Abstract
There have been a growing number of acute disseminated encephalomyelitis cases following coronavirus disease-19 (COVID-19) infection. Given the rare occurrence, studies eliciting the clinical features, treatment response, and outcomes are still limited. In patients recovering from COVID-19, multifocal neurologic symptoms in the presence or absence of encephalopathy must be closely evaluated by neurologists and physicians. Early radiographic evaluation using magnetic resonance imaging and timely administration of glucocorticoid-based treatment reduces mortality and leads to satisfactory outcomes.
Keywords
Introduction
Acute disseminated encephalomyelitis (ADEM) is an autoimmune demyelinating disease of the central nervous system (CNS) with new-onset polyfocal neurologic symptoms, including encephalopathy in conjunction with multifocal demyelination on neuroimaging. Despite being reported without any preceding evidence of infection in several cases, ADEM has been established as a sequela to infection since the 18th century. 1 Although it can occur at any age, it is common in children and rare in adults. 2
After the worldwide pandemic of coronavirus disease-19 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), neurological complications associated with it have been increasingly recognized, including ADEM. 3 Although rare, the relationship between SARS-CoV-2 and ADEM has been reported in recent literature, where ADEM associated with SARS-CoV-2 was found to share most of the features of classic ADEM with moderate discrepancies, including longer duration from infection to ADEM symptomatology, severity of antecedent infection, poorer outcomes, and lower recovery rates.4,5
The scarcity of post-COVID-19 ADEM mentions in literature, severity of the antecedent SARS-CoV-2 leading to ADEM, increased incidence of post-COVID-19 ADEM in adults and poorer neurological outcomes than those of pre-pandemic ADEM led us to report this case internationally.
Case presentation
A 51-year-old female with no prior comorbidities presented to Tribhuvan University Teaching Hospital, Kathmandu, (TUTH) emergency department with altered sensorium for 28 days. Prior to visiting our institution, she visited a local hospital with presenting complaints of drowsiness, decreased voluntary eye movement, inability to eat by herself, inability to follow commands, absence of spontaneous speech, and incontinence of bladder and bowel for 2 days. The symptoms did not improve during the 5-day stay at the local hospital for which she had to visit the district hospital where she tested positive for COVID-19 and was admitted to the intensive care unit (ICU). After 2 days of ICU stay, she had spontaneous eye opening back, but she became emotionally labile and spoke inappropriate words. Majority of the symptoms improved except the emotional lability and inappropriate words. After 15 days of ICU stay with no further improvement, the patient and her family decided to visit our center (TUTH).
In our emergency department, examination revealed that the patient was alert and cooperative. The patient did not complain of limb weakness, burning sensation, tingling sensation, and numbness. There was no blurring of vision, headache, nausea, vomiting, chest pain, shortness of breath, palpitation, pre-syncope, syncope, vertigo, neck stiffness, trauma, nasal discharge, or abnormal body movement. There was no history of a similar illness in the past, but she had an episode of fever and cough 4 days before the presenting complaints, which was treated as an upper-respiratory-tract infection in the local health care center. There was no history of seizure, trauma, alcohol intake, and thereby alcohol withdrawal. She had a history of burr hole drainage for traumatic intracranial bleed 25 years back.
Her vitals at the time of admission were stable except the high blood pressure (150/90 mm Hg). Respiratory examination showed bilateral normal vesicular breath sound without any added sound. Cardiovascular system examination was normal with audible S1 and S2 without any murmur. On neurological examination, Glasgow Coma Scale (GCS) 6 was 12 (E4V2M6). Her memory was intact, and speech comprehension was preserved, but the patient showed dysarthria. Her muscle tone increased, and reflexes were 3+ in all the limbs with normal bulk and power. Plantar responses were improving. She had normal sensations for touch, pain, temperature, vibration, and joint position. She did not have any sign of cerebellar abnormalities. There were no neck rigidity and no abnormal body posturing. The RAPD (Relative Affererent Pupillary Defect) test was negative. Her facial symmetry was intact.
Emergency management of the patient in our center included intravenous hydrocortisone 100 mg and fluids, followed by oral prednisolone 60 mg per day. She was fed orally as her swallowing was intact. She was then shifted to the neuromedicine ward for further evaluation. Preceding the COVID-19 infection, unexplained encephalopathy, multifocal neurologic signs and symptoms, absence of a similar past history, and no history of previous demyelinating disorders led us to the provisional diagnosis of ADEM.
To establish the provisional diagnosis and to rule out differential diagnoses, we carried out cerebrospinal fluid (CSF) examinations; looked for metabolic derangements; evaluated for endocrinopathy; assessed liver function tests, thyroid function tests, and renal function tests; and interrogated for possible intoxication. She was not recently vaccinated against COVID-19, which ruled out vaccine-associated ADEM. The other causes of encephalopathy including toxins, metabolic causes, and infection were ruled out. From history, intoxication was less likely, and toxicologic screening was not performed. Other investigations were limited as far as possible to address the economical constraints of the patient and with the further plan of performing magnetic resonance imaging (MRI). Based on clinical features alone, a single clinical episode could not accurately distinguish between multiple sclerosis (MS) and ADEM, which warranted MRI.
Results of some important laboratory tests are listed in Table 1. Erythrocyte sedimentation rate and serum C-reactive protein tests were negative. Renal function tests and liver function tests showed no derangements. A lumbar puncture was performed, and in CSF analysis, protein count was normal with normal cellularity and glucose levels. CSF immunoglobulin G (IgG) value and adenine deaminase value did not raise.
Laboratory parameters at the time of admission.
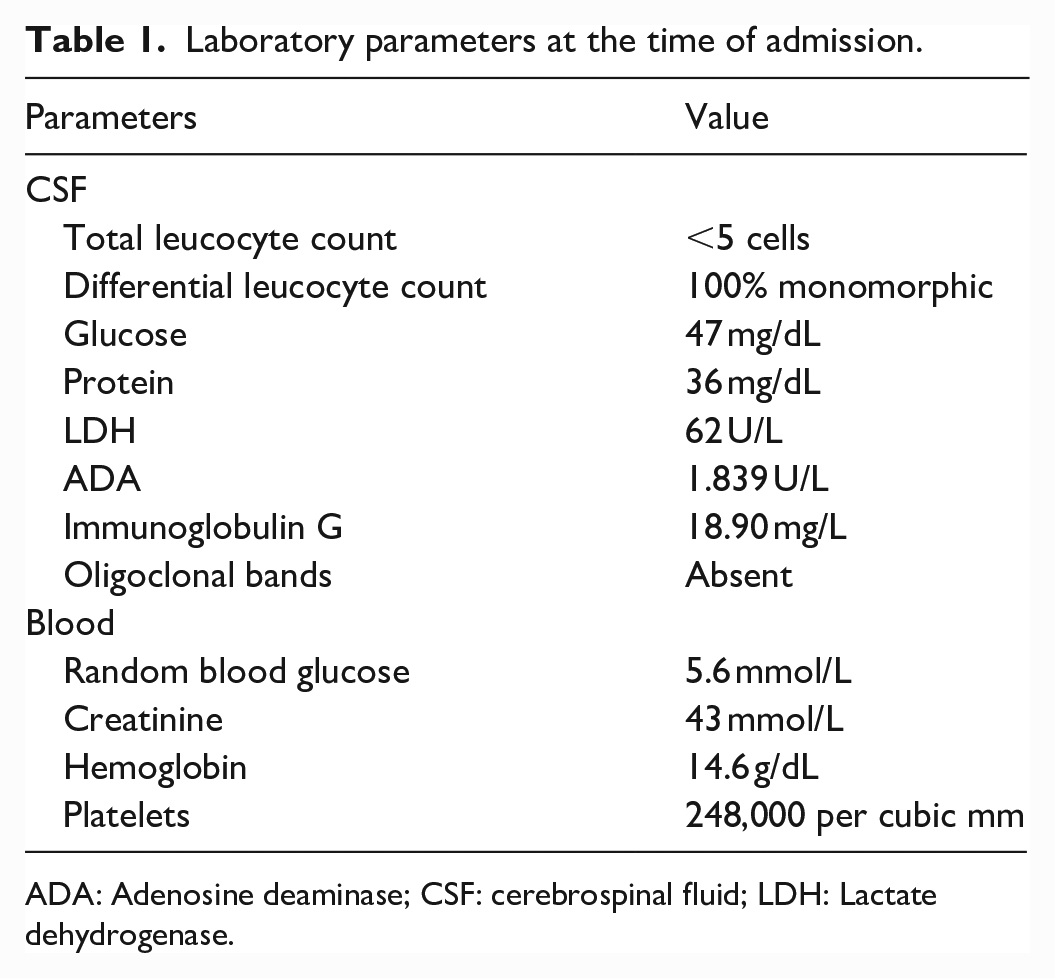
ADA: Adenosine deaminase; CSF: cerebrospinal fluid; LDH: Lactate dehydrogenase.
The encephalitis/meningitis panel was negative. India ink staining of CSF was negative for the Cryptococcus spp. Polymerase chain reaction (PCR) tests for neurotropic viruses (adenovirus, varicella-zoster virus, Epstein–Bar virus, herpes simplex virus, enterovirus, and cytomegalovirus) were negative. Oligoclonal bands were not found on CSF analysis, which concurrent with polyfocal symptoms (vs monosymptomatic in MS) ruled out MS. CNS infections were ruled out from the negative CSF panel. All these findings were consistent with the immune-mediated process and helped us rule out other causes of CNS involvement, limiting investigations to as little as possible.
Serology was non-reactive for antinuclear antibody, Treponema pallidum hemagglutination, and antibodies for toxoplasma. PCR of nasopharyngeal and oropharyngeal swabs was positive for SARS-CoV-2. Owing to limited resources, SARS-CoV-2 was not tested in CSF. NMO-IgG (Neuromyelitis Optica-IgG) and MOG-IgG (Myelin Oligodendrocyte Glycoprotein-IgG) autoantibody tests were negative. Malignancy screening was negative with no detection of serum tumor markers.
Suspecting ADEM, the patient was put on oral prednisolone 60 mg daily, and we proceeded to neuroimaging for diagnostic certainty. MRI of the brain and whole spine was performed. Supportive treatment included catheterization for urinary incontinence, speech therapy, calcium and vitamin D supplements, and antiemetics.
The initial MRI (Figure 1) showed multiple bilaterally asymmetrical, poorly marginated T2 high-signal-intensity foci, juxtacortical and subcortical foci in bilateral frontal lobes, left temporal and occipital lobes, bilateral basal ganglia (Figure 1(a)), right periventricular region (Figure 1(b)), bilateral cerebral peduncle of midbrain (Figure 1(c)), and spinal cord at the C6-C7 level.

Initial magnetic resonance images. (a) Multiple bilaterally asymmetrical, poorly marginated T2 high-signal-intensity foci, juxtacortical and subcortical foci in bilateral frontal, left temporal, and occipital lobes, as well as in bilateral basal ganglia. (b) Similar T2 high-signal-intensity foci in the right periventricular region. (c) T2 high-signal-intensity foci involving the bilateral cerebral peduncle of the midbrain. (d) Short-segment T2 high-signal-intensity foci of spinal cord at the C6-C7 level.
The patient’s symptoms resolved after 10 days of steroid therapy, and the patient was discharged while on oral prednisolone 60 mg per day. The patient had been on routine follow-up. Follow-up MRI after 3 months (Figure 2) of discharge showed high-signal-intensity lesions in the right periventricular region at the level of the body of lateral ventricle in FLAIR (Fluid-attenuated Inversion Recovery) axial scan (Figure 2(a)) and T2 axial scan (Figure 2(b)). Foci of high signal intensity in the right periventricular region and juxtacortical area of the left frontal lobe were seen in a T2 coronal scan (Figure 2(c)). Compared to the previous scan, the size and number of signal intensities were decreased (Figure 3).

Follow-up magnetic resonance imaging after 3 months. (a) FLAIR axial scan at the level of the body of lateral ventricle displaying high-signal-intensity lesions in the right periventricular region. (b) T2 axial scan at the same level. (c) Coronal T2 scan displaying foci of high signal intensity in the right periventricular region and juxtacortical area of the left frontal lobe.

Comparison of the initial MRI (a and b) to the follow-up MRI after 3 months (c and d). (a and b) T2-weighted axial images at the level of basal ganglia and body of the lateral ventricle. (c) T2-weighted axial image at the level of basal ganglia and body of the lateral ventricle. (d) FLAIR image at the level of basal ganglia and body of the lateral ventricle.
Discussion
Given the unavailability of specific biomarkers and heterogeneous clinical presentations, diagnosis of ADEM remains challenging, which puts emphasis on detailed history and neurologic examination revealing multifocal symptoms and encephalopathy in alignment with antecedent infection. 7 ADEM has also been considered a diagnosis of exclusion given that other CNS demyelinating and inflammatory syndromes (ADEM mimics) are ruled out, especially MOG antibody-associated disorder and MS. 8 In our patient, the initial clinical course with antecedent COVID-19 infection (altered sensorium and multifocal neurologic deficits and non-existence of other causes of encephalopathy) was consistent with ADEM, and we worked up for investigations down the line.
Apart from history, ADEM and MS distinction are based on MRI findings, CSF oligoclonal bands and time, and there are no distinct criteria to foretell whether an episode of demyelination will lead to MS or not. 9 In our patient, late age of onset, pertinent MRI findings, absence of CSF oligoclonal bands, no new lesions in follow-up MRI after 3 months, and resolving follow-up scans with no new symptoms on follow-up interval in the background of supportive evidence provided by COVID-19 infection make MS less likely to be the diagnosis.
Neurological manifestations after COVID-19 infection are being increasingly reported in the literature, including its rare association with ADEM. A lack of consensus on set diagnostic criteria of ADEM for the adult population has often led to generalization of IPMSSG (International Pediatric Multiple Sclerosis Study Group) criteria itself for diagnosis. Although similar to the pre-pandemic disease course on the grounds of clinical features and timing between infection and ADEM symptomatology, post-COVID-19 ADEM is associated with more frequency in adult population, a more severe preceding infection, rare MOG antibody seropositivity, and increased morbidity and mortality. 5 Whether or not SARS-CoV-2 infection is the preceding factor for ADEM development is confirmed by the identification of this agent through PCR tests in most cases in the absence of other responsible agents. However, ADEM has been reported in asymptomatic to mild to severe cases of COVID-19 infection, supporting the hypothesis of multifactorial etiology; not just the extent of previous viral exposure and magnitude of initial immune response. 10
Association with the preceding infection has been found in 50%–75% of adult cases and in 50%–70% of pediatric cases. 11 As asymptomatic and milder presentations of COVID-19 infection do commonly occur, there are chances of missing COVID-19 preceding CNS demyelination in such cases, which puts emphasis on serologic testing of COVID-19 in cases of ADEM.12,13
The exact pathogenesis of ADEM associated with COVID-19 is not yet elicited. What is plausible is that immune-mediated mechanisms or molecular mimicry leads to anomalous neuro-inflammatory loops, not requiring the virus to cross the blood-brain barrier to cause CNS damage. This has been deduced from the fact that most cases had no viral detection in CSF yet neuroinflammation (pleocytosis and hyperproteinemia) was evident.10,14,15
A recent review including 46 patients (including children and adults) reported encephalopathy as the most common neurologic complication (78%), followed by focal neurologic deficits (43%). 5 Zelada-Ríos et al. 10 reported encephalopathy followed by pyramidal signs, brainstem signs, cerebellar signs, seizures, and peripheral nerve compromise as the neurologic manifestations among 30 post-COVID-19 ADEM patients. In our patient, encephalopathy manifested as altered sensorium, drowsiness, decreased voluntary eye opening, inability to understand commands, bowel bladder incontinence, and emotional liability, which was the initial presentation. Our patient had an unremarkable computerized tomography scan. No enhancement on MRI scans and negative CSF panels argued against infectious encephalitis. Temporality (parallel presentation of COVID-19-specific symptoms and neurological manifestation) and less-frequent pleocytosis in cases of COVID-19-associated encephalitis, another neurological sequela of COVID-19, delineate it from ADEM.10,16
There have been various viewpoints of neuroimaging for ADEM. MRI is regarded as highly sensitive in demonstrating white matter lesions of both MS and ADEM. However, a single episode of demyelination on MRI cannot fairly distinguish ADEM and MS. 17 MRI features suggestive of ADEM typically include reversible, poorly defined lesions of white matter in the brain and spinal cord, concurrent with involvement of thalami and basal ganglia, which are hyperintense on T2-weighted and FLAIR sequences. Follow-up MRI is useful in diagnosing ADEM retrospectively. 8
A randomized study elucidating ADEM treatment is still lacking. 18 In a multicenter study, Zelada-Ríos et al. recommend a glucocorticoid-only regimen as effective as combined treatment for post-COVID-19 ADEM, which would be a boon to low- and middle-income countries. Hence, early glucocorticoid-based treatment after an MRI-based diagnosis can have a good prognostic impact altogether. 10 As per an observational study in adults, early treatment with a short course of intravenous methylprednisolone followed by a tapering dose over a 4- to 6-week period led to better outcomes. 2
Our patient responded well to the glucocorticoid therapy and was discharged after 10 days of hospital stay. Antecedent COVID-19 infection, non-inflammatory CSF analysis, absence of oligoclonal bands, absence of anti-MOG seropositivity, good response to steroids, resolving follow-up MRI scans, and no new symptoms in 3-month follow-up scans in our patient were in line with ADEM.
There have been studies emphasizing on spontaneous recovery of ADEM cases over the course of weeks to months. Although initially more severe, the recovery rate has been reported to be better than that of MS. 19 As the basis for accurately diagnosing ADEM is still in debate, prospective validation from multicenter studies is required to differentiate the spectrum of multifocal demyelinating diseases. 20
Being a rare occurrence, this case can be an important reminder to be on the lookout for new-onset polyfocal neurologic symptoms after COVID-19 infection. Overall, this demands more clinical research to clarify the relationship between this neuropathogen and the ADEM, which can thereby extract intimate associations and interventions in the days to come, considering its severity and outcomes.
Conclusion
Along with other multisystemic impacts of the SARS-CoV-2 virus, the association of ADEM with COVID-19 infection has been increasingly reported. Moreover, post-COVID ADEM can be different from pre-pandemic ADEM in relation to the gap between occurrence of COVID-19 infection and onset of ADEM symptoms, severity, and mortality. Neurologists must be vigilant about the chronology of ADEM occurring after COVID-19 infection and possible neurological complications that might occur after COVID-19 infection. Till date, observational data show better prognosis with early diagnosis and treatment; key clinical features (presenting with novel neurologic deficits, fever, and encephalopathy) along with MRI findings (acute demyelinating lesions in the absence of older lesions) being the best tool for diagnosis. Given the novelty of pandemic, the better understanding of the spectrum of neurological manifestations in ADEM following COVID-19 relies on further studies.
Footnotes
Acknowledgements
The authors are grateful to the patient and her family and the Department of Neuromedicine at TUTH for their valuable support in the preparation of this manuscript.
Author contributions
A.A., S.K.Y., K.Y., and Y.K. wrote the initial draft of the manuscript. G.N. and R.O. edited the draft and reshaped it into this manuscript. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the editor-in-chief of this journal.
