Abstract
Granuloma annulare is a benign, inflammatory condition of unknown etiology, characterized by erythematous annular plaques, frequently on distal extremities. Generalized granuloma annulare can be difficult to treat, with varying success in therapeutic approaches. We present the case of a 59-year-old female with refractory generalized granuloma annulare successfully managed with adalimumab, requiring ongoing 40 mg q2weekly treatment for 2 years. While there are a handful of published case reports/series suggesting that adalimumab can be used to treat generalized granuloma annulare, dosing regimens and the need for long-term use remain inconsistent. This case adds further evidence for considering adalimumab as a sustained therapeutic option for recalcitrant generalized granuloma annulare. The patient responded to adalimumab, a tumor necrosis factor-alpha antagonist, administered biweekly for a year, then switched to weekly intervals. Most granuloma annulare lesions improved within 2 months and continued to improve throughout the treatment. Adalimumab may be proposed as a therapeutic treatment for recalcitrant forms of generalized granuloma annulare.
Introduction
Granuloma annulare (GA) is a poorly understood, benign inflammatory granulomatous skin condition. GA can manifest in different forms, the most common being localized and generalized GA, and less frequently as perforating, patch, and subcutaneous GA. 1 Generalized GA is more difficult to manage, as lesions are more resistant to treatment, less likely to resolve spontaneously, and may have significant impacts on a patient’s quality of life. 2 Currently, there is no gold standard for treatment, with numerous proposed therapies ranging from localized to systemic therapies such as biologics, systemic drug therapy, and immunosuppressants. 3 All treatments have been used anecdotally and with variable degrees of success. We report a case of generalized GA improving following the use of adalimumab, a tumor necrosis factor – alpha (TNF-α) antagonist, for over a year in an outpatient dermatology clinic.
Case
A 59-year-old woman with a 3-year history of generalized GA was referred for a dermatologic assessment. Upon examination, the patient had erythematous annular plaques distributed to her upper and lower extremities and trunk (Figure 1). Past medical history included hypertension. Prior to presenting, the patient had undergone several failed therapies, including hydroxychloroquine, methotrexate 20 mg, intralesional kenalog (ILK) injections 2.5 mg/mL, and ultraviolet light-B phototherapy, many of which were prescribed in combination. This led to the initiation of adalimumab (Humira) 40 mg q2 weeks off-label in addition to ILK 2.5 mg/mL in April 2021. At a follow-up 2 months after initiating the treatment regimen, there was a significant improvement in the GA lesions, with decreased erythema and flattening of the lesions and no adverse events. At the follow-up 10 months later, she continued to have improvement; however, she had developed new lesions on her arms, neck, and face; thus, treatment was altered to adalimumab 40 mg qweekly and methotrexate 20 mg weekly, with ILK as needed (PRN). In the follow-up 9 months after the change in treatment (almost 1.5 years after initiating adalimumab), the patient noted stopping methotrexate due to feeling fatigued. She also had no new active lesions and only mild post-inflammatory hyperpigmentation (Figure 2).
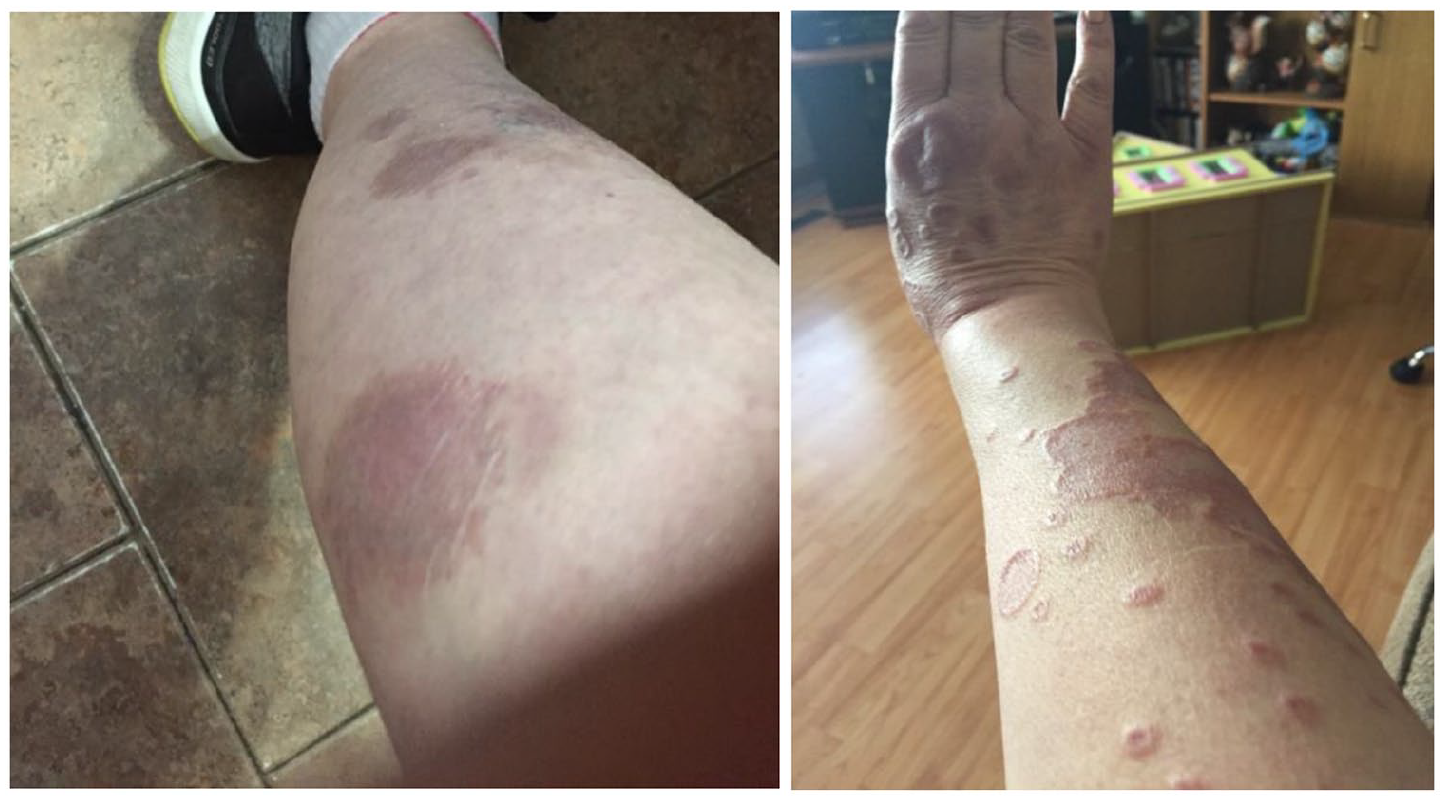
Disseminated distribution of erythematous plaques on lower and upper extremities prior to initiating treatment with adalimumab.

Mild post-inflammatory maculated hyperpigmented residuum after 18 months of treatment with adalimumab.
Discussion
GA was first described by Colcott Fox in 1895 as an asymptomatic or mildly pruritic condition with annular skin-colored or erythematous papules and plaques with a female predilection. 4 It can occur anywhere on the body but most commonly on the hands and feet. 1 Although the pathogenesis is not well understood, histological findings reported delayed-type hypersensitivity reactions and cell-mediated immune responses due to histiocytic and lymphocytic granulomatous dermal infiltrate. 5 GA’s inflammatory infiltrate is composed of CD3+ cells and macrophages, which secrete TNF-α. 6 As a foreign-body granuloma formation induced by an unknown inciting antigen is suspected.
There are multiple different subtypes of GA. Localized GA is the most classic subtype of GA, typically presents with few lesions, affects a population under 30 years, and is self-limiting with spontaneous resolution within 2 years. It usually does not require therapy unless the patient wishes to pursue treatment for cosmetic or symptomatic reasons. 6 Generalized GA is the presence of 10 or more lesions. It is characterized by a more widespread involvement of the trunk and extremities, with a bimodal distribution, mostly occurring in children younger than 10 years or adults older than 40 years.7,8 Generalized GA is less likely to resolve spontaneously and more difficult to manage, as lesions are more resistant to treatment. 9 Although various therapeutic modalities have been described in the literature, there is a lack of definitive data demonstrating efficacy, highlighting the need for alternative, more efficacious, and safe treatment options.
Currently, there are few successful cases of treatment of generalized GA with TNF-α antagonist reported. One systemic review found that in previously reported cases of generalized GA treated with biologics, 14 out of 16 patients treated with adalimumab demonstrated improvement or resolution of GA. 10 The efficacy of these drugs is related to the presumed role of TNF-α in the GA pathogenesis. The biggest challenge to this medication is that, on rare occasions, TNF-α antagonists may induce auto-immune diseases and increase the incidence of infectious diseases (including tuberculosis reactivation). 10
In the present case, adalimumab treatment enabled a fast improvement of generalized GA that had been refractory to previous systemic therapies. When the condition later flared despite being on treatment, increasing the dosing interval resulted in recapture, and adalimumab ultimately has been able to serve as long-term maintenance therapy. To date, no adverse effects have been noted by our patient. The case demonstrates that TNF-α antagonist, adalimumab, may be an effective treatment to implement following previous failed treatment attempts using topical or systemic therapies.
Conclusion
We suggest TNF-α antagonists such as adalimumab may be an efficacious and well-tolerated therapeutic option for forms of recalcitrant generalized GA preceding failed systemic or topical treatments. If patients flare following initial response increasing the dosing interval from every 2 weeks to weekly can recapture patients. Finally, some patients may not achieve remission, but adalimumab can be used as maintenance therapy for generalized GA with good results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent
Informed consent was obtained for the publication of the details in this report.
