of APS based on the Sydney criteria in a population-based study from 2000 to 2015, reported that only 55% were females. 7 CAPS occurs usually as a primary disease in 59%, with SLE in 26.9%, with lupus-like diseases in 3.4% and the remaining with other autoimmune diseases. 8 CAPS is often precipitated by infections, neoplasms, surgical procedures, withdrawal of anticoagulation or low levels of international normalized ratio. 8 In our series, all three patients were females with SLE. The precipitating factor in the first two cases was disease flare while it was unclear in Case 3.
In a 10-year follow-up of 1000 patients with APS, stroke was reported in 8.1%, transient ischemic attacks in 4.7%, deep vein thrombosis in 4.3% and pulmonary thromboembolism in 3.5% cases. 5 The disease has a rapid progression and can involve the kidneys, lungs, brain, heart, skin, peripheral vessels, intestines and other organs. 8 Renal involvement manifests with acute kidney injury, hypertension, haematuria and proteinuria. 9 Pulmonary manifestations include acute respiratory distress syndrome, pulmonary thromboembolism and pulmonary haemorrhage. 10 Nervous system involvement manifests as encephalopathy, stroke, seizures, headache and coma. Cardiac involvement includes heart failure, myocardial infarction and defects. 11 Other complications include skin necrosis, digital gangrene, and thrombocytopenia. 12 In our series, Case 1 had extensive organ system involvement with stroke, cerebral venous thrombosis, splenic infarct and pulmonary thromboembolism; Case 2 had cerebral venous thrombosis with digital gangrene involving the right thumb and bilateral lower limbs; and Case 3 had anterior spinal artery thrombosis and lower limb arterial occlusion that occurred within 1 week of symptom onset.
The treatment of CAPS is directed at two different pathologies: the thrombotic events and the suppression of cytokine cascade. 8 Anticoagulation with low molecular weight or unfractionated heparin in the acute setting that is bridged with warfarin is the mainstay of treatment. In our series, all cases received low-molecular-weight heparin except for Case 1, who received fondaparinux (due to thrombocytopenia). To suppress the cytokine cascade and for immunomodulatory effects, the use of high-dose systemic glucocorticoids such as methylprednisolone (0.5–1 g intravenously for 3 days) followed by oral glucocorticoid equivalent to prednisolone 1 mg/kg have been reported.13–16 The other modalities to control inflammation and mitigate the effect of antiphospholipid antibodies include intravenous immunoglobulin (400 mg/kg/day for 5 days) and plasma exchange. In our series, all three cases were managed with high-dose steroids and intravenous immunoglobulin, and Case 3 was managed with plasmapheresis.
In patients resistant to the above therapies, treatment options include rituximab in the presence of microangiopathic haemolytic anaemia 17 and cyclophosphamide and eculizumab in CAPS refractory to therapies including plasma exchange.13,15 Cyclophosphamide is recommended in the treatment of CAPS associated with SLE. 18 However, there is no literature that compares the effectiveness of these therapies in the prevention of mortality. In CAPS secondary to SLE, it is recommended to continue hydroxychloroquine for its immunomodulatory effects.
CAPS has high mortality in its initial phase of the disease due to organ system failure or complications arising from therapy. In our series, one patient survived and is being followed up on an outpatient basis for recurrence of thromboembolic events and therapeutic monitoring of warfarin dosage.
CAPS presents with a wide range of clinical manifestations across multiple organ systems and is associated with a rapidly deteriorating course, and high mortality despite initiation of optimum management. A high index of suspicion is required while dealing with such cases.
Footnotes
Acknowledgements
We thank Thinley Jamtsho Tshering for generating the image plates and the patient’s family members for proving consent.
Data availability
All data and images relevant to this case series are presented in the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
