Abstract
Pancreatitis can produce several complications such as pseudocyst, which can happen in acute and chronic pancreatitides. Pseudocysts are typically found in the abdomen but can rarely extend into the mediastinum. Atypical symptoms such as dyspnea, dysphagia, coughing, vomiting, abdominal or chest pain, and hemoptysis are usually the notable complaints. CT scan, MRI, and endoscopic ultrasound are valuable diagnostic modalities. Drainage and surgical removal of the pseudocyst are the treatment options. Herein, we outline the case of a young female with episodic chest and epigastric discomfort, dysphagia, and weight loss. Previously, she was incorrectly diagnosed with gastroesophageal reflux disease and peptic ulcer. A mediastinal pseudocyst secondary to chronic pancreatitis was found to be the cause. The patient underwent surgical removal of the pseudocyst and a pancreaticojejunostomy. Significant improvement was noticed at follow-up. This article highlights the possibility of such unusual conditions and the importance of a proper assessment while treating patients with epigastric pain.
Clinical message
Though pancreatic pseudocyst is a common complication of pancreatitis, its extension and presence in the mediastinum are unusual and rare. The initial history of abdominal pain can be nonspecific and brushed aside, which can lead to a missed or delayed diagnosis. Early identification is crucial to avoid life-threatening complications. This article emphasizes that a detailed history and a structured evaluation are essential for diagnosing the condition and the pancreatitis that is causing it. Imaging tools such as a CT scan or MRI can be used to differentiate other similar-appearing pathologies in the mediastinum.
Introduction
A pancreatic pseudocyst is composed of amylase/lipase-rich pancreatic fluid enclosed in fibrous and granulomatous tissue (lacking an epithelial lining, hence named pseudocyst) that leaks into the peripancreatic space due to obstruction or disruption of the pancreatic duct. 1 It can occur at any age and be caused by acute or chronic pancreatitis, trauma, and malignancies. Chronic pancreatitis is the most common cause in adults, with an incidence of 20%–40%, whereas in children, >60% of cases are attributable to blunt trauma. 2 Pseudocysts are primarily located in the abdominal cavity but can sometimes spread to other areas, such as the liver, spleen, and testes. Rarely can they pass through the openings in the diaphragm and into the mediastinum above the diaphragm. 3 Very few cases of a mediastinal pancreatic pseudocyst are described in the literature, with the first occurring in 1951.2,4
Patients with a mediastinal pseudocyst experience breathing difficulties, chest pain, dysphagia, vomiting, weight loss, coughing, hemoptysis, and palpitations.5,6 Due to the pressure effects caused by its proximity to vital organs, fatal complications such as respiratory failure, airway obstruction, hypotensive shock, pleural effusion, and congestive cardiac failure may occur.7,8 A delayed diagnosis and a higher risk of complications can result from an inadequate evaluation and workup. This paper reports a rare case of extensive mediastinal pseudocyst in a young female initially diagnosed with gastroesophageal reflux disease (GERD) and peptic ulcer disease (PUD) in other settings but did not improve with several months of pharmacotherapy.
Case presentation
An 18-year-old girl presented to our outpatient department with complaints of chest discomfort, dysphagia, vomiting, and increasing weakness for one and a half months. She had no comorbidities. She had a history of epigastric pain and anorexia for 1 year. She had several consultations for these symptoms and was labeled with GERD and PUD. However, despite the constant use of Proton pump inhibitors (PPIs) and prokinetics, she saw no improvement. A detailed interview disclosed a chronic history of episodic abdominal pain, diarrhea, and weight loss. Her vital signs were within normal range, and she appeared pale. She weighed 41 kg. Mild epigastric tenderness, increased bowel sounds, and a faint systolic ejection murmur were noted on systemic examination. Her Electrocardiogram (EKG) and chest x-ray were normal. Serum investigations were done that showed hemoglobin of 7.8 g/dL, raised serum amylase of 134 U/L (<90) and lipase of 58 U/L (13–60), normal liver enzymes, and negative serology for HAV (hepatitis A virus), HBV (hepatitis B virus), HCV (hepatitis C virus), and HIV. Stool analysis was done for chronic diarrhea, and it showed no ova or parasites. However, the fecal fat content was elevated. Distorted gastric contours and reduced pancreas size were appreciated on transabdominal ultrasound. A CT scan of the thorax and abdomen was then ordered and showed an atrophic pancreas along with calcification and dilated main pancreatic duct (Figure 1(a)). Surprisingly, a well-defined, thin-walled cyst was seen that extended from the superior peripancreatic region into the posterior mediastinum as far as the subcarinal area anterior to the descending thoracic aorta (Figure 1(b)–(d)). The stomach fundus and diaphragmatic crus were pushed anterolaterally. Serologic testing with ELISA was done that ruled out hydatid cyst disease. Based on the symptoms, laboratory investigation, and radiographic findings, the patient was diagnosed with chronic pancreatitis complicated by pseudocyst extending into the mediastinum. Following initial management with blood transfusion and painkillers, consultation with a general surgeon was done, and exploratory laparotomy was advised. The patient subsequently underwent surgical resection of the pseudocyst and lateral pancreaticojejunostomy. Her postoperative course was unremarkable. At follow-up visits, significant improvement in the patient’s condition was noted with resolution of the symptoms and gain of weight. A timeline of care is summarized in Table 1.
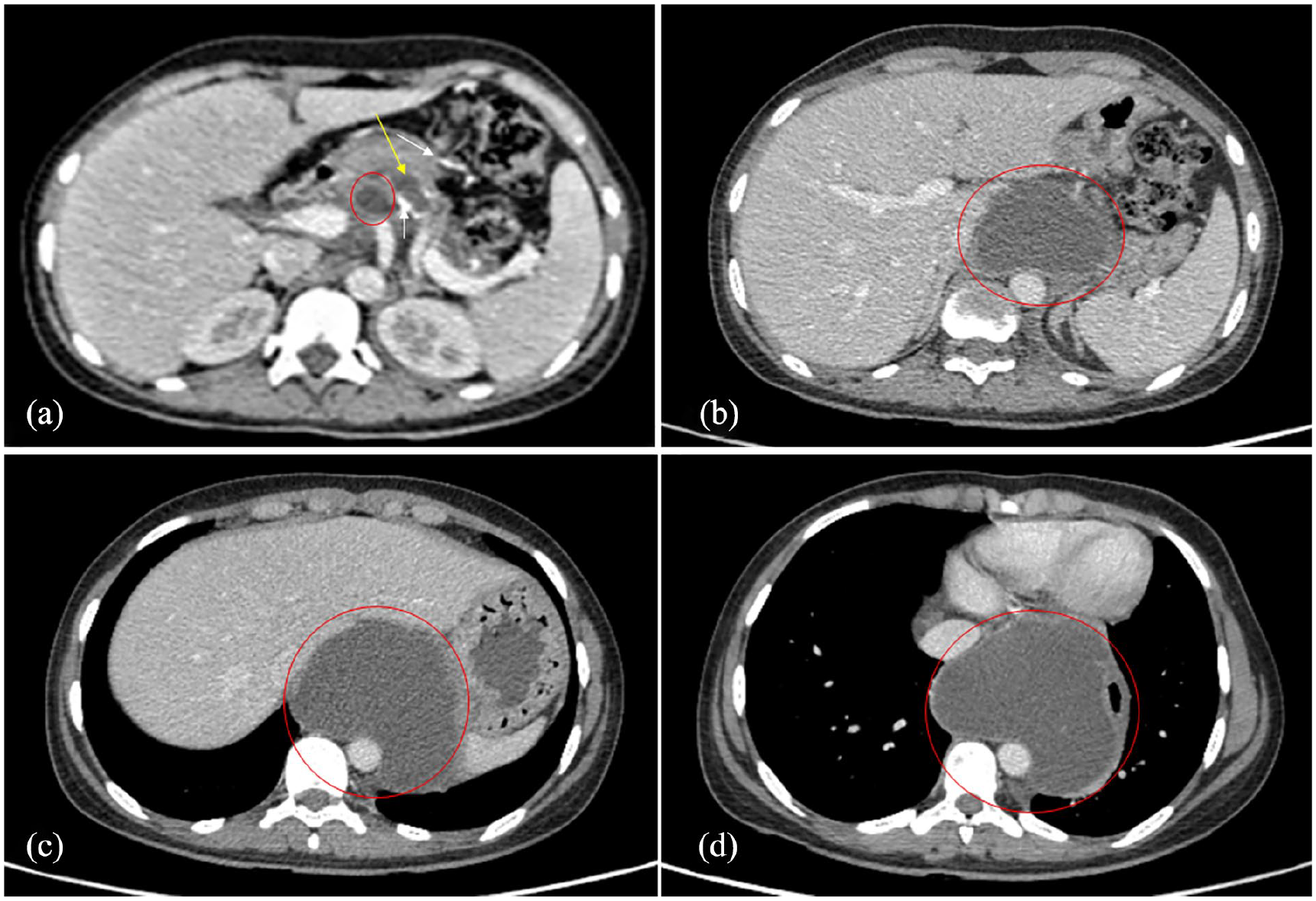
Contrast enhanced CT abdomen and thorax. (a) axial section at the level of pancreas showing atrophic pancreas, pancreatic calcifications (white arrows), dilated main pancreatic duct (yellow arrow), and pseudocyst (red circle). (b) and (c) axial section at the level of proximal abdominal aorta. A well-defined fluid density (red circle) that is the pseudocyst can be seen extending proximally through the aortic hiatus. The pseudocyst is related anteriorly to the left lobe of liver (b) and the body of stomach (c), and posteriorly to the aorta. (d) Intrathoracic extension of the pseudocyst (red circle) into the posterior mediastinum. It is related to the left atrium anteriorly. The esophagus is compressed and displaced anterolaterally.
Timeline of care.

Discussion
Pancreatic pseudocyst is a rare clinical condition with an incidence of 7%–25% after acute pancreatitis and 20%–40% after chronic pancreatitis. In a minority of cases, it develops after trauma to the pancreas or surgery. 1 Eighty percent of pancreatic pseudocysts arise in the head and body of the pancreas. The remaining 20% are outside the pancreas and found in the abdomen, pleura, mediastinum, liver, spleen, and pelvis. 9 Inflammation and fibrosis in the peripancreatic regions create areas of lesser resistance, forming thoracopancreatic fistulas, and terminating in pleural, mediastinal, bronchial, or pericardial regions. 9 Mediastinal extension occurs when peripancreatic fluid enters through the diaphragmatic openings into the mediastinum. In our case, the pseudocyst extension occurred through the aortic hiatus. The fluid extension can also occur through the inferior vena cava hiatus or by directly penetrating through the diaphragm into the anterior or middle mediastinum. Most pseudocysts would be found to have communication with the main pancreatic duct if carefully investigated. 1
Pancreatic pseudocysts typically present with early satiety, abdominal pain, nausea, vomiting, jaundice, and bleeding. In contrast, mediastinal pseudocysts lack the typical symptoms and usually present with cardiopulmonary and upper Gastrointestinal (GI) symptoms such as dysphagia and dyspnea.5,6 Our patient complained of chest discomfort, difficulty swallowing, postprandial emesis, and fatigue. The presentation of chronic pancreatitis itself can be vague and overlooked. Our patient’s longstanding history of intermittent epigastric pain, diarrhea, and weight loss indicated the possibility of pancreatitis. However, she was never investigated and was misdiagnosed. Delayed diagnosis of mediastinal pseudocyst increases the risk of complications, including sepsis, airway obstruction, hemothorax, bleeding, congestive heart failure, cardiogenic shock, and thrombosis of nearby vessels.1,3,7,9,10 Differential diagnoses of pancreatic pseudocyst in the mediastinum include herniations, schwannoma, neurenteric cysts, paraspinal abscess, and meningocele. 7
Ultrasonography can detect pancreatic pseudocysts but is of less significance in cases of mediastinal counterparts. CT scan is an ideal modality for delineating a mediastinal pseudocyst and its connection with the pancreas and features of pancreatitis.5,9 MRI and Magnetic resonance cholangiopancreatography (MRCP) are alternative tools for diagnosis. A pseudocyst usually appears as a uniloculated and encapsulated fluid collection with homogenous density. 1 Pathologies in the pancreatic duct, such as stricture, dilatation, and communication with the pseudocyst, are better visualized with MRCP.5,9 Endoscopic ultrasonography is a recent advancement and has a therapeutic role.7,9 The CT scan of our patient showed an atrophic pancreas, pancreatic calcifications, dilated pancreatic duct, and a pseudocyst extending into the posterior mediastinum.
Repeated acute pancreatitis, trauma, hereditary pancreatitis, cystic fibrosis, and pancreatic duct stones are some of the possible causes of chronic pancreatitis. However, in this particular case, no underlying cause could be identified and hence labeled as idiopathic.
Management of the pseudocyst depends upon its location, size, relation to adjacent structures, communication with the pancreatic duct, symptomatology, and complications. 5 Spontaneous resolution occurs only rarely. Uncomplicated pseudocysts are usually drained through an endoscopic or percutaneous route. Excision by laparotomy or laparoscopy is ideal for pseudocysts that grow large or produce complications. 11 The patient in our case underwent surgical resection and pancreaticojejunostomy with good outcomes. Surgical removal of the pseudocyst was considered because of its large size and extraabdomial extension as well as the need for pancreaticojejunostomy.
Conclusion
Although uncommon, a mediastinal pancreatic pseudocyst should be considered in the differential diagnosis of individuals with dysphagia, vomiting, chest pain, anorexia, and features of pancreatitis. A detailed history is crucial in identifying the primary pathology and guiding further workup. Primary care physicians must rule out other potential causes of epigastric pain that could be mistaken for PUD.
Footnotes
Author contributions
SA, AS, AK, MG, AL, HU, K, and AQ performed draft writing and data analysis, literature search, and data acquisition. SA, AS, and AK contributed to images postprocessing. SA and AK supervised the activities. All authors participated in conceptualization, revision of the manuscript, and read and approved the final manuscript.
Data availability statement
The original contributions presented in this study are included in this article/supplementary material, further inquiries can be directed to the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
