Abstract
Enteric fever is a systemic bacterial infection caused by enteroinvasive, gram-negative bacilli, named Salmonella enterica serovar typhi and Salmonella enterica serovar paratyphi. It presents with hectic fever, headache, malaise, bowel habit changes, and abdominal pain. Diagnosis is usually confirmed by blood culture. Gastrointestinal complications of enteric fever include intestinal bleeding, bowel perforation, pancreatitis, and cholecystitis. We encountered a case of lower gastrointestinal bleeding (hematochezia) as a complication of enteric fever. A 35-year-old male patient presented to Aster CMI hospital, India, with an intermittent fever of 2-week duration associated with dry cough, loss of appetite, abdominal pain, and generalized body weakness. Four days after admission, he experienced three episodes of lower gastrointestinal bleeding. Upon physical examination, he was hemodynamically stable and had a high-grade fever, mild hepatomegaly, tipped splenomegaly, and lower abdominal tenderness. Blood culture grew Salmonella typhi. Abdominal ultrasound showed ileocolonic thickening with enlarged mesenteric lymph nodes. Abdominal computed tomography scan displayed enlarged mesenteric lymph nodes with surrounding fat strands. A colonoscopy revealed multiple shallow, punched-out, and punctate ileocolonic ulcerative lesions, with stigmata of active bleeding at caecal ulcers. Colonoscopy-guided biopsy suggested multifocal active colitis favoring infective etiology. Diagnosis of blood culture-confirmed enteric ulcer was made. He was treated with ceftriaxone 1 g iv twice daily for 10 days and rehydrated with intravenous fluids. Adrenaline injection was done at the site of bleeding ulcers, and hemostasis was secured. Other additional medications were antipyretics, anti-emetics, multivitamins, and proton pump inhibitors. He was fever-free on the third day of admission and discharged after 10 days of hospital stay. He was appointed to follow-up clinic after a week. He was completely healthy on the day of the first follow-up clinic visit and planned to resume his duties. Enteric fever remains a common public health problem in most developing countries. Early suspicion and prompt institution of appropriate antibiotics are crucial in the reduction of systemic and local complications of enteric fever. Since gastrointestinal complications of enteric fever are less often encountered in the antibiotic era, clinicians should be cognizant of an enteric ulcer as a cause of lower gastrointestinal bleeding.
Introduction
Enteric fever is a systemic bacterial infection caused by enteroinvasive, gram-negative bacilli, named Salmonella enterica serovar typhi (S. typhi) and Salmonella enterica serovar paratyphi (S. paratyphi). Worldwide, as of 2019, an estimated 9 million people get sick from enteric fever and 110,000 people die from it every year. The recently estimated annual incidence of enteric fever in sub-Saharan Africa was 762 cases per 100,000 person-years, which doubled that of India (360 cases per 100,000 person-years).1–4 It remains to be a common problem in the developing world. Humans are the only natural host reservoirs of Salmonella typhi. Hectic fever, headache, anorexia, constipation, diarrhea, and abdominal pain were the usual presentations of enteric fever. Gastrointestinal bleeding was the frequently observed local complication (10% of affected patients), followed by bowel perforation (3% of affected patients) before the antibiotic era.5–7 Nowadays, the gastrointestinal complications were reduced by early and effective antibiotic treatment. Here, we present a case of blood culture-confirmed enteric fever complicated by lower gastrointestinal bleeding.
Case presentation
A 35-year-old male patient was admitted to the medical ward, Aster CMI Hospital, India, with the complaint of high-grade intermittent fever of 2-week duration associated with loss of appetite, dry cough, generalized body weakness, and diffuse, moderate to severe abdominal pain which was not settling with analgesics. No history of vomiting, bowel habit change, or yellowish discoloration of the eye. He did not complain of hematuria, dysuria, urgency, or frequency of micturition. No history of headache, neck stiffness, or altered mentation. Upon physical examination, he was acutely sick looking with vital signs, blood pressure (BP) = 110/70 mmHg, pulse rate (PR) = 80 beats per minute, respiratory rate (RR) = 18 breaths per minute, temperature (T°) = 38.3°C, and body mass index = 23.3 kg/m2. He had no pallor of conjunctivae or icteric sclerae. There was no significant lymphadenopathy at accessible sites. There were no remarkable findings on the respiratory and cardiovascular systems. On abdominal examination, the abdomen was soft and moved with respiration. He had mild lower abdominal tenderness, but no rebound tenderness. Mild hepatomegaly and tipped splenomegaly were appreciated. Bowel sounds were audible. He had no costo-vertebral angle tenderness. There were no remarkable findings on neurologic examination. Laboratory values revealed white blood cell count = 5600/µl (neutrophils = 81%, lymphocytes = 13%) (normal range = 4.5–11.0 × 103/µl), hemoglobin (Hgb) = 12.1 gm/dl (normal range = 12–16 gm/dl), and platelet count = 202,000/µl (normal range = 150–450 × 103/µl). The erythrocyte sedimentation rate was 44 mm in the first hour (normal range = 0–22 mm/h). Liver biochemical tests showed alanine transaminase = 74 IU/L (normal value ⩽ 36 IU/L), aspartate transaminase = 94 IU/L (normal value ⩽ 40 IU/L), bilirubin (total) = 0.46 mg/dl (normal range = 0.3–1.2 mg/dl), bilirubin (direct) = 0.22 mg/dl (normal range ⩽ 0.3 mg/dl), alkaline phosphatase = 70 IU/L (normal range = 50–150 IU/L), total protein = 6.1 gm/dl (normal range = 6.0–8.0 gm/dl), and albumin = 3.4 gm/dl (normal range = 3.5–5.5 gm/dl). Serum Cr = 0.81 mg/dl (normal range = 0.6–1.2 mg/dl), serum K+ = 3.6 meq/L (normal range = 3.5–5.5 meq/L), and serum Na+ = 130 meq/L (normal range = 135–145 meq/L). QuantiFERON-TB Gold test was negative. The Widal test was negative. Blood culture grew Salmonella typhi. Ultrasound of the abdomen showed thickening of the terminal ileum, ileocecal valve, and caecum, and enlarged mesenteric lymph nodes (Figure 1). The abdominal CT scan displayed multiple enlarged mesenteric lymph nodes with surrounding fat strands (Figure 2). Diagnosis of blood culture-proven enteric fever with gastrointestinal complication (enteric ulcers) was made, and started on antibiotics, ceftriaxone 1 g iv twice daily and azithromycin, 500 mg po daily. The fever spike continued, hence antibiotic was escalated to meropenem, 1 g iv thrice daily until drug-sensitivity testing (DST) was available. He was continued on ceftriaxone to complete 10 days of treatment after Salmonella typhi was sensitive to ceftriaxone. Other medications were intravenous fluids (75–100 ml/h), paracetamol, 1 gm iv prn, pantoprazole, 40 mg iv daily for 5 days, ondansetron, 4 mg iv prn, providac (probiotics) daily, and zincvit tablets daily. His temperature starts to slow down after 72 h of antibiotics. After 4 days of admission, he developed three episodes of frank, red, rectal bleeding (hematochezia) over a day, but had no shortness of breath, change in urine volume, or altered mentation. On repeated physical examination, he was conscious and oriented. Vital sign was BP = 100/60 mmhg, PR = 90 bpm, RR = 19 bpm, T° = 37.2°C. He had no pallor of conjunctivae. The buccal mucosa was moist. No remarkable findings on cardiovascular examination. He had no new findings on abdominal examination. A colonoscopy was done after bowel preparation for the complaint of hematochezia, which showed multiple transverse, shallow, punched-out, and punctate ulcerative lesions in the terminal ileum, ileocecal valve, and caecum, respectively. Caecal ulcers showed stigmata of recent bleeding (Figure 3). Colonoscopy-guided biopsy from the caecal ulcers revealed multifocal active colitis favoring infective etiology. Colonic mucosa showed multifocal ulceration. Granulation tissue with mixed inflammatory infiltrate was noticed in lamina propria. Focal neutrophilic cryptitis was seen. There was no evidence of crypt architectural distortion. Mallory cells (macrophages engulfed lymphocytes, bacilli, and red cells) were not seen. No evidence of parasites, granuloma, dysplasia, or malignancy (Figure 4). Diagnosis of ileocolonic enteric ulcers was made. Adrenaline injection was done at bleeding ulcer sites, and hemostasis was secured. He was hemodynamically stable and had no episodes of lower gastrointestinal bleeding. He was discharged home after 10 days of stay in the hospital, and appointed to follow-up clinic after a week. He was completely healthy on the day of the first follow-up clinic visit and planned to resume his duties.

Thickening of the ileum with enlarged mesenteric lymph nodes.

Multiple enlarged mesenteric lymph nodes around the ileum and ileocaecal junction.

Multiple transverse shallow, punched-out, and punctate ulcers in the terminal ileum, ileocaecal valve, and caecum, respectively.
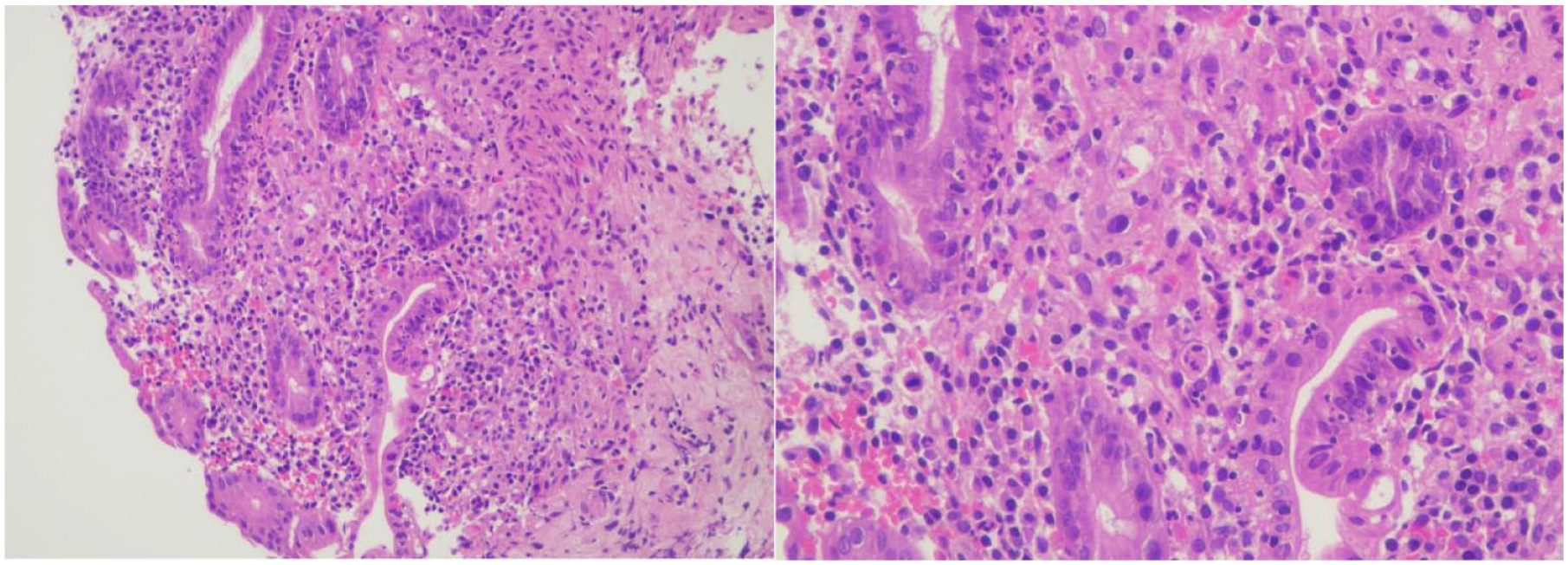
H&E image (10× and 40× magnifications) showing colonic mucosa with neutrophilic infiltration of crypts and surface epithelium. No parasites, granuloma, or dysplasia were seen.
Discussion
Enteric fever is caused by an enteroinvasive, gram-negative bacteria, named Salmonella typhi and Salmonella paratyphi. Enteric fever remains a public health problem in most developing countries due to poor hygiene and sanitation, overcrowding, lack of access to safe water, and impoverishment.1–4 Humans are the only natural host reservoirs of Salmonella typhi. The host acquires the organism via fecal-oral route from contaminated food or water. The ingested organism penetrates the intestine wall, invades submucosal Peyer’s patches, and reaches the reticuloendothelial tissues (liver spleen, lymph nodes, bone marrow) via the lymphatic and hematogenous route. The bacilli proliferate intracellularly within the reticuloendothelial tissue and then disseminate to involve various organs to cause symptomatic disease. Re-infection of intestinal Peyer’s patches occurs following the release of organisms into bile from reticuloendothelial tissues. Hyperplasia of Peyer’s patches occurs following re-infection and replication of Salmonella typhi, and results in intestinal ulceration, bleeding, necrosis, and ultimately full thickness perforation.5–7 Clinical presentation occurs after 7–21 days of microbial exposure and includes high-grade fever, headache, chills, malaise, constipation, diarrhea, and abdominal pain. Other atypical presentations include pneumonia, endocarditis, myocarditis, cholecystitis, osteomyelitis, meningitis, and orchitis. Inappropriate use of antibiotics led to the development of multidrug-resistant (MDR) strains and extensively drug-resistant strains. Complication rates were higher in MDR strains than in sensitive strains.1–5 Gastrointestinal bleeding and bowel perforation were the frequently observed complications before the antibiotic era and were usually noticed in the second and third weeks of illness. Studies reported that these complications are rarely encountered nowadays due to timely and effective antibiotic treatment.8,9 Our patient presented with hematochezia after 2 weeks of systemic symptoms onset. Diagnosis of enteric fever is confirmed by isolation of Salmonella typhi from blood, bone marrow, stool, and urine specimens. Blood culture is a diagnostic investigation of choice and is often positive in the first week of illness. Stool and urine have a high diagnostic yield in the second and third weeks of illness.5–7 In our patient, blood culture grew Salmonella typhi after 1 week of systemic illness. Serological tests such as the Widal test for ‘febrile agglutinins’ are available, but have little diagnostic value as they are neither sensitive nor specific to diagnose enteric fever. Third-generation cephalosporin is the first choice in the empirical treatment of enteric fever. Azithromycin is an alternative drug of choice.6,7 The patient was scheduled to complete the 10-day course of intravenous ceftriaxone after DST. World Health Organization introduced and prioritized typhoid conjugate vaccines (Ty21a and Vi CPS) for children and adults younger than 45 years in countries with the highest burden of typhoid disease or high levels of antibiotic resistance to Salmonella typhi. 10 Colonoscopy is the procedure of choice to localize the site of intestinal bleeding. The usual site of gastrointestinal bleeding in enteric fever is the terminal ileum followed by the ileocecal valve, owing to the abundance of submucosal lymphoid follicles (Peyer’s patches). Other less frequent sites of bleeding include caecum, ascending colon, and transverse colon, while the left-sided colon is usually spared.11–13 Colonoscopy of the patient visualized multiple shallow, punched-out, and punctate ulcers in the terminal ileum, ileocecal valve, and caecum, which were representative findings of enteric ulcers. Colonoscopic differential diagnoses of punched-out appearing enteric ulcers include intestinal tuberculosis, Yersinia enterocolitis, amoebiasis, and Crohn’s disease. 13 Majority of gastrointestinal bleeding due to enteric ulcers is managed conservatively with appropriate parenteral antibiotics and hemodynamic support. Life-threatening bleeding requires invasive measures including angiographic coil embolization, endoscopic therapy (adrenaline injection, thermal coagulation, or hemoclipping, alone or in combination), and surgical interventions (hemicolectomy or segmental resection).6,9,12,14,15
Conclusion
Enteric fever remains a common public health problem in most developing countries. Early suspicion and prompt institution of appropriate antibiotics are crucial in the reduction of systemic and local complications of enteric fever. Since gastrointestinal complications of enteric fever are less often encountered in the antibiotic era, clinicians should be cognizant of an enteric ulcer as a cause of lower gastrointestinal bleeding.
Footnotes
Acknowledgements
We are grateful to the medical personnel who were caring for the patient.
Author contributions
Z.A. and N.B. diagnosed and treated the patient, and wrote the draft of the case report. A.T. took a major part in drafting, critically revising, and reviewing of the case report. All authors gave final approval of the case report to be published.
Availability of supporting data
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual(s) case reports or case series.
Informed consent
Informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
