Abstract
Objective:
Understanding the minimum infective dose is significant for risk assessment in the performance of suitable infection control strategies in healthcare centers. However, the literature lacks strong evidence regarding this value for severe acute respiratory syndrome coronavirus 2. Therefore, the aim of this study was to investigate the minimum infectious dose of coronavirus disease 2019.
Methods:
We searched the databases of PubMed, Scopus, Web of Science, and Cochrane and retrieved all the relevant literature by 25 July 2021. The records were downloaded into the EndNote software and underwent title/abstract and full-text screenings. A summary of included studies was organized into tables for further analysis, interpretation, and drafting of the results.
Results:
Nineteen studies including the laboratory data on human and animal hosts were selected based on the eligibility criteria. All the literature reported on the infective dose, particularly in humans. The main methods for measurement of infection were through tissue culture infectious dose (TCID50) and counting plaque-forming units. The range of minimum infective was 1.26–7 × 106.25 PFU.
Conclusion:
In this study, we have presented a range of minimum infective doses in humans and various animal species. Such numbers can possibly vary between the individuals based on numerous demographic, immunologic, or other factors.
Keywords
Introduction
The World Health Organization (WHO) declared coronavirus disease 2019 (COVID-19) disease as a global public health emergency on 30 January 2020.1,2 COVID-19 started its devastating trajectory into a global pandemic in Wuhan, China.3 –5 COVID-19 spread rapidly around the world. Globally, as of 28 March 2022, more than 480 million confirmed cases and 6.1 million deaths due to COVID-19 were reported to the WHO. 6 Most patients with COVID-19 are asymptomatic or show mild symptoms such as fever, upper respiratory tract symptoms, shortness of breath, and diarrhea, but the more severe cases can lead to pneumonia, multiple organ failure, and death. 7
The newly emerged virus that caused the COVID-19 was named as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) by the International Committee on Taxonomy of Viruses (ICTV) on 11 February 2020. 8 Coronaviruses are a large family of single-stranded RNA viruses that infect animals and humans and cause respiratory, gastrointestinal, liver, and neurological diseases.
Coronaviruses are further divided into four genera: alpha-coronavirus, beta-coronavirus, gamma-coronavirus, and delta-coronavirus. 9
There were six most identified coronaviruses before 2019 that affected the humans: (1) human coronavirus 229E (HCoV-229E), (2) human coronavirus OC43 (HCoV-OC43), (3) severe acute respiratory syndrome coronavirus (SARS-CoV), (4) human coronavirus NL63 (HCoV-NL63), (5) human coronavirus HKU1 (HCoV-HKU1), and (6) Middle East respiratory syndrome coronavirus (MERS-CoV).
The first four are recognized as endemic (that is, these are associated with mild and self-limiting diseases), the last two cases lead to severe illness and death. The seventh strain of the corona virus is SARS-CoV-2, also called COVID-19. This strain causes severe disease and respiratory infections in humans and is widespread. 10
The SARS-CoV-2 genome has great sequence similarity (89%–96.3%) with two bat coronaviruses, bat-SLCoVZC45 and bat-SL-CoVZXC21, and 79%–82% with that of human SARS-CoV. The genome consists of 14 kilo bases that encode 27 proteins. The 5′ terminus encoding for 15 nonstructural proteins collectively involved in virus replication and possibly in immune evasion. The 3′ terminus of the genome encodes for structural and accessory proteins. 11 Dose means the number of particles to cause a detectable infection. For understanding viral pathogenicity, determining the number of particles that trigger infection is crucial. Low infectious doses mean that the organism is highly contagious from person to person through contact with infected surfaces. In a US study, isolation of SARS-CoV-2 from a sample of the oropharynx and nasopharynx, one patient and inoculation into Vero cells showed that SARS-CoV-2 proliferated rapidly, reaching 105 TCID50/mL within 24 h. 12
SARS-CoV-2 infection requires a minimal dose of infection because lower doses can be safe. The minimum infectious dose indicates how much virus has entered the body and caused the infection. To determine the pattern of transmission, we need the minimum infectious dose of the virus. Given the importance and necessity of the topic, we aimed to review the literature on the minimum infective dose of SARS-CoV-2 to identify what was the lowest range of SARS-CoV-2 dose that caused the COVID-19 in the related studies.
Methods
This review study was designed to encompass all the evidence related to the infective dose of COVID-19 that has been published by 25 July 2021. The authors aimed to investigate the minimum infective dose of novel coronavirus (SARS-CoV-2) and to ensure the reliability and validity of the results, the preferred reporting items for systematic reviews and meta-analyses (PRISMA) checklist was applied (Supplemental Material 1).
Data sources
The relevant literature was retrieved by a systematic search of the keywords on the online databases comprising PubMed, Web of Science, Scopus, and Cochrane, we extracted all pertinent records published from December 2019 to 25 July 2021. The search strategy contemplated numerous keyword combinations that were identified through the MeSH (medical subject headings) database or previous articles. All the search strategies were recorded in the Supplemental Material 2, but the final search for PubMed is presented below in the query [C] (all the keywords were searched as title/abstract):
A. “COVID-19” OR “SARS-CoV-2” OR “SARS-CoV2” OR “2019-nCoV” OR “Novel Coronavirus.”
B. “Infective dose” OR “Infectious dose” OR “Minimum infective dose” OR “Minimum infectious dose” OR “Minimal infective dose” OR “minimum viral load” OR “minimum infectious viral load” OR “minimum infective level” OR “minimum infectious level” OR “Tissue culture infectious dose” OR “Plaque forming unit.”
C. [A] AND [B].
Study selection
Three independent researchers screened the retrieved studies and selected that serve the objectives of the present review by titles and abstracts. Later, the full texts of these articles were inspected carefully and based on the eligibility criteria, the most relevant studies were included in the qualitative synthesis. We included the original studies (including laboratory, animal, and human studied) that assessed the COVID-19 minimum infective dose. We also included the pre-prints as the number of studies on this matter is limited.
As part of the eligibility protocol, we deemed the exclusion criteria as below:
Non-original studies, including review articles and meta-analyses.
Publications with inaccessible full texts, conference abstracts, and abstract articles.
Ongoing clinical trials that lack published results.
Suspicion of data duplication, for example, two studies reporting the results of a single research
Data extraction
A summary of findings and minimum infective dose of COVID-19 were prepared and included in two tables by five researchers. The first table describes the characteristics of the studies and participants including first author information, type of study (e.g. experimental), publication year, country of origin, study population, age and gender, signs and symptoms, comorbidities, and laboratory data. The second table addresses host type, sampling site and method, mode of transmission, minimum infective dose, symptomatic cases (%), and clinical outcomes (%). Ultimately, other co-authors double-checked each of the selected publications to avoid potential duplications or overlaps, if any exist in the results.
Quality/risk of bias assessment
We used the Newcastle–Ottawa scale (NOS) to assess the quality of the included studies. 13 A maximum score of 9 will be allocated to the studies based on three categories of selection, comparability, and exposure/outcome.
Statistical analysis
As this study is a systematic review and not a meta-analysis, we did not aim to conduct any statistical analysis.
Results
Following a systematic search on online databases, 439 total records were identified. After duplicate removal and title/abstract screening, 298 and 77 of them remained, respectively. Finally, 19 eligible studies including human and animal subjects were included to assess the minimum infective dose, route of transmission, signs and symptoms, and outcomes of COVID-19 infection (Figure 1). Studies were conducted in six countries. Most of the studies were from the United States (n = 8), followed by China (n = 6) and Canada (n = 2). The majority of studies were experimental (n = 13), and some of them were human studies (n = 6). Experimental studies were conducted on golden Syrian hamsters (n = 5), mice (n = 3), African green monkeys (n = 3), Rhesus macaques (n = 1), and ferrets (n = 1) (Table 1).
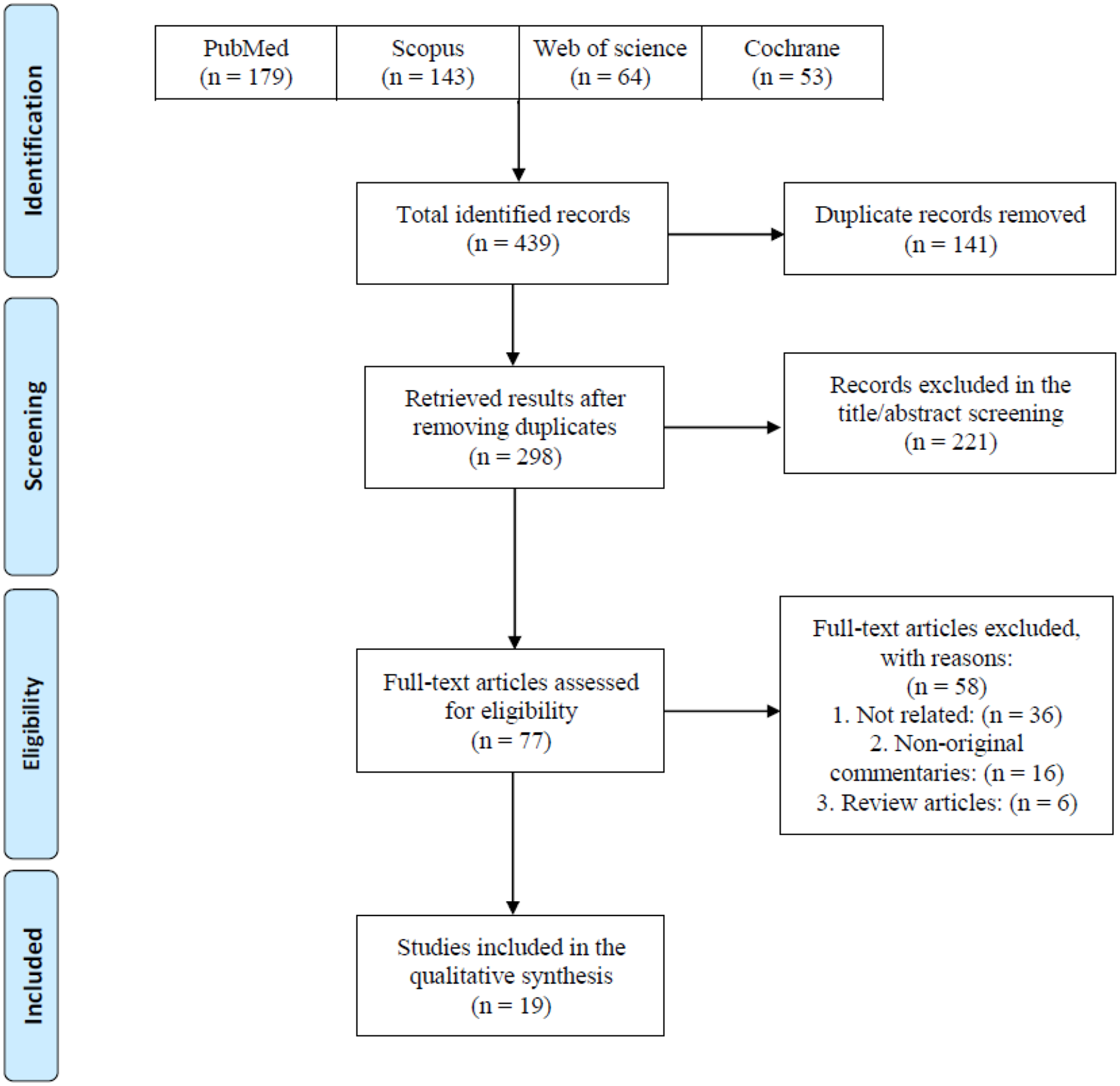
PRISMA flow diagram of the study selection process.
Characteristics of the studies and participants.

SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; COVID-19: coronavirus disease 2019; qRT-PCR: quantitative real-time polymerase chain reaction; ALT: alanine aminotransferase; CRP: C-reactive protein; aPTT: activated partial thromboplastin time; CJ: conjunctivally; IT: intratracheally.
The main methods for reporting the infective dose were through tissue culture infectious dose (TCID50) and by counting plaque-forming units (PFU). 33
In TCID50, the viral dose in 5% of inoculated tissue culture made pathological changes or cell death. PFU is estimated of viral concentration in plaque-forming units by measuring the number of particles that form a plaque. 34 The minimum infective doses have been summarized in Table 2.
Minimum infective dose of the subjects and related details of the studies.
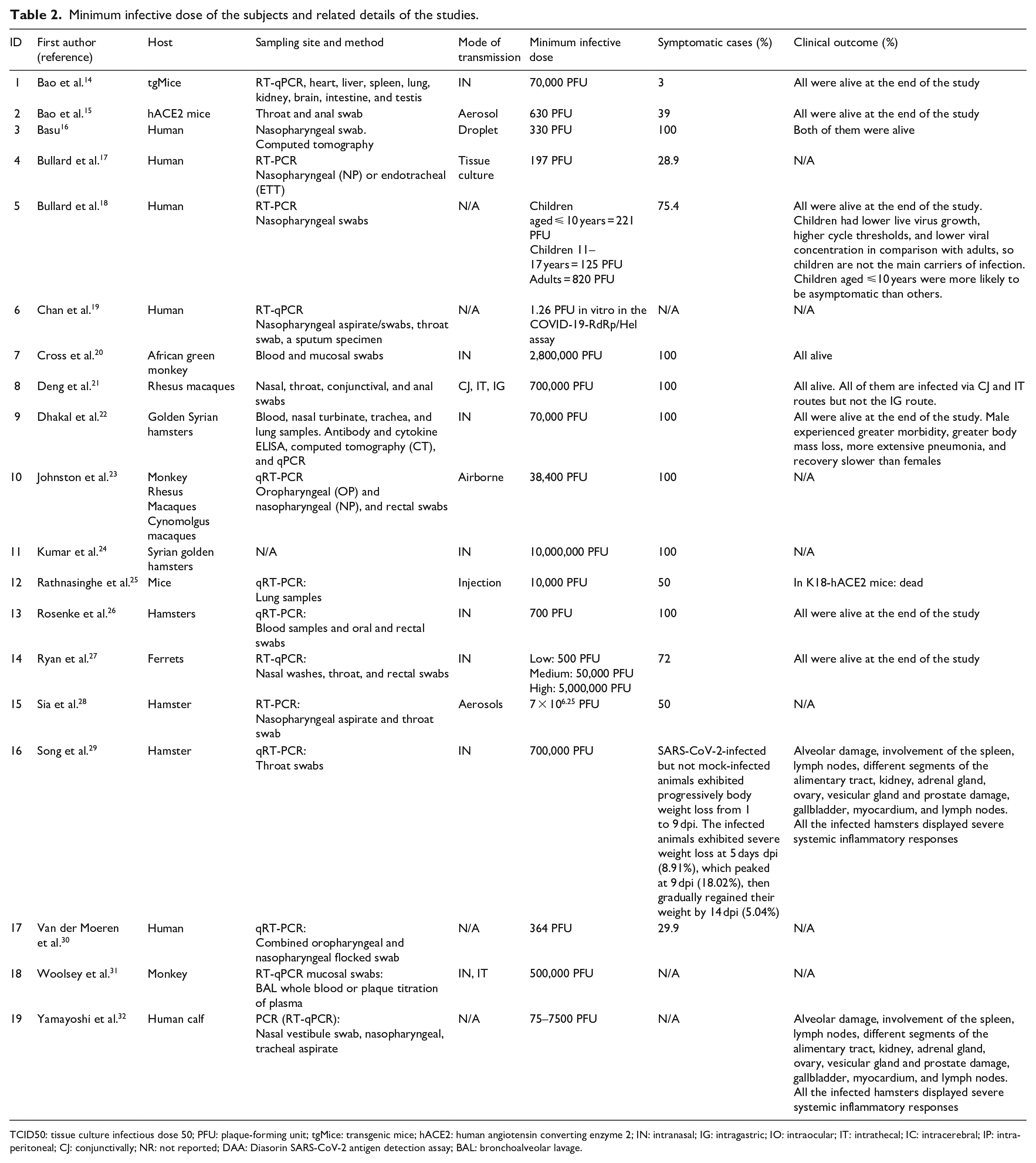
TCID50: tissue culture infectious dose 50; PFU: plaque-forming unit; tgMice: transgenic mice; hACE2: human angiotensin converting enzyme 2; IN: intranasal; IG: intragastric; IO: intraocular; IT: intrathecal; IC: intracerebral; IP: intraperitoneal; CJ: conjunctivally; NR: not reported; DAA: Diasorin SARS-CoV-2 antigen detection assay; BAL: bronchoalveolar lavage.
Human studies on infective dose of SARS-CoV-2
We found no experimental studies that assess the infective dose in human, so we included observational human studies. The minimum infective dose of SARS-CoV-2 causing COVID-19 in humans in assessed cross-sectional and case-series studies was low; in a case-series study that investigated infective dose in 273 specimens from 15 SARS-CoV-2-positive patients, detected minimum infective dose was 1.26 PFU in vitro in the COVID-19-RdRp/Hel assay. 1 In another study, 248 oro-nasopharyngeal samples of COVID-19 individuals were assessed, and infective dose was reported to be 364 PFU. 2 In a case-series study which assessed 97 children 10 years and lower, 78 children aged 11–17 years, and 130 adults, the infective dose in 11–17 years children was lower than two other groups (125 PFU). Children had lower live virus growth, higher cycle thresholds, and lower viral concentration in comparison with adults, so children are not the main carriers of infection. Children aged ⩽10 years were more likely to be asymptomatic than others. 3
Animal studies
Tables 1 and 2 summarize experimental studies of different animals on SARS-CoV-2.
Ferret
Intranasal inoculation of SARS-CoV-2 virus in three level of low (500 PFU), medium (50,000 PFU), and high (5,000,000 PFU) caused symptoms in 72% of ferrets. 27 In all ferrets of medium and high group (n = 12; 66%) that were infected at a dose of 50 000 PFU, and 5,000,000 PFU, respectively, mild multifocal bronchopneumonia was observed. Also, in medium and high groups and one ferret of low group, viral RNA shedding in the upper respiratory tract was observed. At the end of study, all of the ferrets were alive.
Mice
A study on 23 transgenic mice after aerosol inoculation at a dose of 630 PFU of SARS-CoV-2 showed weight loss and 43.5% (n = 10) of them were infected based on serological analyses. 15 Another study on 34 transgenic mice after infection at a dose of 70,000 PFU by intranasal route showed weight loss, interstitial pneumonia, and virus replication in the lungs. 14 In K18-hACE2 mice infected by the intraperitoneal injection at a dose of 10,000 PFU of SARS-CoV-2, 75% body weight loss, lethargy, ruffled fur, hunched posture, and labored breathing. Also, at the end of study, the mice died. In Ad-hACE2 Mice, infected with same route and dose, viral titers at the day 2 post-infection were seen in the lungs. 25
African green monkey
All six African green monkeys exposed to 280,000 PFU by the intranasal route had pulmonary consolidation with hyperemia and hemorrhage. Three of them had multifocal neutrophilicbroncho-interstitial pneumonia. Also, declined appetite, increased temperature, hypercapnia, gas distinction of small intestines showed in three of them. 20 African green monkeys inoculated by combined intrathecal and intranasal routes at a dose of 5.0 × 105 PFU showed decreased appetite, fever, hyperemia multifocal lesions of each lobe, mild lymphoid enlargement, pulmonary edema and pulmonary hemorrhage, pneumonia, and the small intestine was somewhat loose and dilated with gas and yellow fluid. 31 At a dose of 38,400 PFU, they showed fever, lung opacity, and mild hypoxia. 23
Rhesus macaques
After airborne inoculation at a dose of 38,400 PFU macaques showed inflammatory responses like lymphocyte and neutrophil fluctuations, fever, erythema around the eyes, and rectal bleeding. 23 Exposure to higher dose leads to moderate pulmonary symptoms in a group of rhesus macaques infected at a dose of 700,000 PFU by combined intranasal, intrathecal, and conjunctivally routs. 21
Cynomolgus macaques
All cynomolgus macaques infected with SARS-CoV-2 at a dose of 38,400 PFU via airborne presented fever, mild hypoxia, rectal bleeding, and lung opacity. 23
Hamsters
In two groups of hamsters infected with SARS-CoV-2 by intranasal routes at a dose of 700 PFU and 70,000 PFU, respectively, weight loss was shown.22,26 Also, higher dose–infected hamsters presented lung injury, and morbidity, body mass loss, and pneumonia were more severe in male hamsters. 22 All of Syrian golden hamsters infected via intranasal route at a high dose of 10,000,000 PFU showed clinical presentation. 24
The risk of bias assessment also demonstrated an acceptable quality (score of 5 or more) for all the included studies (Table 3).
Newcastle–Ottawa scale (NOS) quality assessment of the study.

Discussion
SARS-CoV-2, a lately found coronavirus accountable for COVID-19, was first reported in China in late 2019 and then extends rapidly worldwide. 35 In this systematic review, we have aimed to discover the average minimum infective dose of SARS-CoV-2. The range of minimum infective was found to be 1.26–7 × 106.25 PFU. Many of the findings that illustrate the reaction of the immune system against the virus have been experimented on animals such as mice, and the studies on minimum infective dose yield consistent results.36,37 It is vital to study the factors affecting transmissibility and mortality of the COVID-19 to curb the current pandemic, in order to predict the virus behavior in the host body and inform the preventive measure to adjust accordingly, it is essential that we find the average minimum infective dose of SARS-CoV-2. 12 Although, the infective dose differs significantly by virus and the way of administration, for coronavirus mostly hundreds or even more virus particles are needed to begin an infection. 12 Due to the rapid spread of the COVID-19 across the world, there are now more infected cases. Many people have recovered from this viral infection; however, more apprehension of virus biological behaviors is needed to control the virus. 38
One of the factors that determine the spread of the virus is its transmissibility. Finding the minimum infective dose of the virus is one of the steps which bring us closer to measuring the virus transmission power. To our knowledge, minimum infective dose is required to start an infection, because very low dosages of virus can be harmless. To substantiate this theory, the study by Bao et al. 15 showed that SARS-CoV-2 can be experimentally spread among hACE2 mice by close contact or through respiratory droplets, but it is hardly transmitted through aerosol inoculation. This could be justified by the amount of virus in the aerosol, which is less than droplets, in some cases is not even enough to cause infection; hence, a cut-off point for causing an infection known as minimum infective dose for the virus is a very useful indicator for virus infectivity in different exposure occasions. According to Basu, 16 based on a new computational strategy to quantify the infectious dose, it is estimated that the particles needed to infect humans is possibly in the order of hundreds.
A minimum infective dose indicates the amount of the virus that causes infection and reflects on the probability of infection. In line with this hypothesis, Ryan et al. show that there is a relationship between the viral load and the severity of the disease. These findings suggest that the low dose of the virus could only cause mild infection, and even in five of six ferrets did not cause infection. 27 Another method that indicated the number of virus titers is TCID50/mL, particularly it measures the amount of virus needed to kill 50% of tissue culture cells. Bullard et al. stated that the cycle threshold value was highly prognostic of culture positivity. As opposed to this statement, symptoms to test time were not capable of discriminating between cases with positive and negative cultures. 17 This dose-response relationship between the mild and high viral dose group could reflect the relationship between minimum infective dose and disease severity.
Finding the minimum infective dose of the virus can be extremely useful in determining the transmission pattern. This represents itself in inconsistent results across the included studies; similar viral load did not cause the same outcome. This indicates that despite having a similar minimum infective dose, the infection rate could differ so this minimum is not the same across the same population. On the other hand, there are some human studies which have shown some hypothetical infective viral dosages. One of the most well discussed one is the study done by Basu et al., the main goal of which was to evaluate the size of the droplets which have high probability of causing infection. But besides this finding, they also had some points related to the viral load which can cause the infection. They found that the number of virions placing at a closely situated individual’s nasopharynx over the 2.5 h duration approximates to (11/5) virions per minute × 60 min × 2.5 h = 330. They also mentioned some related past studies done on ferrets (which showed 500 as the lower tested limit for the number of virions needed to launch an infection) and on humans but with SARS-CoV (the SARS-CoV dose that correlated to 10% and 50% adverse responses (i.e. illness) was estimated, respectively at 43 and 280 PFU). 16
This controversial point should be discussed in further studies, to find whether there can be a period of minimum infective dose or not, so the idea of minimum infective dose would be useful in determining the transmission of the disease.
This study has several limitations. First, we did not aim to conduct statistical analysis as the articles were limited, and the data and their ways of presentation were heterogeneous. Moreover, we had to include the pre-print studies (e.g. on bioRxiv) because of the limited number of studies and the existing evidence, and this problem could cause some studies that are not peer-reviewed and may have some errors in methodology and results, to leak into our data. Many of the studies were also laboratory or animal studies so the average minimum infective dose could vary from the actual human circumstances. Therefore, future studies are required to address the shortcomings of our study and to compensate for the lack of information regarding human models.
Conclusion
The results of this review suggest that one of the key factors to control the pandemic could be the study of virus transmission. The minimum infective dose is one of the main components of virus transmission. In this study, we have presented a range of minimum infective doses in humans and various animal species, yet such numbers can possibly vary between the individuals based on numerous factors. Measuring the minimum infective dose can provide a clearer overall understanding of the disease and its transmissibility and help better halt its spreading.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221115053 – Supplemental material for Minimum infective dose of severe acute respiratory syndrome coronavirus 2 based on the current evidence: A systematic review
Supplemental material, sj-docx-1-smo-10.1177_20503121221115053 for Minimum infective dose of severe acute respiratory syndrome coronavirus 2 based on the current evidence: A systematic review by SeyedAhmad SeyedAlinaghi, Amirali Karimi, Hengameh Mojdeganlou, Zahra Pashaei, Pegah Mirzapour, Ahmadreza Shamsabadi, Alireza Barzegary, Fatemeh Afroughi, Soheil Dehghani, Nazanin Janfaza, Amirata Fakhfouri, Sepideh Khodaei, Esmaeil Mehraeen and Omid Dadras in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121221115053 – Supplemental material for Minimum infective dose of severe acute respiratory syndrome coronavirus 2 based on the current evidence: A systematic review
Supplemental material, sj-docx-2-smo-10.1177_20503121221115053 for Minimum infective dose of severe acute respiratory syndrome coronavirus 2 based on the current evidence: A systematic review by SeyedAhmad SeyedAlinaghi, Amirali Karimi, Hengameh Mojdeganlou, Zahra Pashaei, Pegah Mirzapour, Ahmadreza Shamsabadi, Alireza Barzegary, Fatemeh Afroughi, Soheil Dehghani, Nazanin Janfaza, Amirata Fakhfouri, Sepideh Khodaei, Esmaeil Mehraeen and Omid Dadras in SAGE Open Medicine
Footnotes
Acknowledgements
This study was conducted in collaboration with Khalkhal University of Medical Sciences, and Iranian Research Center for HIV/AIDS, Tehran University of Medical Sciences.
Author contributions
All the authors have read and approved the final version of the article. The conception and design of the study were performed by E.M. and S.S. Methodology was given by E.M., S.S., and A.K. Screening of the articles and acquisition of data were done by Z.P., P.M., N.J., H.M., S.D., F.A. Writing—original draft preparation was by A.B., A.F., S.K., A.K., and A.S. Writing—review & editing was by S.S. and O.D. Validation E.M., O.D., and S.S.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
