Abstract
Erosive lichen planus is a chronic auto-inflammatory disease which affects the stratified squamous epithelia resulting in painful ulcerations of both the skin and mucosal surfaces, and has a known malignant potential. Management of erosive lichen planus has proven to be difficult; however, recent reports of treatment with Janus kinase inhibitors such as Upadacitinib, are encouraging. This report outlines the third reported case of erosive lichen planus to be successfully treated with Upadacitinib in a 70-year-old woman with treatment-resistant disease. In addition, we report the complication of oral squamous cell carcinoma which became apparent once the extensive erosive lichen planus had healed. This case report highlights the importance of monitoring for mucosal squamous cell carcinoma in areas affected by erosive lichen planus, as squamous cell carcinoma can mimic the erosions of erosive lichen planus.
Introduction
Erosive lichen planus (ELP) is a chronic auto-inflammatory disease which affects the stratified squamous epithelium resulting in painful ulcerations of both the skin and mucosal surfaces. Management of ELP has proven to be difficult; however, recent reports of treatment with Janus kinase inhibitors (JAKi) such as Upadacitinib, are encouraging. This case reports the successful treatment of ELP with Upadacitinib in a 70-year-old woman with recalcitrant disease. This is the third known case of oral lichen planus (LP) that has been successfully treated with Upadacitinib; 1 however, we report the added complication of oral squamous cell carcinoma (SCC) which became apparent once the extensive ELP had healed.
Case report
A 70-year-old woman presented with a 6 year history of recalcitrant ELP. She had extensive, painful erosions and ulcerations of the oral mucosa, tongue, oropharynx, and esophagus (Figure 1) despite treatment with multiple systemic agents. These agents included: methotrexate, azathioprine, secukinumab, cyclosporine, sulfasalazine, intravenous immunoglobulin (IVIG), acitretin, hydroxychloroquine, and systemic steroids from which she had had been unable to wean without severe flares of ELP.

Upper endoscopy performed prior to initiation of treatment with Upadacitinib. Erosions, ulcerations and strictures were present within the esophagus at this time.
Treatment with standard dosing of Upadacitinib, 15 mg daily, in conjunction with methotrexate was initiated and she rapidly showed improvement with decreased strictures, improvement in erosions of the esophagus, and almost complete clearance of oral and vaginal mucosa, with the exception of one erosion on the tip of her tongue. Some months later the lesion on the patient’s tongue had continued to progress despite the esophageal lesions continuing to improve (Figure 2). The patient was urgently referred to otolaryngology due to the suspicion of oral SCC. This suspicion was confirmed with a biopsy of the lesion which revealed invasive oral SCC. This patient’s SCC had invaded skeletal muscle and involved four out of thirty-one regional lymph nodes, two of which were positive for extranodal invasion. Upadacitinib was discontinued at this point and a glossectomy with bilateral lymph node dissection was performed. Staging revealed T3N3b SCC and sadly, this patient passed away.
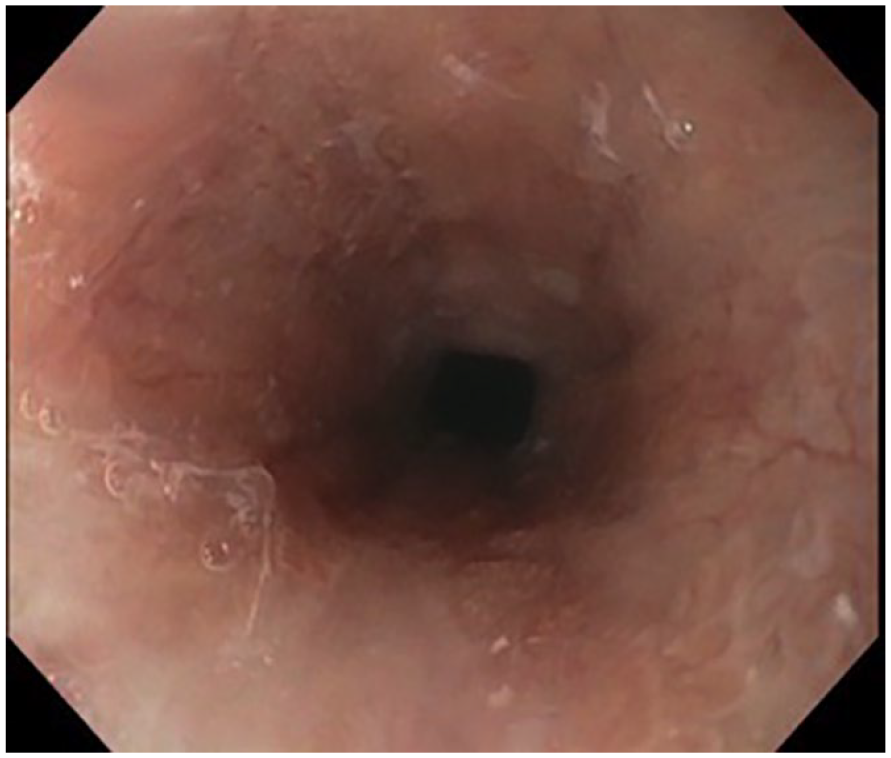
Upper endoscopy was performed again, several months after the initiation of treatment with Upadacitinib. Fewer esophageal erosions and ulcerations were seen at this time.
Discussion
Traditionally, the treatment of ELP has included systemic corticosteroids, immunosuppressants and/or oral retinoids 2 ; JAKi such as Upadacitinib, present a new possible line of treatment in ELP. Janus kinases, JAKs, are tyrosine kinases located on the intracellular portion of type I and II cytokine receptors. These consist of JAK1, JAK2, JAK3, and TYK2; each of which presents a potential drug target in inflammatory mediated diseases. Upadacitinib is a JAKi engineered to have a high affinity for JAK1. 3 This drug has been approved by Health Canada for the treatment of rheumatoid arthritis, 4 moderate to severe atopic dermatitis, 3 psoriatic arthritis, ankylosing spondylitis, and ulcerative colitis.
The successful treatment of LP with JAKi, such as Upadacitinib, has led to a greater understanding of the pathogenesis of LP. Within LP lesions there is an elevated expression of IL-21 and IFN-γ cytokines 5 both of which contain JAK1 within their receptors in addition to JAK3 and JAK2 respectively. Upon activation of these cytokine receptors JAKs become activated, leading to the subsequent activation of STAT1 and STAT3 transduction factors. 5 The translocation of STATs to the nucleus can have varying actions including the differentiation and proliferation of immune mediators. Within LP lesions there is increased activation of the JAK-STAT pathway, with an associated elevation in the levels of activated JAK1 and JAK3. 5 JAKi’s, such as Upadacitinib, prevent the transduction of this signal cascade through JAK1, thereby blocking the expression of any downstream inflammatory mediators.
Despite the success of JAKi in the treatment of inflammatory mediated disorders, there is a suggested association of the JAKi, Tofacitinib, with a risk of lymphomas and solid organs malignancies. Instances of these adverse events were documented in Phase 3 studies with a higher frequency of malignancies seen in patients receiving higher doses of Tofacitinib. 6 Tofacitinib preferentially inhibits cytokine receptors containing JAK1 or JAK3 while also inhibiting JAK2 and TYK2 to a lesser extent. 7 Despite differences in their selectivity and mechanism of action, all subsequent JAKi’s, including Upadacitinib, have been labeled with a similar risk of malignancy to Tofacitinib. 8
Based on a review of the literature, there is evidence to support the malignant potential of oral LP. 9 One particular review found that those with erosive or atrophic oral LP are more likely to undergo malignant transformation than any other subtype of LP. 10 While another found that those with LP lesions on the tongue had 1.82 times the risk of developing oral SCC. 11 Unfortunately, it remains a challenge to determine the individual risk of malignancy in those with oral LP as there is currently no prognostic biomarker available to aid in identifying which oral LP lesions are at the highest risk for malignant transformation. 11 In this particular case, the identification of oral SCC was only possible after the clearance of the LP lesions by Upadacitinib. This presents another challenge in monitoring for malignancy if SCC is being camouflaged by extensive disease.
In conclusion, ELP is a disabling condition that is often resistant to treatment. There are currently two published cases of oral/esophageal ELP that have been successfully treated with Upadacitinib as well as six published cases of LP that have been successfully treated with Tofacitinib.1,12 We therefore report the third documented case of oral LP that has been successfully treated with Upadacitinib and the only case to be complicated by malignant transformation of oral ELP to oral SCC. This particular case demonstrates that JAKi’s, particularly Upadacitinib may offer a potential new therapeutic option in those with recalcitrant ELP. Additionally, we highlight the importance of monitoring for mucosal SCC in areas affected by ELP, especially since these lesions can mimic the erosions of ELP. Finally, the influence of immunosuppressants, such as JAKi’s, on the rates of malignant transformation in ELP is not well understood and has not been previously researched. Further research in this area may contribute to our understanding of the pathogenesis of malignant transformation in ELP and how to identify patients most at risk.
Footnotes
Acknowledgements
No acknowledgment to make.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Declaration of Conflicting Interests for Dr. Ian Landells
No other conflicts of interest to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent
Informed consent was received from the patient.
