Abstract
It is a rare condition in twin pregnancies for a fetus to coexist with a complete hydatidiform mole, present with complications, and result in a healthy neonate. Only a few cases have been reported upon review of the literature. Early diagnosis is essential because this type of pregnancy is associated with serious complications and management challenges. The complications associated with these pregnancies include antepartum hemorrhage, hyperthyroidism, preeclampsia, prematurity, fetal death, and gestational trophoblastic neoplasia. Here, we describe a case of dizygotic twin pregnancy in which a complete mole coexists with a normal fetus, complicated by hyperthyroidism, that resulted in the birth of a 1700-g female alive neonate who is euthyroid to a 25-year-old primigravida at a gestational age of 33 weeks by emergency cesarean section for an indication of a twin pregnancy molar coexisting with an alive fetus in labor. The mother had been on conservative management and treated as an inpatient for hyperthyroidism. In our country, there have been three case reports of partial moles with coexisting alive fetuses, but this is the first case report of a complete mole with a coexisting alive fetus.
Introduction
Multiple pregnancy, in which hydatidiform mole coexist, with a healthy fetus, was a rare condition in the past, first described by Gordon Luker MD in 1887, but it is now occurring more frequently, at a rate of 1 in 22,000–100,000 pregnancies, as a result of the liberal use of ovulation induction.1–3 The majority is a complete hydatidiform mole with a coexisting fetus. However, 0.005–0.01% of pregnancies are known to have a partial hydatidiform mole with a coexisting fetus. 4 Twin pregnancies with a hydatidiform mole and a normal fetus seldom progress to term due to spontaneous or induced abortions due to maternal complications. 5
The coexistence of an alive fetus and molar gestation can occur in four different types pregnancy: the first type is dizygotic twins with normal fetuses and complete hydatidiform moles (diploid with a normal placenta); the second type is dizygotic twins with normal fetuses and partial hydatidiform moles; the third type is single pregnancy with a triploid fetus and partial hydatidiform; and the fourth occurrence is twin gestation in which one fetus is normal diploid with the normal placenta and the other is triploid with the partial hydatidiform molar placenta.6,7
Compared to singleton or non-molar twin pregnancies, there is a higher risk of maternal complications in twin pregnancies with a complete hydatidiform mole.8,9 These include fetal complications such as spontaneous abortion, intrauterine growth restriction, and intrauterine fetal death, as well as obstetrical complications such as hyperemesis gravidarum, antepartum hemorrhage, early-onset preeclampsia or eclampsia, hyperthyroidism, preterm premature rupture of the membranes, and preterm labor.8–10
Survival of the normal fetus varies and depends on associated comorbidities from the molar component, such as hyperemesis gravidarum, preeclampsia, hemorrhage, and serum human chorionic gonadotropin (hCG; below 400,000 mIU/mL have favorable outcomes).10–13
For this kind of cases, it is recommended to have conservative management with close follow-up at a tertiary center involving gynecologic oncologists, maternal–fetal medicine specialists, and a pediatrician with a level four neonatal intensive care unit (ICU) after strongly counseling the mother about the risks. This may entail hospitalization in some cases, especially for patients with hemorrhage or other obstetric problems. Although expectant treatment is an option, many women may decide to end the pregnancy if it is diagnosed early. 12
Case
This is a 25-year-old primigravida mother whose gestational age from a reliable last normal menstrual period was 29 weeks and 6 days. She came for an antenatal care follow-up after she was referred from a private clinic with the diagnosis of second trimester pregnancy and coexisting molar pregnancy at 26 weeks of pregnancy, which was her first visit. She had no aneuploidy screening or genetic testing. She had symptoms of excessive vomiting and nausea in the first trimester and early in the second trimester; otherwise, she had no history of vaginal bleeding, headache, abnormal body movement, or raised blood pressure, and she had no self-history or family history of diagnosed chronic medical or surgical illness. On her second visit, she was evaluated and diagnosed with the same assessment as the private clinic at 27 weeks and 5 days, and she was appointed to have regular follow-ups both at our hospital and the private clinic every 2 weeks. On her current presentation, she was diagnosed with hyperthyroidism in addition to the above diagnosis, for which she was admitted to the hospital after she presented for her routine follow-up. Except for palpitation, she had no symptoms or danger signs of pregnancy in her current presentation.
Upon arrival, the physical examination revealed a blood pressure of 120/70, a pulse rate of 108, a respiratory rate of 18, and temperature = 36.5°C The thyroid gland size was normal, with no nodules, and on obstetric abdominal palpations, the fundal height was 32 weeks, with a longitudinally lie, cephalic presentation, and a fetal heartbeat of 144. And also, no uterine contraction was detected, and on genitourinary examination, there was no vaginal bleeding, and the cervix was closed, posterior, and firm.
On ultrasound evaluation, the findings were as follows: twin intrauterine pregnancy, one normal and the other molar. The normal twin was cephalic, with a positive fetal heartbeat and no gross anomaly, an average gestational age of 29 weeks and 4 days, and an estimated fetal weight of 1200 g. The placenta was fundal and posterior. The biophysical profile findings were adequate amniotic fluid volume with intact acute variables, and the Doppler study was normal. There was a 13 × 7 cm echo complex mass anteriorly with a diffusely cystic and snowstorm appearance (Figures 1 and 2). The twin peak (lambda sign) and a thick inter-twin membrane are also seen.

2D ultrasound showing normal placenta, fetal forearm, and molar mass.

2D ultrasound showing molar mass and the fetus.
Laboratory investigation results were as follows: on complete blood count (white blood cells = 6500 cells/μL, hematocrit = 35.8%, and platelet = 236,000/μL), thyroid function tests (thyroid-stimulating hormone (TSH) = 0.19 mIU/L, free T3 = 8.21 nmol/L, free T4 = 275 nmol/L), liver function test (serum glutamic-oxaloacetic transaminase = 24 IU/L, serum glutamic-pyruvic transaminase = 19 IU/L), renal function test (creatinine = 0.8 mg/dL), serum electrolytes are in a normal range, blood group and Rh is O positive, hepatitis B surface antigen = negative, venereal disease research laboratory = negative, hepatitis C antibody = negative, urinalysis = unremarkable, and serum β-hCG was 88,387.21 mIU/mL.
She was diagnosed with an early preterm twin pregnancy in which normal fetus coexists with molar + hyperthyroidism based on the clinical presentations, ultrasound findings, and laboratory results. Following a thorough explanation of the risks of developing complications related to a molar pregnancy, she was admitted to the obstetric ward with the aforementioned diagnosis for inpatient management.
She received two courses of dexamethasone 6 mg intramuscularly twice a day (BID) for 2 days for lung maturity, as well as propylthiouracil 100 mg per os (PO) BID, propranolol 10 mg PO BID, and was followed with antepartum fetal surveillance with maternal kick count, a biweekly biophysical profile, and a fetal Doppler study. The Doppler study of the normal fetus was done, and all the results were normal throughout the follow-up period. Serum β-hCG done after 2 weeks of admission, and it was 94,387 mIU/mL. A thyroid function test was also done every 2 weeks (once antepartum after the initial presentation and twice postpartum), and it was in the normal target range, which is TSH suppressed and free T4 in the upper range of normal during antepartum and in the normal target range postpartum after 7 days and 2 weeks. She had improved from hyperthyroidism clinically as well as by biochemical tests, and the antithyroid drug was continued at the same dose.
On the 22nd post-admission day, at 33 weeks of gestation, she started having labor. Then, she underwent a cesarean section (C-section) for an indication of twin pregnancy molar coexisting with an alive fetus in labor, resulting in the delivery of a 1700-g female neonate who was alive and had APGAR scores of 8 and 9 at the first and fifth minutes, respectively. The fetus’ cord and placenta were healthy. The other finding was an excessive vesicular mass, which is distinct from a normal placenta and membrane (Figure 3).

Normal placenta and evacuated molar mass.
The woman had a smooth postoperative period; except the neonate was admitted to the neonatal ICU with the diagnosis of preterm + low birthweight + respiratory distress syndrome, and after being treated for the above diagnosis and worked up for congenital hypothyroidism (thyroid function tests were in the normal range in the first week and 2 weeks after the discharge), both the mother and neonate were improved and discharged on the fifth postoperative day.
After the operation, the sample was sent for histopathology, and the results showed hydrophilic villi with a circumferential proliferation of trophoblasts (Figure 4). The index was a complete mole, whereas the second placenta showed a normal placenta. The chest X-ray done postpartum was normal, and following 3 months, post-molar surveillance demonstrated a normal pattern of serum β-hCG decrease, which was then negative until 6 months of monthly monitoring.
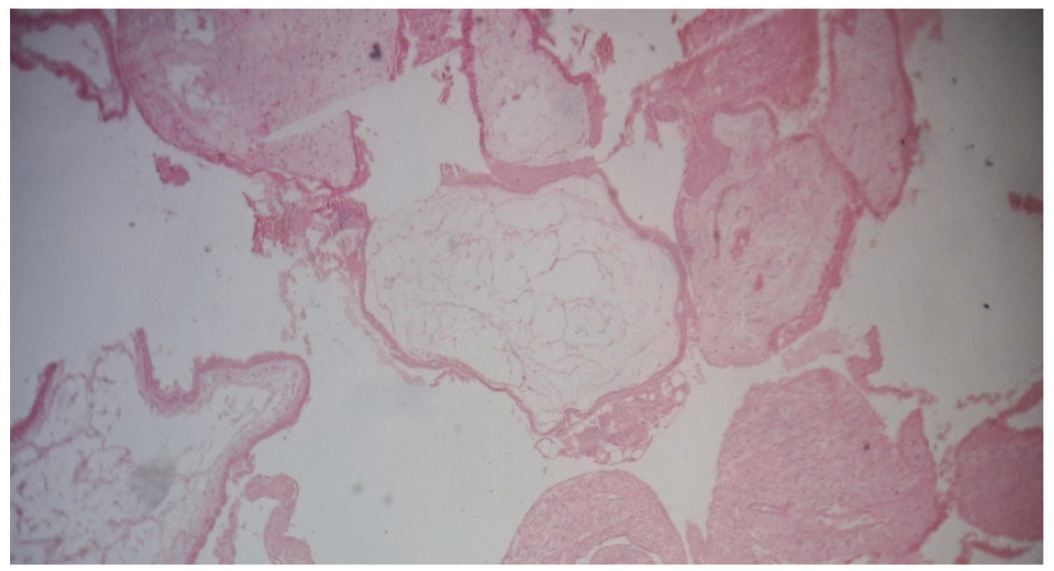
Histopathology of complete mole (hydrophilic villi with a circumferential proliferation of trophoblasts).
Discussion
It is a rare and a high-risk pregnancy when a hydatidiform mole coexists with a live fetus. The incidence of a complete hydatidiform mole and a partial hydatidiform mole coexisting with a live fetus is about 1 in 20,000 to 1 in 100,000 times and 1 in 10,000 to 1 in 20,000, respectively.14–16 Because some of the cases belong to periods when the division between partial and complete mole was not clear, the rates are quite varied. 10
About 200 cases of both complete and partial hydatidiform moles and coexisting live fetuses have been reported thus far, but no case of monozygotic twin partial molar pregnancy has been documented.14,17 However, there has been a reported increase in recent years of complete and partial moles coexisting with live fetuses due to the widespread use of ovulation induction.18,19 In a series of 30 occurrences of twin pregnancies with a coexisting fetus and a complete hydatidiform mole, 30% of them were preceded by ovulation induction. 19 A live fetus coexisting with a complete mole in a twin pregnancy is also known as sad fetus syndrome because the coexisting fetus is usually chromosomally normal and has the potential to survive.18,20
Recent developments in cytogenetics and ultrasonography allow for early antenatal identification and may make it possible to differentiate between complete and partial moles.14,15 The important point is to distinguish a complete mole and a coexisting live fetus with a survival chance for the fetus from partial mole pregnancies. Therefore, the placenta and other structures (i.e., another sac, the umbilical cord) must be examined attentively; in this way, a possible second sac negligence risk may also be decreased. 10
First trimester abdominal ultrasonography can be used to make the diagnosis of multiple gestations with molar pregnancy and a coexisting fetus. A study showed that 68% of patients with hydatidiform mole and coexisting fetus were diagnosed correctly by abdominal ultrasound. However, early ultrasound is less able to detect this rare existence than a complete or partial molar pregnancy.14,16
A complete mole has the typical snowstorm appearance, so the association with a normal gestational sac can be accurately determined around 12–14 weeks. 15 Even if the molar placenta partially covers the normal placenta, substantial overall swelling of the villous tissue with significant hemorrhagic areas can be seen on ultrasound. 17 A complete hydatidiform mole in a twin pregnancy will appear on an ultrasound scan as a normal fetus and placenta next to a molar mass.
On the other hand, distinguishing between focal partial molar degeneration and twins with complete mole can be difficult using ultrasound, as both show two distinct regions of the placenta, a practical approach is to follow the fetal umbilical cord. If it fuses with the molar placenta, twin pregnancies can be ruled out, but if the umbilical cord insertion is in the normal placental site, the two entities cannot be distinguished by ultrasound. 18 The diagnostic differentiation of placental mesenchymal dysplasia and the exclusion of myometrial invasion require the use of magnetic resonance imaging. 14 In our case, it was a complete molar with a coexisting, alive fetus and diagnosed at 26 weeks of pregnancy without difficulty.
While ultrasound examination aids in pre-evacuation diagnosis, histological evaluation of the products of conception provides the definitive diagnosis. 19 Maternal hCG levels that may be used together with ultrasound for diagnosis are not sufficient enough for exact diagnosis since these levels may increase in multiple pregnancies.10,20 But maternal serum α-fetoprotein measurements and β-hCG measurements help confirm the diagnosis and a cell-free DNA screening test can also suggest the presence of a complete molar pregnancy. Molar placental karyotype should also be included in the diagnosis.21,22
The prognosis and continuation of the pregnancy depend on the results of prenatal testing, which must include at least the fetal karyotype. 10 Although fetal karyotype was not available for our patient, as in most documented cases,10,22 we are convinced that our case was a complete hydatidiform mole with a coexisting live fetus due to the normal newborn, ultrasound demarcation between the normal and molar placenta, and histopathological examinations.6,10,20
Because pregnancies complicated by complete hydatidiform mole and a coexisting live fetus are very rare, there is a scarcity of literature to help guide management. Furthermore, reported outcomes vary significantly. 12 As a result, the decision to continue or end the pregnancy is more complicated in molar pregnancies with a normal, living fetus. 23
The first consideration in a complete hydatidiform mole with a coexisting viable fetus is to decide whether it is possible to continue the pregnancy by evaluating both maternal and fetal risks.9,23 According to different literature, the current consensus, is to follow the pregnancy conservatively if the woman and fetus are stable, after discussing the risks with the patient and family, with close clinical monitoring, and to consider an urgent delivery if complications arise.9,23,24 In about 60% of cases, live births occurred without increased maternal risk. 25 A retrospective study of 130 cases revealed that termination of pregnancy was recommended in 41% of them when faced with high maternal risk. 20 This implies that clinical management should be extremely cautious and give maternal risk factors great attention.
Another study found preterm birth is an important factor in determining outcomes, and live birth rates vary between 16% and 56%. 26 In our case, too, the therapeutic interruption of pregnancy was by preterm labor.
In the largest series of twin pregnancies in which a live fetus coexisting with complete hydatidiform mole (77 patients) to date, almost 40% of the women who decided to continue pregnancy had a live birth after 32 weeks. 27 However, it is important to consider the risks associated with premature birth, according to a study by Massardier et al., 26 since most complications seen in newborns are those commonly associated with preterm birth.
In our case, we have discussed the potential hazards of conservative management with the patient and her family. Despite having hyperthyroidism, she had a successful maternal and fetal outcome, which is quite unusual.
hCG and TSH share a molecular resemblance that leads to gestational trophoblastic hyperthyroidism, which is also linked to an increase in maternal and fetal problems. 28 In our case, she had gestational trophoblastic hyperthyroidism, for which she was treated with thioamide and beta-blocker, underwent inpatient follow-up and care, and improved and gave birth to alive euthyroid newborn.
Some authors reported an increased risk of severe preeclampsia (25.8%) in complete hydatidiform mole with a coexisting live fetus, while others found no significant differences with singleton complete molar.20,29 Steller et al. 29 suggest that the risk of persistent trophoblastic disease is higher in cases of complete hydatidiform mole and a coexisting live fetus compared with single molar pregnancies. However, a different study revealed that the risk of persistent trophoblastic disease after complete hydatidiform mole with coexisting a live fetus is not significantly higher than in single molar pregnancies (16% vs 19%). 30
According to a meta-analysis, C-section is more common than vaginal delivery among patients with a complete hydatidiform mole with a coexisting fetus: 72.2% versus 27.8% for vaginal delivery. 31 However, there is no substantial difference in the incidence of gestational trophoblastic neoplasia between the two modalities of delivery. 31
We recommend patients with similar circumstances in the late second and third trimesters to have a C-section to prevent a potential postpartum hemorrhage while still delivering a healthy fetus and minimizing other risks. In our case, she delivered by C-section without any complications.
Conclusion
Twin pregnancy of hydatidiform mole coexisting with a normal fetus is an extremely rare clinical condition with management dilemmas. As the diagnosis is rare, and since our case was complicated by hyperthyroidism and resulted in a healthy euthyroid baby after controlling hyperthyroidism, and it is the first case so far from our country, Ethiopia, we report this case, considering sharing similar cases and consequences could add more evidence to the current literature. In our case, expectant management in the hospital was made until the C-section was decided after the onset of labor. Both the mother and neonate were discharged healthy.
Footnotes
Acknowledgements
I would like to thank Dr Betelihem Admasu and Miss Selamawit Getachew for editing and commenting on this case report.
Author contributions
All authors are involved in diagnosing and treatment including operation while the corresponding author wrote the case report. All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, agreed to the journal submitted, and agreed to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Patient consent
Informed written consent has been obtained from the patient for all case details and images to be reported in the journal.
