Abstract
Mature cystic teratoma of the ovary is the most common ovarian neoplasm (10%–20%). These tumors are generally benign but may undergo malignant transformation, 80% of which is squamous cell carcinoma, in postmenopausal women (1%–2%). Due to nonspecific findings, the preoperative diagnosis is challenging; most patients are detected at an advanced stage and have poor outcomes. A 40-year-old female, P2A0, complained of worsening lower abdominal pain and palpable abdominal mass. She had not been having menstrual periods for almost 4 months. For the last 3 months, she had asthenia, anorexia, and lost 10 kg. Abdominal examination was notable for an immobile, well-defined cystic mass measuring 12 × 8 × 8 cm with an irregular surface in the lower abdomen. Its overlying skin appeared reddish. A bimanual examination confirmed the adnexal origin of the cystic mass. Laboratory examination was notable for leukocytosis, thrombocytosis, increased neutrophil-to-lymphocyte ratio, and reduced absolute lymphocyte count. There was an increase in her cancer antigen 125. An abdominal ultrasonography examination showed bilateral mature cystic teratoma. A bilateral salpingo-oophorectomy was performed. A postoperative diagnosis of bilateral dermoid ovarian cysts was then established. The patient histopathology examination concluded squamous cell carcinoma (malignant transformation from benign cystic teratoma) in bilateral ovaries. The patient was referred to a gynecology oncology center for further examination and management. However, she missed the appointment for an unknown reason. The patient passed away 4 months after surgery. Clinicians should be aware of the possibility of malignant transformation of teratoma if a tumor has been present for a long time, its diameter is greater than 10 cm, there is a thickening of the cyst wall with papillary growth, there is an increase in tumor markers, and the patient is postmenopause. Malignant transformation of mature cystic teratoma remains a diagnostic and therapeutic puzzle and the prognosis is poor despite extensive initial surgery and optimal debulking.
Introduction
Mature cystic teratoma of the ovary (MCTO), commonly called a dermoid cyst, is the most common ovarian neoplasm, accounting for 10%–20% of all ovarian tumors and 60% of all benign ovarian tumors.1,2 It consists of well-differentiated derivates of germ cell layers.1,3 Over 80% of cases are present in reproductive periods, especially those under 40 years of age. 4 The most common signs and symptoms include abdominal pain and distention, constipation, urinary frequency, and a palpable abdominal or pelvic mass, but these are usually asymptomatic. 2 These tumors are generally benign but may undergo a malignant transformation (MT) in 1%–2% of the cases, especially in postmenopausal women.1,4,5 Squamous cell carcinoma (SCC), which arises from the ectoderm, accounts for 80% of MT, followed by adenocarcinoma, carcinoid tumor, melanoma, and sarcoma.4,6 The preoperative diagnosis is difficult because of nonspecific findings; therefore, most patients are diagnosed in advanced stages and have poor outcomes. Hence, diagnosing MT of MCTO at an early stage is essential for longer survival. 4 In this study, we describe a case of MT-MCTO, which was not clinically suspected preoperative and was diagnosed by postoperative histopathologic results. The patient details had been fully de-identified.
Case presentation
A 40-year-old female, P2A0, complained of worsening lower abdominal pain and palpable abdominal mass. She had not been having menstrual periods for almost 4 months. For the last 3 months, she had asthenia, anorexia, and lost 10 kg. The patient had no comorbidities.
The vital signs were within normal limits. Abdominal examination was notable for an immobile, well-defined cystic mass measuring 12 × 8 × 8 cm with an irregular surface in the lower abdomen. Its overlying skin appeared reddish. A bimanual examination confirmed the adnexal origin of the cystic mass.
The patient’s laboratory examination was notable for leukocytosis (13,000/µL), thrombocytosis (470,000/µL), increased neutrophil to lymphocyte ratio (NLR) (6.42), and reduced absolute lymphocyte count (1560). Her cancer antigen 125 (CA125) was raised at 83.3 U/mL. An abdominal ultrasonography examination shows two unilocular cystic masses with complex echogenicity sizes 11 × 7.4 and 8 × 6 cm. Dermoid mesh and rokitansky nodule were positive. Both masses did not exhibit increased intralesional blood flow. The result showed bilateral mature cystic teratoma (Figure 1).
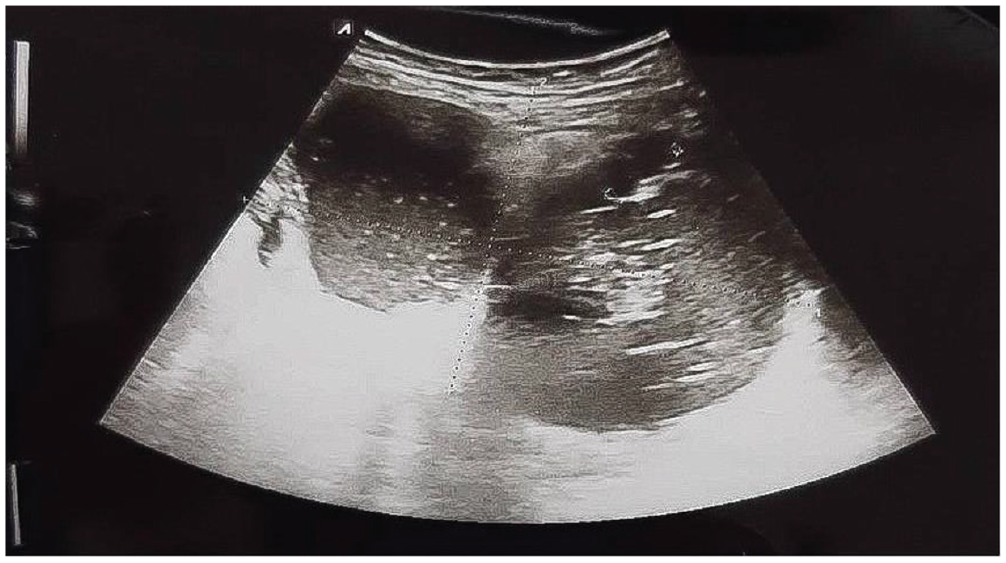
Abdominal ultrasound showed bilateral mature cystic teratoma.
A bilateral salpingo-oophorectomy was performed. The uterus was normal in size. Two ovarian cysts with irregular surfaces were found. The left ovarian cyst measured 12 × 8 × 6 cm with adhesions to the omentum, colon, and posterior corpus of the uterine. The right ovarian cyst measured 10 × 8 × 4 cm with adhesions to the posterior corpus of the uterine (Figure 2(a) and (b)). The left ovarian cyst ruptured spontaneously during adhesiolysis, leaking out a mixture of mucinous and serous fluid, hair, and bone (Figure 2(c) and (d)). The right ovarian cyst was completely resected. A postoperative diagnosis of bilateral dermoid ovarian cysts was established.
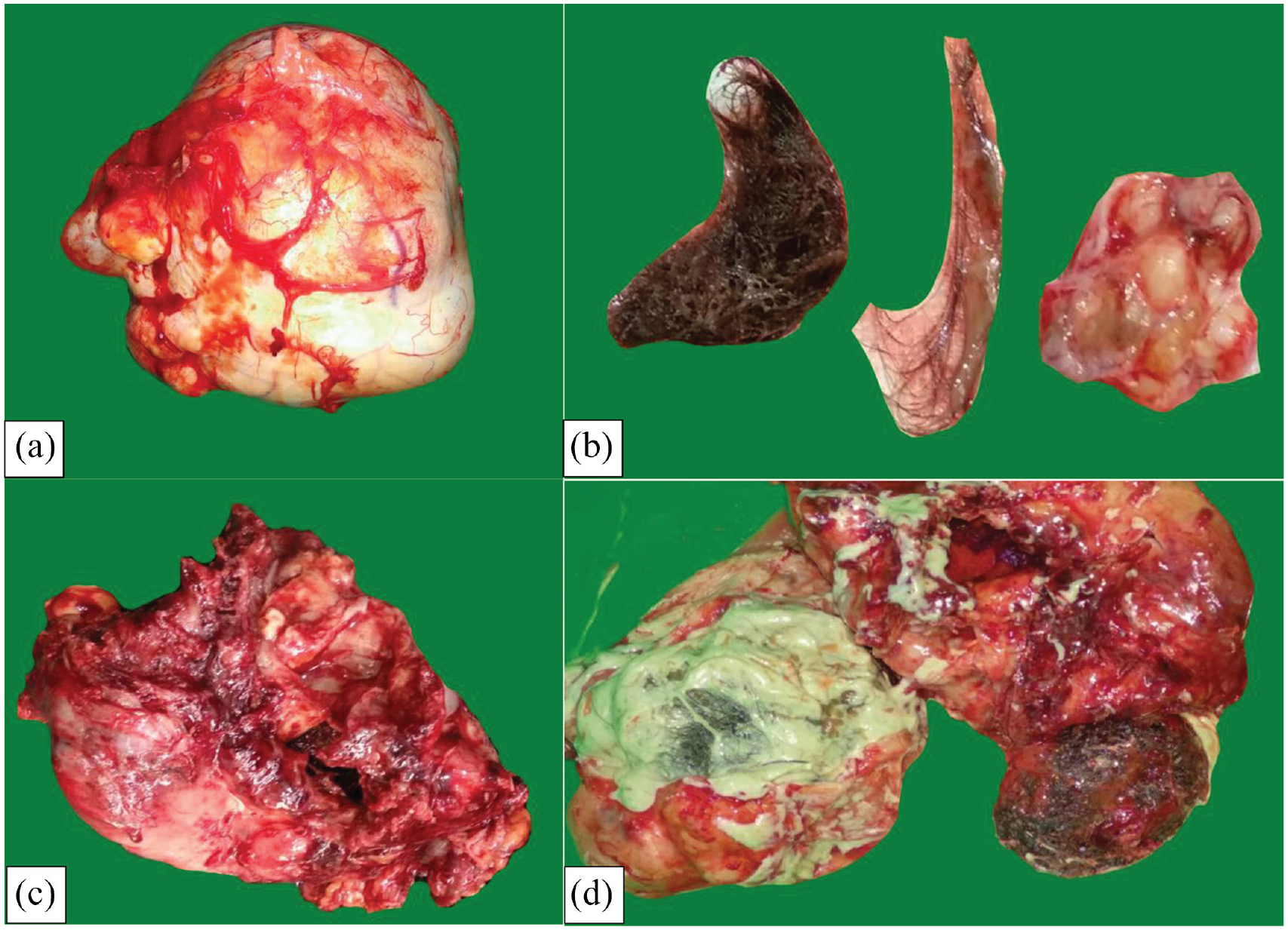
(a) Right ovarian cyst. (b) Hairs and bone inside the right ovarian cyst. (c) Ruptured left ovarian cyst. (d) Mucinous and serous fluid, with hairs and bone inside the ruptured left ovarian cyst.
On gross examination, the pathologist found the white-brownish left and right ovarian cysts with a size of 12 × 9 × 7 cm and 10 × 8 × 5 cm, respectively (Figure 3(a) and (c)). On the cut section, a unilocular cyst containing porridge-like fluid, typical soft yellowish butter-like material, hair, and a solid mass of bone was found (Figure 3(b) and (d)). Some of the inner layer of the wall was brownish and smooth. The wall thickness of the left and right ovaries was 0.5–1.5 cm and 0.1–0.2 cm, respectively. However, some parts of the cystic wall were rough and irregularly thickened, up to 2 cm in thickness. The inner surface of the cyst showed a focal tan area along the thickened part of the wall, showing the MT of the cyst.

Macroscopic specimen: white-brownish left and right ovaries containing porridge-like fluid, soft yellowish butter-like semisolid material, hairs, and bones. (a & b) Left ovarian cyst. (c & d) Right ovarian cyst.
Bilateral ovaries showed a cystic structure lined by a stratified squamous epithelium. There are solid areas that showed disorderly arranged invasive nests of atypical cells showing hyperchromatic, pleomorphic, and vesicular nuclei, with prominent nucleoli, mitotic figures and moderate to abundant eosinophilic cytoplasm. Keratin pearls were seen frequently.
The right and left fallopian tubes were normal. There was no malignant tumor invasion to the right and left fallopian tubes. The conclusion was SCC, an MT from benign cystic teratoma in bilateral ovaries (Figure 4(a)–(e)).
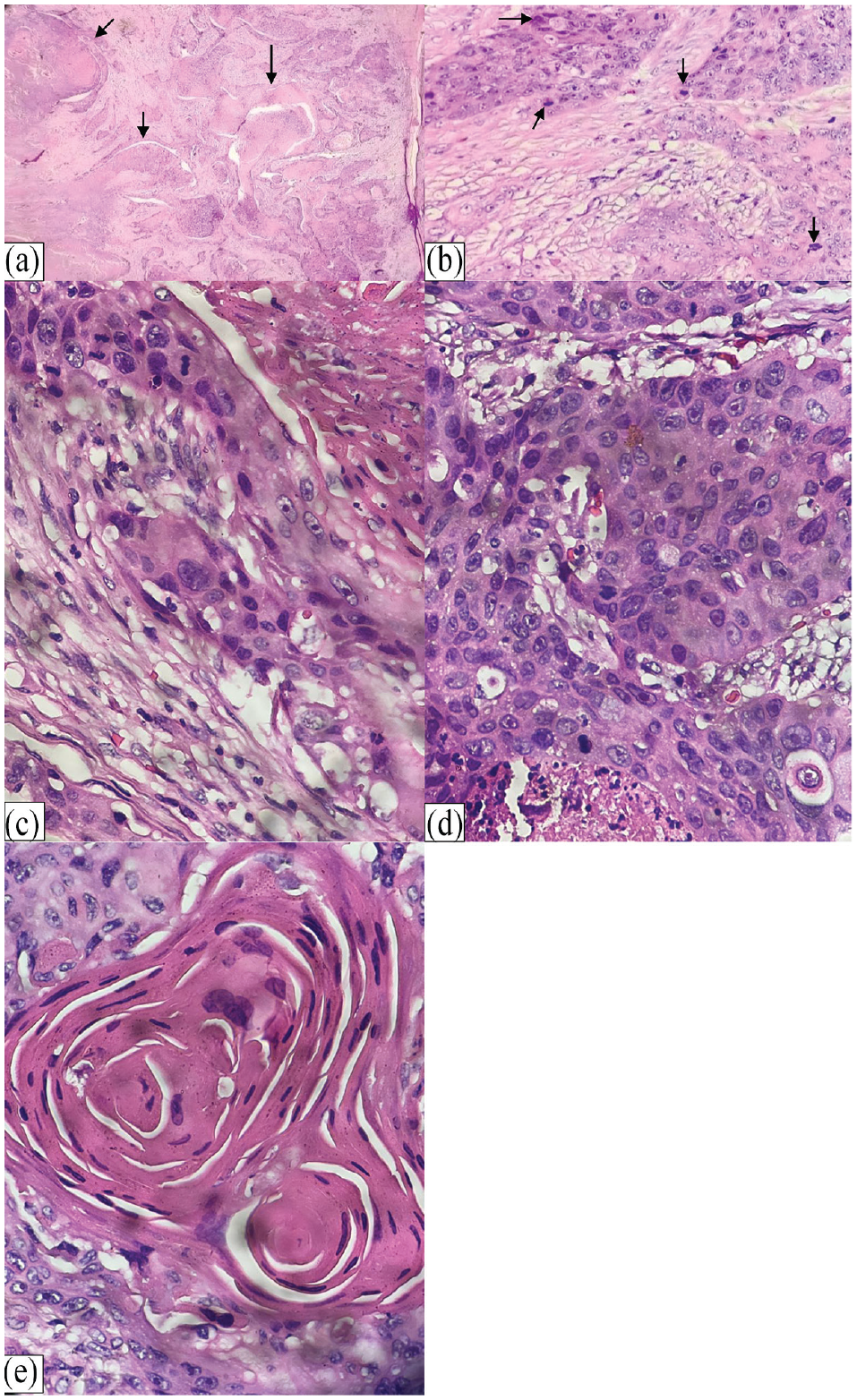
Pathological findings: (a) tumor nests, (b–c) hyperchromatic, pleomorphic, and vesicular nuclei, with prominent nucleoli and mitotic figures of the right ovary. (d) Hyperchromatic, pleomorphic, and vesicular nuclei, with prominent nucleoli and mitotic figures of the left ovary. (e) Keratin pearls.
The patient was referred to a gynecology oncology center for further examination and management. However, she missed the appointment for an unknown reason. The patient passed away 4 months after surgery.
Discussion
Mature cystic teratoma, also known as a dermoid cyst, is a benign tumor that arises from germ cells in the ovary and primarily affects young women of reproductive age. 7 The reported incidence of mature cystic teratoma is 1.2–14.2 cases per 100.000 per year. 8 A complication such as MT of the various tissue components of MCTO can occur. 8 It occurs when the cells within the dermoid cyst begin to divide and proliferate rapidly, leading to the development of cancerous cells. 9 Although rare, it occurs in approximately 1%–3% of all MCTO cases. However, one report shows that the frequency was as high as 6.67%.10–12 Lower in the previous study in which MT occurs is 0.17%–2%. 8 More than 80% of MTs are squamous cell carcinomas arising from the ectoderm; the rest are carcinoid tumors or adenocarcinomas. 13 The risk of MT increases with age and is more common in postmenopausal women. A family history of ovarian cancer and smoking are other risk factors. 8
Due to the lack of distinct clinical indications, tumor markers, or imaging findings, diagnosing MT both preoperatively and during surgery is extremely difficult, if not impossible.14,15 The tumor is asymptomatic in the early stage and the diagnosis of malignancy is made by histopathologic examination. 6 Several risk factors are believed to lead to the diagnosis of MT in MCTO cases, including age, menopausal status, imaging features, tumor size, serum levels of several tumor markers, stage of disease, and tumor remnants. 4 Unfortunately, the risk factors in this case were nonspecific.
In the most recent meta-analytical study, the median age of patients diagnosed was 53.5 years old, and 72.1% were ⩾ 45 years old. 16 Research by Hackethal et al. also shows that the mean age of patients diagnosed was around 55 years, and the group most at risk of developing malignancy was postmenopausal women. 8 Another two cases reported were postmenopausal women, 51 and 53 years old. 4 In contrast to our case, the patient was younger, aged 40 years, and had not yet experienced menopause.
SCC transformation in MCTO had two key clinical manifestations: palpable abdominal mass and abdominal pain. 16 Our patient also presented with palpable abdominal mass and abdominal pain. Bloating, loss of appetite, and weight loss are other symptoms reported previously.6,8
MT-MCTO tumors are typically located in one ovary but can involve both ovaries in 10%–17% of the cases. The tumor mass size is usually large (10–20 cm in diameter), 8 and larger than benign tumors, possibly due to the presence of additional areas of necrosis or hemorrhage. Tumor size is a potential indicator of MT of Mature Cystic Teratoma (MCT).2,5 According to some studies, tumors larger than 99 mm or tumors demonstrating rapid growth may be associated with an increased risk of MT, 17 and tumors larger than 150 mm are associated with more aggressive behavior. 18 In a meta-analytical study, the mean tumor size was 14.8 (range 3.5–40.0) cm. There is no difference in the survival rate between patients with the diameter of the tumor ⩽10 cm and >10 cm. 16 In our case, the left and the right ovarian mass were 12 × 8 × 6 cm and 10 × 8 × 4 cm, respectively, which are smaller than the mean size in the above studies.
Serum tumor marker levels are another risk factor for MCT MT that can be observed. Certain tumor markers (SCC antigen, CA125, cancer antigen 19-9, and carcinoembryogenic antigen) are frequently high in patients with SCC arising from MCTO. 8 A study also shows that elevated serum calcium levels might be a marker of SCC presence. 9 However, there is no association between the quantity of these tumor markers and the size of the tumor or the stage of the illness. 18 But preoperative SCC and CA125 antigen concentrations were associated with adverse outcomes. It is also suggested that a high CA125 is a more reliable prognostic marker than SCC. 8 However, a low level of SCC antigen does not rule out carcinoma.6,15 In this case, CA 125 was the only tumor marker being evaluated, and it was increased. Unfortunately, in our case, due to the technical impossibility of routine clinical practice, SCC antigen was not measured.
An early diagnosis of MCTO mainly relies on ultrasonic examination. The presence of lipid, hair, and cartilage causes MCTOs to exhibit prominent ultrasonic imaging characteristics, such as the dermoid plug, dermoid mesh, tip of the iceberg sign, and fat-fluid level. The similarities of ultrasound features between benign and malignant teratomas do not allow accurate differentiation. 4 Certain imaging features of the tumor may contribute to the diagnosis of MT, using transvaginal Doppler ultrasound to measure the blood flow resistance in the intratumoral vessel. This method perhaps is more accurate in differentiating benign from malignant MCT. Interestingly, Emoto et al. report that Doppler detection was a more helpful indicator than serum SCC antigen levels. 19 In our case, abdominal ultrasonography examination shows two unilocular cystic masses with complex echogenicity sizes 11 × 7.4 and 8 × 6 cm. Dermoid mesh and rokitansky nodule were positive. Both masses did not exhibit increased intralesional blood flow. The result shows bilateral mature cystic teratoma. Compared to ultrasound, magnetic resonance imaging (MRI) and computed tomography (CT) scans are more effective and specific in locating tumors and making a qualitative diagnosis. 14 The diagnostic value of CT and MRI examinations are essential when identifying malignant manifestations, such as necrosis, nodules, cyst wall growth, invasion of surrounding structures, or pelvic and abdominal metastases.4,20 Unfortunately, MRI or CT scan was not performed in this case.
Radical surgery (bilateral salphingo-oophorectomy, total hysterectomy) and comprehensive surgical staging (peritoneal washing, omentectomy, appendectomy, peritoneal biopsies, and pelvic plus paraaortic lymphadenectomy) should be the standard treatment, while the best cytoreductive surgery should be used for advanced disease.2,6 Cystectomy or unilateral oophorectomy and comprehensive surgical staging are justifiable if the patient wants to preserve fertility, especially in stage IA disease. Patients with stage IA tumors can only undergo the observation stage following a comprehensive surgical staging. 2 Due to retrospective design and limited data available in earlier research, there are still disagreements over the efficacy of a particular treatment, such as adjuvant chemotherapy or radiotherapy in this disease. A recent systematic study suggests that early hysterectomy and platinum-based chemotherapy may be linked to improved survival. Nevertheless, this finding needs to be interpreted with caution. 16 In our case, a bilateral salpingo-oophorectomy was performed. The uterus was normal in size. Two ovarian cysts with irregular surfaces were found.
A postoperative diagnosis of bilateral dermoid ovarian cysts was established. On histopathologic examination, the conclusion was SCC (MT from benign cystic teratoma) in bilateral ovaries. There was no malignant tumor invasion to the right and left fallopian tubes. However, the frozen section could not be performed in our case due to a lack of facility. Hence, surgical staging could not be carried out. Age >45 years, advanced staging, large tumor, high cancer grade, absence of hysterectomy, residual tumor lesions, absence of adjuvant chemotherapy, and non-platinum-based chemotherapy are poor prognostic factors. 6 The prognosis, in this case, could not be determined due to a lack of surgical staging. However, the fact that the patient died 4 months after the surgery for an unknown reason posed difficulties explaining the cause. This case is one of the supporting evidence that the management of MT of MCTO is not enough just with surgery. The risk factors that our patient had and the lack of adjuvant therapy significantly affected the patient’s prognosis. Early suspicion of MT should be raised in MCTO patients who have risk factors for MT.
Conclusion
Malignancy in MCTO is a rare entity. However, clinicians should be aware of the malignant possibility of teratoma if the tumor has been present for a long time; the patient is an older woman, particularly if she is in the postmenopausal age group; the tumor diameter is greater than 10 cm; or there is a thickening of the cyst wall of papillary growth as well as an increase in tumor markers. In addition, diagnosis and treatment of SCC arising from MCTO is still a diagnostic and therapeutic puzzle, especially in women with advanced disease, since, despite extensive initial surgery and optimal debulking, the prognosis in these patients is poor.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The institutional review board has exempted our case report from a formal ethical review.
Informed consent
Written informed consent was obtained from the legally authorized patient’s representative for their anonymized information to be published in this article.
Consent for publication
Written informed consent was obtained from a legally authorized representative regarding the publication of anonymized patient details in this case report.
