Abstract
Acute generalized exanthematous pustulosis is a rare severe cutaneous adverse reaction that classically presents in intertriginous or flexural areas and subsequently spreads diffusely across the trunk and extremities. To date, few cases of acute generalized exanthematous pustulosis arising in a photodistributed pattern are documented. Herein, we describe the second known case of photodistributed generalized exanthematous pustulosis arising in association with oral terbinafine use, providing a summary of the previously documented cases along with exploration of the potential pathophysiological mechanisms for this cutaneous reaction.
Keywords
Introduction
Acute generalized exanthematous pustulosis (AGEP) is a severe cutaneous adverse reaction (SCAR) characterized by non-follicular, sterile pustules overlying confluent erythema. Overwhelmingly, most cases are drug-induced with antibiotics, antifungals, antimalarials, anticonvulsants, analgesics, and calcium channel blockers implicated as the major causative agents.1–4 Classically, AGEP arises in intertriginous sites and generalizes to the trunk and extremities.1,2 Patients may experience fever and lab abnormalities, including leukocytosis, neutrophilia, eosinophilia, hypocalcemia, transaminitis, elevated creatinine, and elevated C-reactive protein (CRP).1–3 Herein, we present a case of terbinafine-induced, photodistributed AGEP.
Case
A 48-year-old female presented for an eruption beginning 8 days after initiation of oral terbinafine. While taking the medication, she traveled to the beach and had frequent sun exposure. While on vacation, she noted a pruritic, red rash in her inguinal creases, which quickly spread to her trunk and extremities. She denied prior use of terbinafine.
On examination, erythematous papules and plaques were noted diffusely on the trunk and extremities with clusters of small, non-follicular pustules on the arms and chest in a photodistributed pattern with sharp demarcation at areas covered by clothing (Figures 1–3). Laboratory studies were significant for neutrophilic leukocytosis, elevated CRP, and mild uncorrected hypocalcemia. Biopsy showed epidermal acanthosis and spongiosis with subcorneal pustules and dense dermal inflammation with eosinophils and neutrophils (Figures 4 and 5). Based on these findings, the patient was diagnosed with AGEP with a EuroSCAR score of 9. 1 She improved with a 5-day course of intravenous dexamethasone and twice daily application of betamethasone dipropionate 0.05% ointment. After 1 week, all pustules resolved with residual desquamation.

Clinical photograph at initial presentation, which demonstrates the striking photo-accentuation of the eruption. Coalescing erythematous patches and plaques on the arms and upper chest are seen with sharp demarcation at areas previously covered by the patient’s clothing. Scattered small papules are noted on the photoprotected sites.
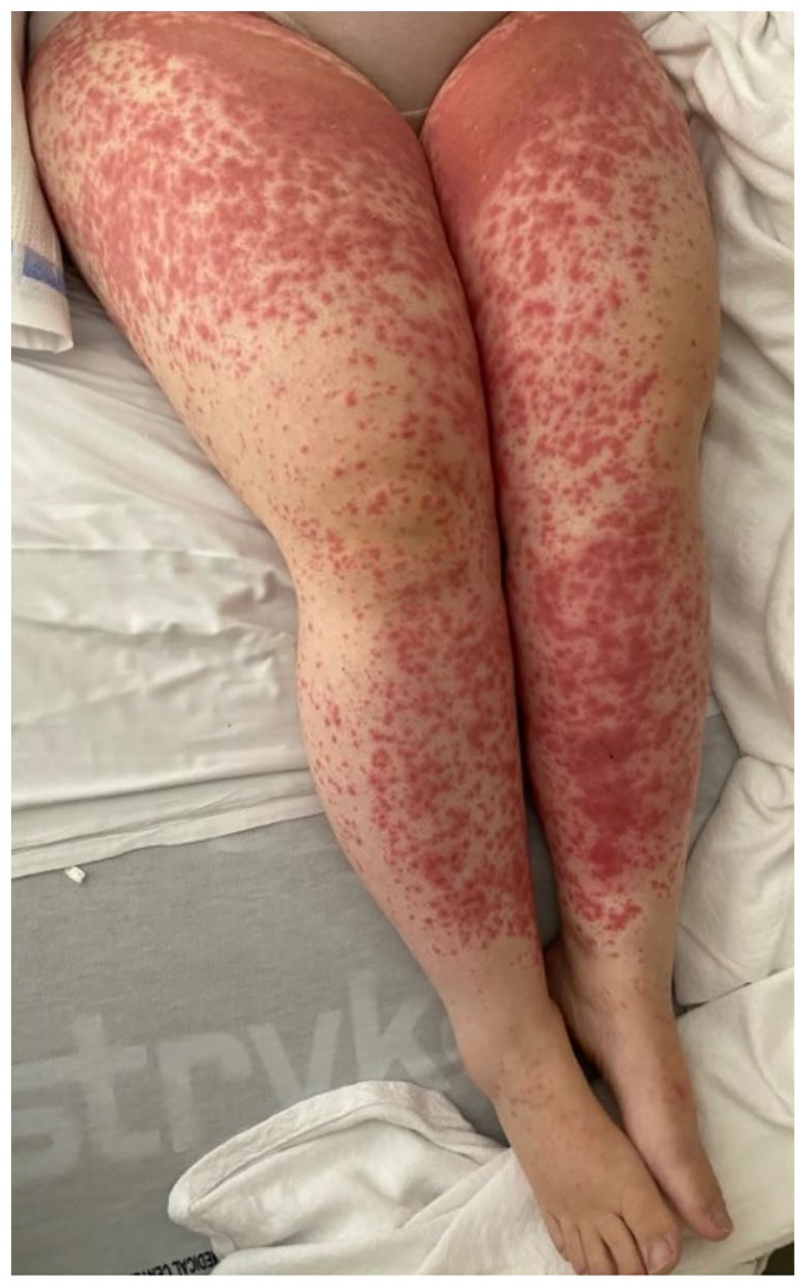
Clinical photograph showing diffuse coalescing erythematous papules and plaques diffusely on the anterior thighs and shin, sparing the feet and ankles previously covered by socks and shoes.

Two-panel photograph of bilateral proximal arms showing large, erythematous plaques studded with numerous pustules.

Punch biopsy of skin (hematoxylin and eosin stain, 4× magnification) shows epidermal acanthosis and spongiosis with a mixed dermal inflammatory infiltrate containing eosinophils and neutrophils.

Higher magnification of the histology specimen (hematoxylin and eosin stain, 10× magnification) demonstrates a spongiotic epidermis with subcorneal neutrophilic abscesses.
Discussion
Putatively, a drug-specific T-cell mediated type IV hypersensitivity reaction typifies AGEP. 5 The formation of pustules is driven by activation of innate and reactive immune responses. Specifically, upon exposure to the inciting agent, drug-specific T-cells migrate to the skin, leading to production of numerous cytokines and chemokines. T-cell–derived C-X-C motif chemokine ligand 8 (CXCL8) recruits neutrophils to lesional skin, while granulocyte-macrophage colony-stimulating factor prevents apoptosis of recruited neutrophils, supporting their accumulation.2,5,6 Upregulation of interleukins (IL) such as IL-17 and IL-22 stimulate keratinocyte production of additional CXCL8 and IL-8, synergistically contributing to chemotaxis and amassment of neutrophils in the stratum corneum and epidermis, as observed in lesional AGEP tissue specimens.5,6 Production of additional inflammatory cytokines such as tumor necrosis factor-alpha, IL-1, IL-6, IL-12, and IL-23 further augment recruitment and activation of neutrophils. 5 A genetic predisposition may underpin the development of AGEP as human leukocyte antigens B51, DR11, and DQ3 are more frequently observed in patients with AGEP. 7 Moreover, mutations in the IL-36RN gene, encoding the IL-36 receptor antagonist, may be responsible for the increased downstream production of IL-1, IL-6, and IL-8.5,6 However, the role of ultraviolet (UV) in triggering or exaggerating AGEP has not been established.
AGEP arising in a photo-distributed or photo-accentuated pattern is rare, with only a few existing reports in the medical literature attributed to hydroxychloroquine-psoralen plus ultraviolet-A light therapy, sertraline, liraglutide, and antimicrobials, as outlined in Table 1, leading some to refer to this entity as acute photodistributed exanthematous pustulosis.8–14 Our case further supports UV exposure as a potential exacerbating factor for terbinafine-induced AGEP.
Summary of prior case reports in the medical literature describing photodistributed, drug-induced AGEP.

Source: Table partially adapted from the table by Knoell and Lynch. 12
ANA: antinuclear antigen; CRP: C-reactive protein; EBV: Epstein-Barr virus; ESR: erythrocyte sedimentation rate; HSV: herpes simplex virus; IV: intravenous; PUVA: psoralen plus ultraviolet-A light; SSA: anti-Sjögren syndrome-related antigen A; SSB: anti-Sjögren syndrome-related antigen B.
In pharmacokinetic studies, terbinafine, an allylamine antifungal, distributes to and concentrates in peripheral body fluids and tissues with a predilection for sebum and skin.15,16 Additionally, the drug’s long terminal half-life has a slow rate of redistribution back to the central plasma from the skin, maintaining cutaneous fungicidal concentrations for extended periods beyond drug discontinuation.15,16 Despite not being classically listed among photosensitizing agents, photosensitivity reactions are recorded as possible adverse effects in the package insert for oral terbinafine. 16
Photosensitizing medications act as exogenous chromophores, absorbing photons from ultraviolet radiation and inducing either a phototoxic or photoallergic reaction.17,18 Photoallergic reactions are less common and typically require prerequisite sensitization to the medication. This response is immunologically mediated wherein a photoproduct acts as a hapten or antigen, eliciting a type IV hypersensitivity reaction.17,19–21 Photoallergic reactions develop ⩾24 h after drug exposure and present as eczematous dermatitis, which may extend to photoprotected sites. Classically, epidermal spongiosis and perivascular inflammatory infiltrates are observed on histology.17,20,21 In contrast, phototoxic reactions are nonimmunologic processes occurring in a dose-dependent manner in any individual exposed to a specific threshold of either drug and/or UV of the appropriate wavelength and without the need for prior sensitization.17,20,21 Pathologically, the generation of photoproducts triggers reactive oxygen species, resulting in cellular injury.17,20 Phototoxic reactions present as pruritic and painful sunburn-like reactions within minutes to hours after UV exposure and demonstrate epidermal keratinocyte necrosis with lymphocytic and neutrophilic dermal infiltrates.17,20,21 After discontinuation of the offending medication, both reactions typically resolve, but in rare instances, photoallergic reactions may persist. 17 However, distinguishing these entities poses challenges as clinical overlap in their features is often observed, and drugs may induce both reactions.17,21 While measuring the minimal erythema dose and photo patch testing may be utilized to assist in distinguishing the diagnoses, especially in cases where continuation of the drug is deemed medically necessary, such testing is not indicated in the majority of patients where simple drug avoidance will suffice in preventing recurrence.17,21
In two prior reports describing terbinafine-induced photodistributed dermatoses, a photoallergic mechanism was proposed based on continued worsening of dermatitis after cessation of drug exposure and the results of photopatch testing, while the other suggested a phototoxic pathway as more likely.18,19 Interestingly, the first case was likely sensitized prior to the use of topical terbinafine. 9 A third case reported solar urticaria resulting from terbinafine exposure. 22 While medications previously implicated in photodistributed AGEP (hydroxychloroquine, sertraline, fluoroquinolones) are well-documented causes of photoallergic or phototoxic reactions, cases of photosensitivity reactions due to terbinafine are rarely reported in the literature.4,10,18,19,21,22
In some instances, terbinafine ingestion may trigger UV-induced photoantigen formation, leading to a type IV hypersensitivity reaction typified by drug-induced photoallergic reactions. 20 While such a mechanism could explain our patient’s presentation based on timing and spongiotic histology, our patient’s eruption began in her groin and then became markedly accentuated in sun-exposed sites, arguing against both phototoxic and photoallergic processes. Another possible pathomechanism serves as the proposed pathway in drug-induced subacute cutaneous lupus erythematosus (SCLE), for which terbinafine is a well-documented offender. In this entity, the induction of photosensitivity by terbinafine is hypothesized to result in the formation of cutaneous lesions of SCLE via a photopharmacological isomorphic (Köebner) response in immunogenetically predisposed individuals. 23 Due to the initial presentation in a photoprotected site, our patient’s clinical course suggests photo-aggravation of AGEP as opposed to a primary photo-induced process, making an isomorphic mechanism the most plausible explanation. Similar patterns of photo-accentuation of AGEP that began classically in intertriginous sites are also documented with sertraline. 9 However, further investigation into the physiologic response to UV exposure in the setting of terbinafine ingestion is warranted to fully elucidate the underlying etiology of terbinafine-induced photodistributed cutaneous eruptions.
To the authors’ knowledge, this case is one of only two reports in the existing medical literature documenting terbinafine-induced AGEP with a photodistributed pattern, highlighting this rare presentation of an otherwise well-described SCAR. While gaps remain in our understanding of the role that UV plays in the pathophysiological mechanisms of AGEP, this case is an important reminder that clinicians should counsel patients to utilize diligent photoprotection and avoid artificial UV sources while taking terbinafine.
Footnotes
Acknowledgements
We are grateful to Jeffrey P. Zwerner, MD, PhD, for his assistance with obtaining the histology photos and descriptions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent
The patient provided written consent to publish the case report and these images.
