Abstract
Cystic lymphangioma develops due to the interruption of lymphatic-venous connections during embryogenesis, leading to the formation of a cystic lesion containing lymph. These lesions fall under the ISSVA classification of vascular malformations. The first documented case dates back to 1828, with further elucidation provided by Sabin in 1909 and 1919. Cervicofacial region is the most common site, often showing early-stage symptoms. Inguinal location is rare, but if complications arise, it may present as a strangulated inguinal hernia. The tumor’s severity lies in its compression and invasion of the aerodigestive tract and adjacent organs. Diagnosis relies on imaging techniques such as ultrasound and computed tomography, which help determine the mass’s nature, boundaries, and relationship with neighboring structures. Asymptomatic lesions are generally monitored, while symptomatic ones require complete surgical excision to minimize the risk of recurrence. At Cheikh Khalifa University Hospital, we present a case highlighting our urology department’s experience in diagnosis, patient care, and surgical treatment.
Introduction
There are several differential diagnoses of inguinal cystic lymphangioma, including inguinal hernia, spermatic cord hydrocele, lymphadenopathy, necrotic lymph node, inguinal abscess, and inguinal hematoma.1,2 Each of these diagnoses has distinct clinical features that can help differentiate them from cystic lymphangioma. However, it is important to consider cystic lymphangioma in the differential diagnosis, as it is a rare but potentially serious condition that can cause compression and invasion of nearby organs.
To date, only six cases of inguinal cystic lymphangioma in adults have been reported in the literature.3,4 This limited number of cases highlights the need for further research and understanding of the mechanisms of cystic malformation. In this review, we will provide a comprehensive overview of the current literature on inguinal cystic lymphangioma and its mechanisms of formation. In addition, we will present new information that can help improve the identification and diagnosis of this rare condition in clinical practice.
Reported case
This is a 64-year-old retired man from Casablanca, living with his spouse and three children. He is the youngest of two siblings. His medical history includes chronic hypertension, which is managed with a calcium channel blocker. He also has hepatitis C, which has resulted in oesophageal varices and was confirmed by a EGD (Esophagogastroduodenoscopy). He has had ligatures for hemorrhoids in 2012. The patient has no family history of medical or surgical issues or toxic habits. He was admitted to the urology department for management of a right inguinal swelling that had been present for 5 months, as shown in Figure 1. The appearance of this right inguinal swelling of a soft consistency, approximately 1 cm in size, initially painless, coincided with the diagnosis of hepatitis C in January 2022. The swelling gradually increased in size, became painful, and had a firm consistency over time, all in the absence of fever and with no impact on the patient’s general health. The patient’s general somatic examination was normal, with a temperature of 37°C, blood pressure (BP) 130/70 mm Hg, pulse 55 bpm, body mass index (BMI) 20.8 kg/m2, and SaO2 of 98%. The clinical examination revealed a conscious patient, hemodynamically and respiratory stable, non-febrile, with a right inguinal swelling of about 3 × 4 cm (Figure 1). The swelling was of firm consistency, non-adherent to the deep plane, non-reducible, slightly painful on mobilization of its base, and without adjacent inflammatory signs. The rest of the clinical examination was unremarkable for all other systems.

Lateral view of the right inguinal swelling measuring 3 × 4 cm.
The medical report requested indicates the following on a biological level: hemoglobin level of 13.6 g/dl, white blood cell count of 4930 elements/mm³, C-reactive protein level at 5 mg/l, and correct ionogram results. The inguinal Doppler ultrasound revealed the presence of a cystic formation in the right inguinal fold, as shown in Figure 2, suggesting a cystic lymphangioma measuring 42 × 28 mm. The cystic lymphangioma appeared non-vascularized on Doppler and was found to be pushing back the ipsilateral spermatic cord medially while following the inguinal canal.

An image of probable inguinal cystic lymphangioma. This formation measuring 42 × 41 × 28 mm.
Due to the recent increase in pain, it was decided to surgically excise the mass and conduct an anatomopathological study for diagnostic confirmation. During exploration via the inguinal route, a lymphangioma was observed (as seen in Figure 3)—a transilluminable cystic mass with a thin wall and liquid content, smooth surface, and slightly vascularized wall—in close contact with the spermatic cord. Macroscopic examination during the anatomopathological study revealed a closed cystic formation measuring 5 × 4 × 2.5 cm with a thin wall. Upon opening the fragment, a pinkish-yellow liquid was noted.
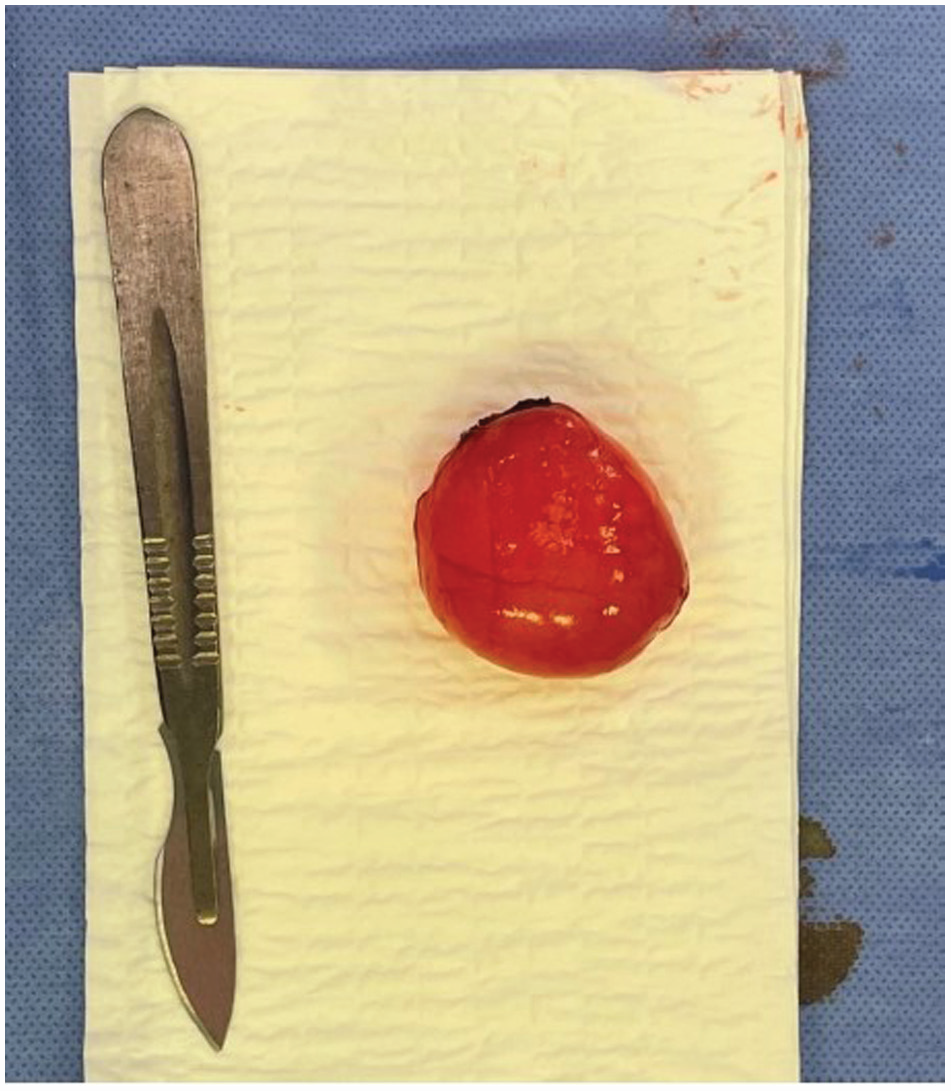
Cystic formation of a 5 × 4 × 2.5 cm.
Hematoxylin- and eosin-stained sections in the microscopic study demonstrated multiple large irregular vascular spaces. These spaces were lined by flattened, bland endothelial cells (as shown in Figure 4). The stroma supporting the cystic spaces was fibroblastic, with lymphocyte aggregates and small, variable-sized congestive vessels (as depicted in Figure 4). There was also mature adipose tissue with thin muscle bundles. No signs of malignancy were found. The postoperative follow-up was uneventful in this patient with normal BP and a clean bandage. Therefore, he was discharged on the first day postoperatively, with a change in bandage every 2 days and thread removal on the 15th day. The patient’s status will only be monitored clinically because the risk of recurrence of the inguinal lymphangioma in adults is almost zero. Therefore, it was decided to review him again at 1 month and then at 3 months.

Microscopic exam of the cystic formation. (a) Large irregular vascular spaces with fibroblastic stroma (H&E Gx50). (b) Vascular spaces lined by flattened bland epithelial cells. Note aggregate of lymphocytes (H&E Gx50).
Clinical discussion
Cystic lymphangiomas are inactive malformations that consist of cysts and atypical lymphatic vessels. They can occur on mucous membranes and the skin. 5 According to the commonly accepted classification of vascular malformations, the 2018 classification of the ISSVA (international society for the study of vascular anomalies) 6 states that the term “lymphatic malformation” replaces that of “lymphangioma” (although these terms are equivalent) to designate congenital malformations consisting of lymphatic vessels. 7 These lymphatic malformations are present at birth and can be diagnosed antenatally for macrocystic forms.
Cystic lymphangiomas are typically diagnosed before the age of 2 years in more than 90% of cases. 8 They do not communicate with the normal lymphatic network, 9 except in a few reported cases in the retroperitoneum10,11 and contain clear lymph or chylous fluid. Lymphangiomas are histologically classified into three types, but the cystic type is of particular interest and is classified according to several parameters, including the lymphatic space of the tumor. 12 As lymphangiomas are congenital malformations, they are most often diagnosed at a very young age and very rarely in adults. 13 The neck and axilla are the most common locations, but the inguinal region is the rarest. 14
Almost all cystic lymphangiomas are asymptomatic, and most affected patients consult for cosmetic reasons or fear of developing invasive cancer. The differential diagnosis of inguinal cystic lymphangioma includes inguinal hernia, spermatic cord hydrocele, lymphadenopathy or necrotic lymph node, inguinal abscess, and inguinal hematoma.
Differential diagnosis between spermatic cord hydrocele and cystic lymphangioma can be challenging due to their similar clinical presentations. 15 Spermatic cord hydrocele is a fluid-filled sac that surrounds the testis and is typically painless, while a cystic lymphangioma is a malformation of lymphatic vessels that contains clear lymph or chylous fluid. Inguinal or scrotal ultrasound can help differentiate between the two, with spermatic cord hydrocele typically presenting as a simple fluid-filled sac with thin walls, while cystic lymphangiomas often have thicker walls and septations. However, in cases where diagnostic uncertainty remains, a histological examination following surgical excision may be necessary to establish a definitive diagnosis.
In our case, the clinical presentation was a right inguinal swelling of about 3 × 4 cm (Figure 1), without adjacent inflammatory signs, of firm consistency, non-adherent to the deep plane, non-reducible, and slightly painful upon mobilization of its base. For our case report, ultrasound, as well as color flow Doppler, was the main paraclinical mean for diagnosis.
The definitive diagnosis of cystic lymphangiomas is established through histological study after an adequate surgical excision. Microscopic examination reveals the presence of attenuated endothelium, which lines the lymphatic spaces and lymphatic dilatation.
Recurrence is the most feared complication when complete excision is not achieved.16,17 During a follow-up period of 3 months, no signs of recurrence have been reported. Surgical excision with preservation of adjacent structures is the key treatment for scrotal and inguinal lymphangiomas.
Conclusion
Surgical excision is the preferred method for treating inguinal cystic lymphangiomas, ensuring caution is exercised to protect both the intra-scrotal structures and the structures within the inguinal canal. It is crucial to achieve complete excision to prevent any future recurrence. In addition, conducting complementary imaging of the surrounding areas is essential to detect any potential extension or presence of coexisting lesions.
Footnotes
Acknowledgements
We thank our colleagues from Department of urology, Department of radiology, and Department of histopathology, Cheikh Khalifa International University Hospital, Casablanca, Morocco, for all their efforts.
Author’s contributions
YK: Responsible for the concept design, writing and editing of all the manuscript including the conclusion and clinical diagnosis; RR: Writing and editing abstract, and data curation (figures and videos) and clinical diagnosis; YE: Writing and editing case presentation; KO: Data curation and microscopic study and histologic diagnosis; NB: Ultrasound diagnosis.
Availability of data and material
Any further data and material can be received through contacting the corresponding author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Guarantor
Yassine Koubbach.
