Abstract
Posterior reversible leukoencephalopathy is a rare radio-clinical entity that has gained increasing recognition over the last two decades. It is associated with various etiologies: arterial hypertension, autoimmune diseases, chemotherapy, and immunosuppressive drugs. Several cases have already been reported following cancer therapy. Posterior reversible leukoencephalopathy is characterized by capital clinical signs (headache, seizures, confusional syndrome, and visual disorders) and radiological abnormalities (cerebral edema predominantly in the posterior regions). We report the case of a 38-year-old female patient diagnosed with posterior reversible leukoencephalopathy after receiving Carboplatin and Paclitaxel chemotherapy for recurrent cervical cancer, which was revealed by a generalized seizure. Brain magnetic resonance imaging showed T2 Flair hyper signals in the parieto-occipital regions. This complication is rare but is probably underdiagnosed due to a lack of awareness and limited hindsight. Rapid diagnosis is essential to prevent acute neurological complications, which can be life-threatening or functionally crippling regardless of neoplasia.
Keywords
Introduction
Posterior reversible leukoencephalopathy (PRES) is an uncommon neurological syndrome associated with a well-defined clinical picture and typical radiological imaging. Headache, nausea, visual disturbances, altered mental status, seizures, and hypertension are the most common signs. Neuroimaging usually reveals bilateral white matter edema of the posterior cerebral hemispheres. 1 Diagnosis holds significant therapeutic and prognostic implications, as reversibility of clinical and radiological signs is linked to early and effective control of blood pressure and/or discontinuation of the causative drug; delayed treatment can lead to irreversible neurological sequelae and even death. 2 After reviewing the literature, numerous cases have reported platinum salts and/or paclitaxel as causative agents of PRES. 3 In this case, we present a patient who developed PRES after the fourth cycle of carboplatin and paclitaxel for metastatic cervical cancer.
Case presentation
A 38-year-old female with no particular medical history has been followed since February 24, 2022 for locally advanced squamous cell carcinoma of the uterine cervix. The patient underwent radiochemotherapy, receiving concomitant cisplatin 40 mg/m2 along with 46 Gy of radiotherapy administered in 23 fractions, followed by brachytherapy consisting of 7 Gy delivered in three fractions. Subsequently, An18 F-fluoro-desoxy-glucose (FDG) PET/CT imaging was conducted, revealing a locally advanced hypermetabolic lesion at the uterine cervix suggestive of recurrent neoplasia. Additionally, a hypermetabolic mass was observed on the right side of the uterus, suspected to indicate secondary extension. Furthermore, moderately hypermetabolic lombo-aortic lymph nodes were detected, indicating lymph node extension of the known neoplasia. Given these findings, the patient underwent palliative chemotherapy: Paclitaxel 175 mg/m2 and carboplatin AUC 5, administered every 3 weeks. Nine days after the fourth chemotherapy course, the patient presented with medullary aplasia. Eleven days later, the patient presented with a generalized tonic-clonic seizure with urine leakage, post-critic amnesia, and ptosis of the right angle of the mouth, preceded by headaches and bilateral visual acuity reduction.
Clinical examination after seizure resolution revealed a conscious patient with a Glasgow Coma Scale score of 15/15, blood pressure was 140/60 mmHg, respiratory rate was 36 breaths per minute, and heart rate was 100 beats per minute. There was no fever and no neurological abnormalities noted, particularly no focal signs or visual field alterations.
A cerebral Computed tomography scan was performed and revealed two areas of cortico-subcortical hypodensity in the right parietal and bilateral occipital areas, with no clear contrast, and no intra- or extra-parenchymal hematoma.
A brain magnetic resonance imaging (MRI) was performed, revealing bilateral juxta and subcortical frontal, parieto-occipital, and cerebellar areas with cortical involvement, high signal on T2, and T2 fluid-attenuated inversion recovery (FLAIR), mostly high signal diffusion and apparent diffusion coefficient, but low signal intensity in the frontal subcortical, right parietal, and left occipital areas in the T1 sequence. Additionally, discrete gyral enhancement was observed post-contrast on the T1 sequence, suggestive of PRES (Figure 1).
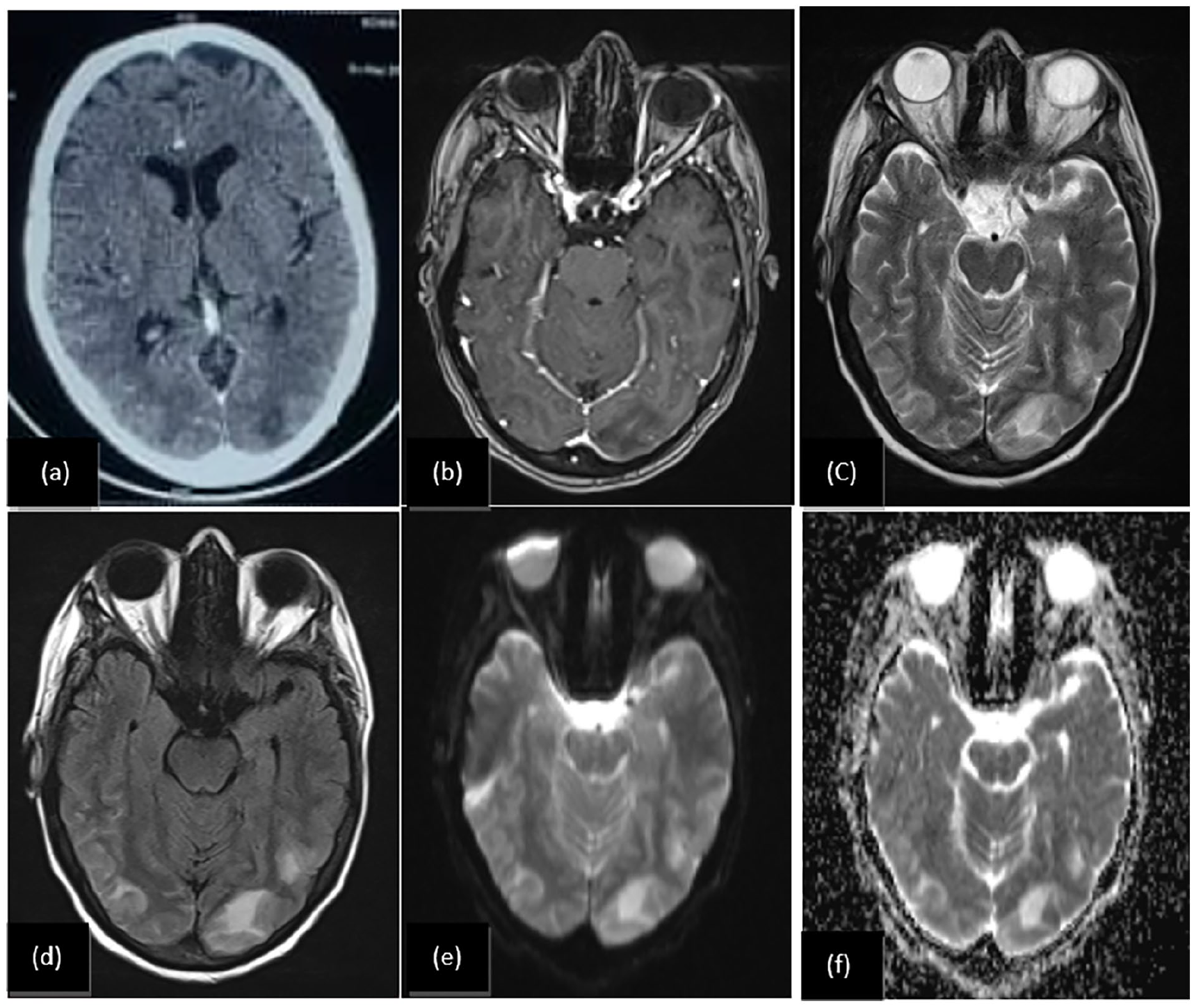
Brain axial CT with occipital hypodensity (a) and brain magnetic resonance imaging with post-contrast T1 (b) T2 (c) T2 fluid-attenuated inversion recovery (FLAIR) (d) apparent diffusion coefficient (ADC) (f) and diffusion (e) axial sequences: We noted bilateral juxta and subcortical parieto-occipital, with cortical involvement, T2 and FLAIR high signal ((c) and (d)), diffusion and ADC high signal ((e) and (f)), and low signal occipital area with discrete gyral enhancement post-contrast T1 (c), suggesting a posterior reversible leukoencephalopathy.
A biological assessment revealed the following results: hemoglobin 9.3 g/dL, neutrophils 4140/mm3, platelets 34000/mm3, C-reactive protein 39.9 mg/L, creatinine 8 mg/L, natremia 137 mmol/L, kalemia 3.8 mmol/L, corrected calcium 90 mg/L.
Due to the absence of clinical evidence of infectious meningitis and thrombocytopenia, a lumbar puncture was not performed. The case was discussed with neurologists, who concluded that it was consistent with PRES.
The patient was managed with symptomatic therapy, including blood pressure control and anticonvulsant therapy. Over a few days, her condition gradually improved with a decrease in headache intensity and an improvement in visual acuity. She was discharged from the hospital with a prescription for anticonvulsant treatment.
One month later, rapid progression of neoplastic disease led to the patient’s death; therefore, a control MRI was not performed.
Discussion
Physiopathology
The physiopathology of PRES remains debatable. Five theories have been proposed:
- The vasogenic theory posits that severe hypertension leads to an interruption in cerebrovascular autoregulation.
- Endothelial theory considers that PRES is mainly due to endothelial dysfunction caused by a systemic inflammatory state (toxins, sepsis, eclampsia, transplant, autoimmune disease).
- Cytotoxic theory states that endothelial dysfunction is induced by endotoxins (chemokines), or exotoxins (chemotherapeutic drugs or immunosuppressants).
- Immunogenic theory asserts that the first marker of endothelial dysfunction is T-cell-mediated inflammation accompanied by chemokine release.
- Hypersecretion of arginine vasopressin (AVP) or an increase in AVP receptor density. Activation of V1a vasopressin receptors would induce cerebral vasoconstriction, leading to endothelial dysfunction and cerebral ischemia. According to this theory, cytotoxic edema is induced by transglial flow dysfunction with increased endothelial permeability, generating vasogenic brain edema.
In the case of this patient, it appears that the cytotoxic mechanism may be involved. Numerous studies have demonstrated that cytotoxic agents can induce endothelial dysfunction, leading to the production of vasoactive substances, vascular leakage, and the development of cerebral edema. 4
PRES risk factors
PRES syndrome is frequently associated with arterial hypertension (essential hypertension, eclampsia, renal failure), but it can also manifest in sepsis, autoimmune diseases, or following neurosurgery: 20%–30% of cases described occur in the absence hypertensive context. Another pathophysiological explanation involves the direct toxicity of certain treatments (immunosuppressants, cytotoxics) via endothelial lesions caused by the production of proinflammatory cytokines. 2
Clinical features and radiological findings
The first acute neurological manifestations of PRES are usually disturbances of consciousness (confusion, lethargy, drowsiness, or coma), visual disturbances, and epileptic seizures; hypertension is also common. 5 In our case, the patient exhibited a convulsive seizure associated with a headache and bilateral visual acuity loss. A retrospective study involving 21 cases of PRES reported a CT diagnosis rate of only 40%, compared with 100% for MRI. Diffuse hypodensity was observed in CT images. 2 MRI typically shows cerebral edema without infarction, affecting the subcortical white matter bilaterally. Particularly in the parieto-occipital regions, MRI shows lesions with hypersignal on T2, T2 FLAIR, and diffusion sequences indicating vasogenic edema. 6 In 2015, three clinico-radiological criteria were suggested for the diagnosis of PRES: acute neurological signs, vasogenic edema on neuroimaging and reversibility of clinical and/or radiological signs. 4
Chemotherapy and PRES
An increasing number of molecules used in oncology have been reported to cause PRES, since it was first described in 1996. 7 These therapies include platinum salts (cisplatin, carboplatin, and oxaliplatin), antimetabolites (gemcitabin), folate antagonists, anthracyclines and vinca alkaloids. The neurotoxicity of platinum salts is frequent. 7 On average, the interval between the last chemotherapy and the appearance of the first symptoms is a few hours to a few weeks, particularly within the first three cycles of chemotherapy. 8 In our case, she developed this syndrome after the fourth cycle of carboplatin and paclitaxel at a three-week interval. Through our literature review, we identified 19 cases of patients experiencing PRES following the administration of chemotherapy drugs with or without targeted therapies, as detailed in Table 1.1–4,6–15
PRES induced by chemotherapy and targeted therapies reported in the literature.
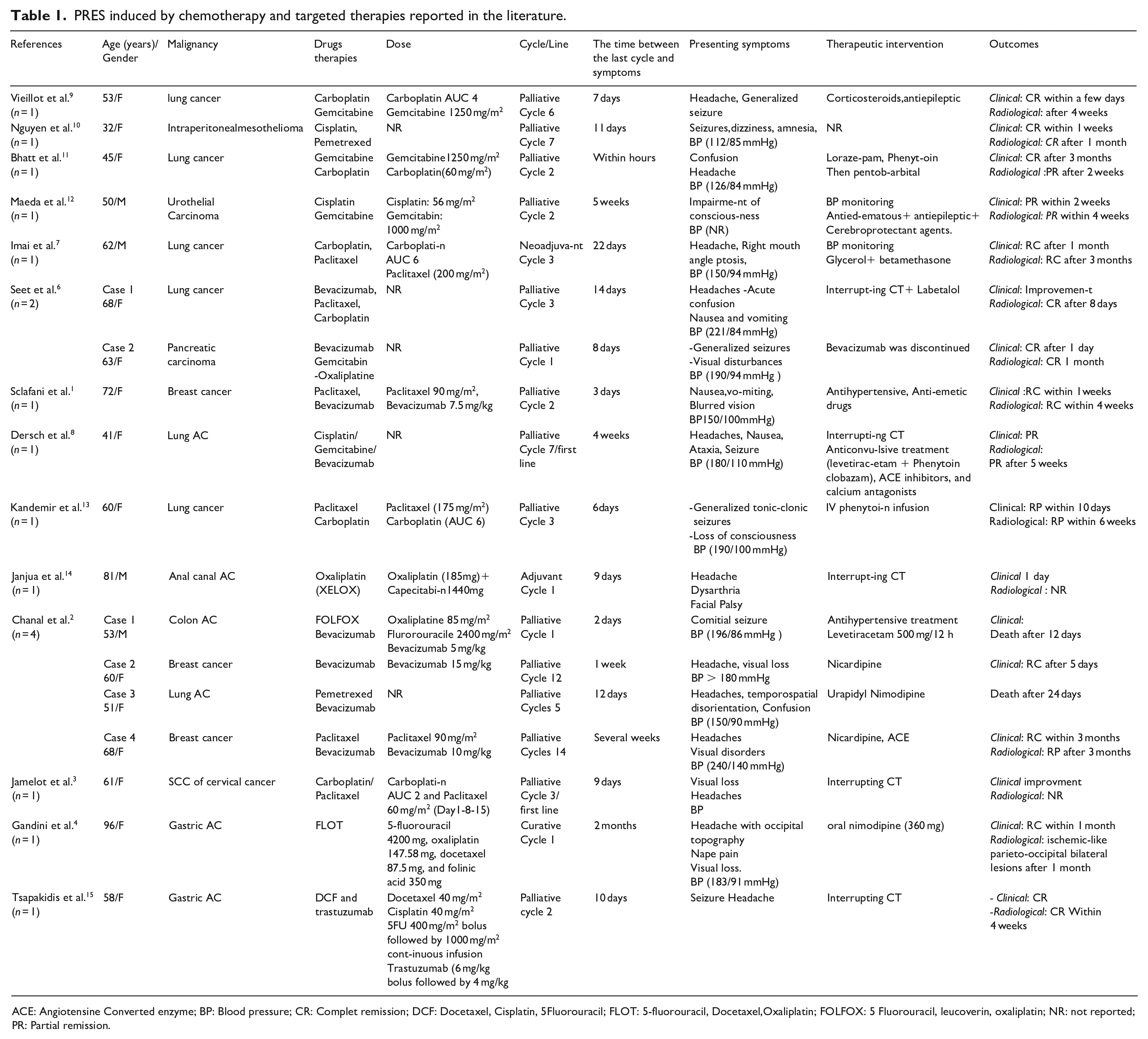
ACE: Angiotensine Converted enzyme; BP: Blood pressure; CR: Complet remission; DCF: Docetaxel, Cisplatin, 5Fluorouracil; FLOT: 5-fluorouracil, Docetaxel,Oxaliplatin; FOLFOX: 5 Fluorouracil, leucoverin, oxaliplatin; NR: not reported; PR: Partial remission.
Treatment
Stopping the triggering or aggravating factor is the first therapeutic measure, as is controlling blood pressure, which is the most important aspect of treatment. Antihypertensive treatment is based on three classes of molecules: calcium antagonists, β-blockers, and diuretics.
Treatment of neurological complications is essential; in the event of epileptic seizures, antiepileptic drugs should be rapidly introduced. 4 The use of corticosteroids to reduce inflammation is also described. 3 Our patient was managed with symptomatic therapy, including blood pressure control and anticonvulsant therapy.
Although PRES is reversible in most cases, failure to recognize the syndrome and correct its underlying cause can lead to severe central nervous system damage and even death. Therefore, it is essential to raise awareness of PRES among patients undergoing chemotherapy with the described drugs.
Conclusion
PRES is a clinic-radiological syndrome; the responsibility of chemotherapy, most commonly with platinum compounds, has been reported for several years. Rapid diagnosis is crucial across a spectrum of suspicious clinical and radiological features that must be rapidly recognized to anticipate the optimal therapeutic strategy and avoid unnecessary complications. Consequently, reporting rare adverse events secondary to chemotherapy is required for a better understanding of the different clinical spectrums.
Footnotes
Acknowledgements
Not applicable.
Author contributions
K.O. contributed to conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, visualization, writing original draft, writing review and editing; F.B. contributed to conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, visualization, validation, and writing original draft; N.B. contributed to supervision, validation; M.B. contributed to validation; T.C. contributed to validation; Z.B. contributed to validation; H.J. contributed to validation; N.T. contributed to validation; S.S. contributed to validation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representatives for anonymized patient information to be published in this article.
