Abstract
Acute necrotic pancreatitis is an emergency of evolution and is often unpredictable because of the potentially life-threatening complications it can cause. We report a unique case of a 56-year-old woman hospitalized for acute necrotic pancreatitis. The evolution of the latter was characterized by the occurrence of two very rare complications, of which the clinical presentations were atypical. The first complication was a gastroduodenal pseudoaneurysm compressing the main biliary tract and causing obstructive jaundice, which evolved well following percutaneous embolization. The second complication was a giant 20 cm pancreatic pseudocyst revealed by obstructive jaundice secondary to biliary compression, which progressed well following surgical treatment.
Introduction
Acute pancreatitis (AP) is one of the main reasons for gastrointestinal-related hospitalizations, and its incidence has been reported to be globally increasing.1,2
Acute necrotizing pancreatitis accounts for 10% of AP cases and is associated with higher mortality and morbidity. 3
This is due to the various complications, more or less frequent, such as parenchyma/peripancreatic fluid collections and necrosis or vascular complications, even rarer, including pseudoaneurysm.
Here we describe an unusual presentation of obstructive jaundice secondary to a gastroduodenal artery pseudoaneurysm and giant pancreatic pseudocyst following AP.
Case presentation
A 56-year-old woman, without any particular medical history, was urgently hospitalized for acute biliary pancreatitis, her staging abdominal computed tomography (CT) scan confirmed grade E pancreatitis of Balthazar with a CT severity index at 8.The initial course was favorable during the first 2 weeks with progressive improvement, until the reappearance of abdominal pain associated with obstructive jaundice. Laboratory studies showed hyper-bilirubinemia (total bilirubin 150 µmol/L) and a deglobalization from 11 to 8 g/dl. Abdominal CT scan with intravenous contrast revealed common bile duct compression by a pseudoaneurysm branching from the gastroduodenal artery (red arrow) measuring 5 × 5 cm (Figure 1) associated with a pseudoaneurysm of the left hepatic artery (white arrow).
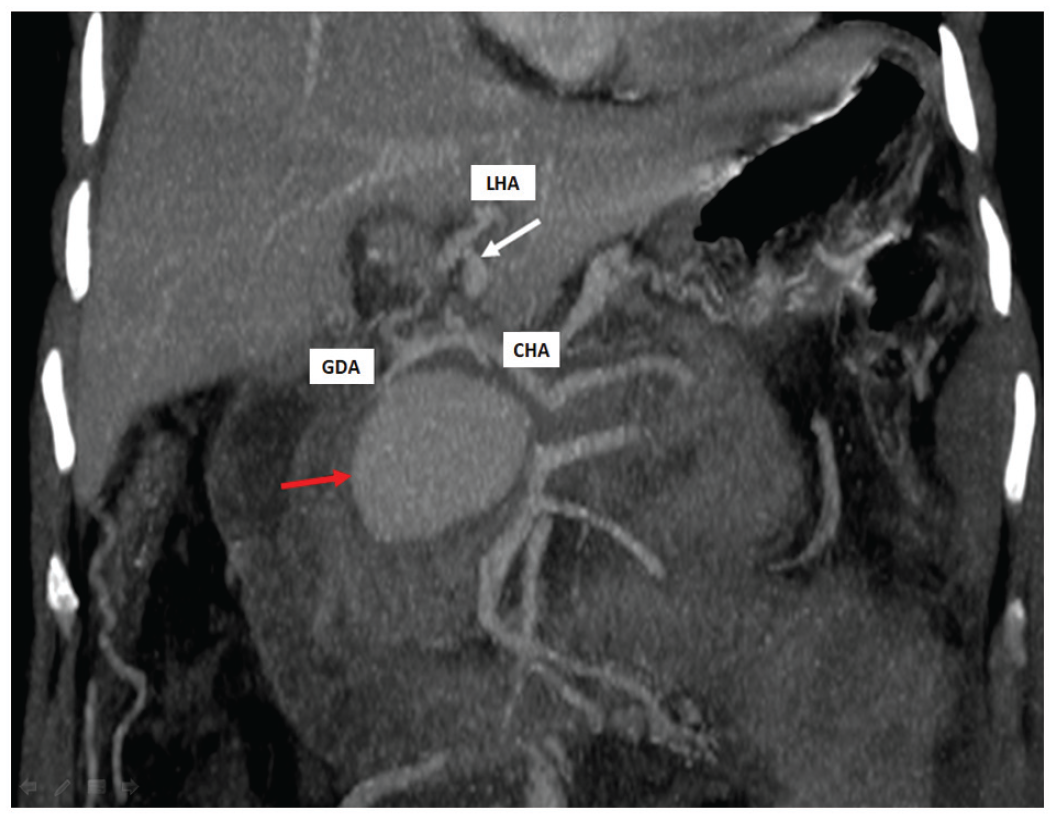
CT angiography showing the coronal cut of gastroduodenal artery pseudoaneurysm.
Angiography confirmed the CT scan results (Figure 2A), and coil embolization of inflow and outflow vessels and the pseudoaneurysm sac was performed (Figure 2B) with a favorable outcome.

(A) Visceral angiography showing the gastroduodenal artery (GDA) pseudoaneurysm (arrows) and the left hepatic artery pseudoaneurysm. (B) No contrast in the pseudoaneurysm after successful coil embolization.
Laparoscopic cholecystectomy was performed 4 months later without incident with simple postoperative care. Three months later, the patient was admitted with severe epigastria pain and vomiting. The CT scan of the abdomen showed a large cystic lesion compressing the common bile duct and occupying almost the entire abdominal cavity, measuring in size 20 cm × 19 cm (Figure 3).

CT angiography shows a giant pseudocyst compressing the common bile duct.
Magnetic resonance imaging (MRI) confirmed the diagnosis of pancreatic pseudocyst and showed the cystic formation along the main pancreatic duct. The patient was surgically managed by cystogastrostomy (Figure 4) with uneventful postoperative care and was discharged on the 5th postoperative day. She remained asymptomatic at 12-month follow-up.

Intraoperative images showed cystogastrostomy.
Discussion
Visceral artery aneurysms are divided into true or pseudoaneurysms
A pseudoaneurysm, which results from the erosion of the pancreatic or peripancreatic artery into a pseudocyst, is a rare vascular complication following pancreatitis, as was the case for the current patient, with an estimated prevalence of less than 10% of cases. 4
The most common locations are the spleen artery (60%), followed by the hepatic artery, involved in 20% of cases and the gastroduodenal location which is even rarer, occurring in only 1.5% of cases. 5
The most common clinical presentation is gastrointestinal bleeding secondary to rupture of the aneurysm (52%), while only 7.5% of cases remain asymptomatic. 6
Obstructive jaundice secondary to compressive hematoma or external pressure by the aneurysm, as was the case in the current observation, is extremely rare, has been only reported in a few cases within the literature.7–10
Performed in the setting of pancreatitis, a CT scan can clarify the origin of the aneurysm and its relationship to surrounding structures, providing data that are useful for planning a treatment strategy. 11
Once the diagnosis is made, angiography with coil embolization is recommended as the treatment of choice in line with the current observation. 5
Indeed, endovascular therapy offers significant advantages in terms of morbidity compared to surgery with proven effectiveness.
Unfortunately, the evolutionary history of necrotic pancreatitis does not end at this stage. Beyond the fourth week of evolution, necrosis of the peripancreatic tissue or parenchyma may cause liquefaction and subsequent organization resulting in pseudocysts. 12
The clinical presentation is quite variable including abdominal pain, surrounding organs compression, especially the main bile duct as was the case for our patient causing obstructive jaundice, or vascular occlusion. 13
Regarding the positive diagnosis, an abdominal CT scan showing a typical appearance in a patient with a history of acute or chronic pancreatitis is virtually pathognomonic of a pancreatic pseudocyst and does not require confirmation by another imaging modality. 12
However, MRI and magnetic resonance cholangiopancreatography, of which the interest is to specify the existence of communication with the pancreatic duct, are important contributions considering that they enable the therapeutic strategy to be adapted, as was the case with the current patient. 14
The term giant pancreatic pseudocyst is traditionally used when the size is greater than 10 cm, 15 but only a few cases of pseudocysts larger than 20 cm, as in our case, had been published in the literature.13,16,17
Therapeutic strategies include observation, endoscopic drainage, percutaneous drainage, and surgical interventions.
The choice of a particular treatment strategy must take into consideration the size and location of the cyst, correlation and dilatation of the main pancreatic duct whether it is symptomatic or not. 18
The patient, in our case, required interventions due to the pseudocyst being large and symptomatic. Despite the advent of endoscopic procedures, surgery still has a place in treating pancreatic pseudocysts, with high success rates and low morbidity and mortality. It still plays an important role in therapy with the development of the laparoscopic approach.
In literature review, few studies were found, mainly case reports, regarding the management of giant pseudocysts.
As for the endoscopic approach, a case report 13 was published in 2018 in which authors suggested internal endoscopic drainage as a possible initial method of management of a giant pseudocyst.
The other alternative, namely, surgical procedures, whether by laparotomy or laparoscopy, have shown excellent outcomes in managing giant pseudocysts.19,20
A Chinese publication 21 on eight patients with retrogastric pancreatic pseudocysts larger than 10 cm, treated by laparoscopic transgastric cystogastrostomy, concluded that this minimally invasive surgical procedure was easy, safe, effective, and had rapid recovery and low recurrence rate.
More recently, almost half of 14 patients with pancreatic pseudocysts larger than 10 cm were successfully managed by laparoscopic anterior transgastric cysto-gastrostomy. 22
This study also illustrated that this technique is a safe and effective method for managing appropriate retrogastric pancreatic pseudocysts.
Even if it seems obvious that laparoscopic transluminal drainage of pseudocyst offers advantage over conventional open surgery in terms of significantly reduced morbidity, less postoperative pain, and early recovery, 23 it is important to compare these two procedures in terms of outcomes.
A randomized clinical trial (RCT), comparing open surgical cystogastrostomy with endoscopic cystogastrostomy, has shown the relative success of both techniques but with a shorter hospital stay in the endoscopic group. 24
Another RCT comparing endoscopic and laparoscopic drainage of pseudocyst 25 concluded that both techniques had similar efficacy for internal drainage of suitable pancreatic fluid collections. The choice of procedure should depend on available expertise and patient preference.
Conclusion
This observation illustrates two very rare complications of AP occurring in the same patient, and of which atypical presentation made the diagnosis more difficult. Rapid diagnosis and early and appropriate treatment are essential to improve the prognosis.
Footnotes
Acknowledgements
Special thanks to Dr. Radhouane Toumi for his assistance.
Author contributions
M.B. and A.R. drafted the manuscript; Y.B.C. reviewed and reported the radiological finding; M.A.C. and Z.B.A. provided conceptual advice; M.S.J., F.H., and S.Y. revised the manuscript and validated the final version. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written Informed consent was obtained from the patient.
