Abstract
Acrodermatitis continua of Hallopeau is a rare, localized variant of pustular psoriasis commonly associated with join disease and severe quality of life impairment. While there are no standard treatment guidelines, therapies used for psoriasis vulgaris are commonly tried. We report a case of severe acrodermatitis continua of Hallopeau in a patient with multiple comorbidities (advanced malignancy, recurrent empyema, psoriatic arthritis) where tildrakizumab lead to a rapid resolution of skin and joint disease which was maintained 1 year later. To date, there are only four cases reporting the use of IL-23 inhibitors class in acrodermatitis continua of Hallopeau and none for tildrakizumab. However, IL-23 inhibitors should be strongly considered among the treatment of choice for acrodermatitis continua of Hallopeau, especially in patients with ongoing malignancy and/or high risk of infections.
Introduction
Acrodermatitis continua of Hallopeau (ACH) is a rare variant of localized pustular psoriasis affecting distal digits and resulting in significant pain and disability.1,2 Comorbid osteitis/osteolysis of affected phalanges or psoriatic arthritis may be present. 2 ACH follows a chronic course and is typically recalcitrant to skin-directed therapy. 2 Given its rarity and the lack of treatment guidelines, systemic treatment options available for psoriasis vulgaris are typically used. 2 We report a challenging case of ACH successfully and safely treated with tildrakizumab. This is the first case report of tildrakizumab for ACH to our knowledge.
Case report
A 62-year-old Caucasian man known for stage III squamous cell carcinoma of the lung status post neoadjuvant chemotherapy (cisplatin–vinorelbine) and complicated right-sided pneumonectomy (repetitive empyema requiring a Clagett window) developed an inflammatory oligoarthritis, enthesitis, and skin lesions. He was started on low-dose prednisone, methotrexate, sulfasalazine for joint disease. However, his skin worsened, and he developed a drug-induced pancytopenia leading to discontinuation of steroid-sparing treatments and dermatology was consulted. Upon presentation to our clinic, painful erythema and swelling were noted on all distal digits (hands worse than feet) with associated sterile pustules and/or collarette scales (Figure 1). Severe nail hyperkeratosis and onycholysis were seen; purulent discharge could be elicited when pressing on the proximal nail folds. Small plaques of psoriasis vulgaris were seen on volar hands, feet, and knees. Clinical suspicion of ACH was confirmed by skin biopsy, demonstrating parakeratosis, sterile subcorneal spongiform pustules. While awaiting biopsy results, we attempted intralesional and ultrapotent topical corticosteroids, but there was no significant improvement 1 month later. Due to the debilitating pain (9/10), difficulty using his hands and bearing weight on his feet (Dermatology Life Quality Index = 26), concomitant arthritis, and comorbid lung cancer/open chest wall, an IL-23 inhibitor was chosen.
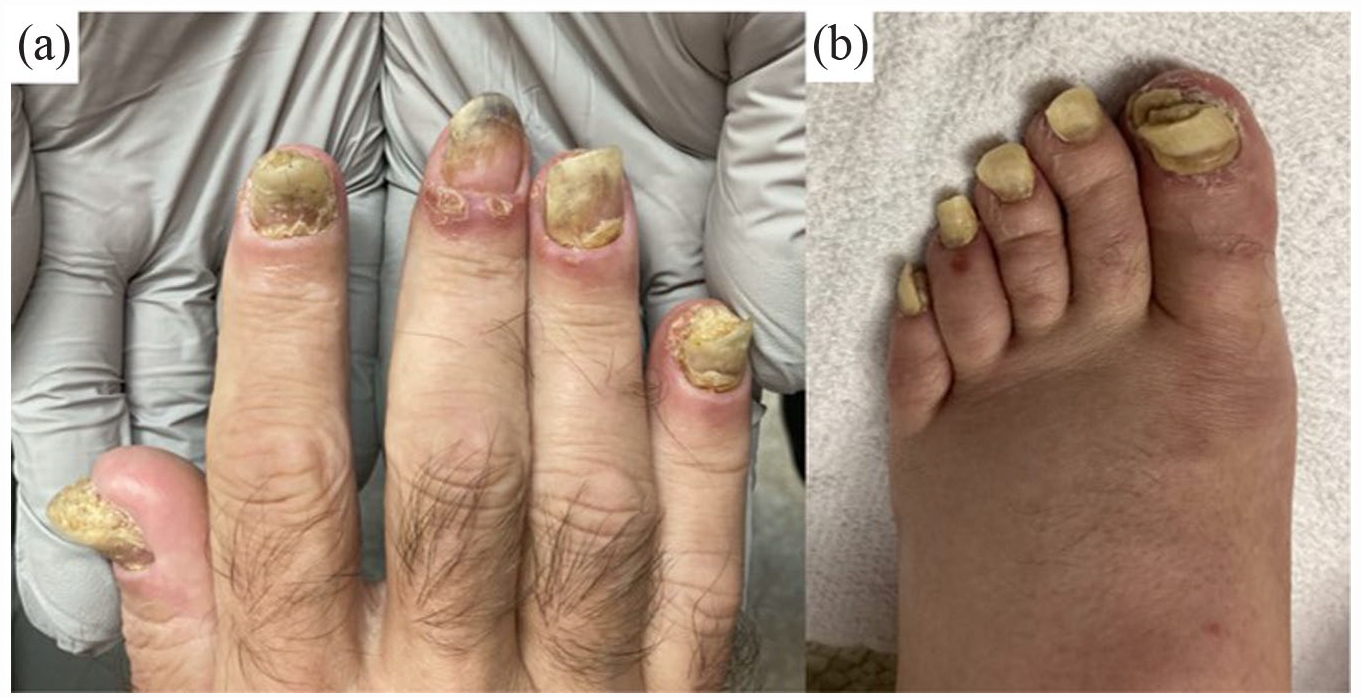
Clinical findings on initial presentation. (a) and (b) illustrate erythema and swelling of the distal digits, pustules, nail hyperkeratosis, and detachment. Of note, these pictures were taken following intralesional corticosteroids injection and ultrapotent topical corticosteroid use.
Tildrakizumab was started in September 2021 (psoriasis dosing, compassionate use). Two weeks later, decreased cutaneous pain was reported. At 3 months follow-up, skin erythema resolved, and nails improved significantly. Joint pain (bilateral knees) remained, and corticosteroids were injected into his knees by rheumatology. At 6 months of follow-up, there was no evidence of residual cutaneous or inflammatory joint disease. In June 2023 (21 months of treatment), he remained completely controlled (Figure 2(a) and (b)). No treatment-related side effects occurred during his treatment.
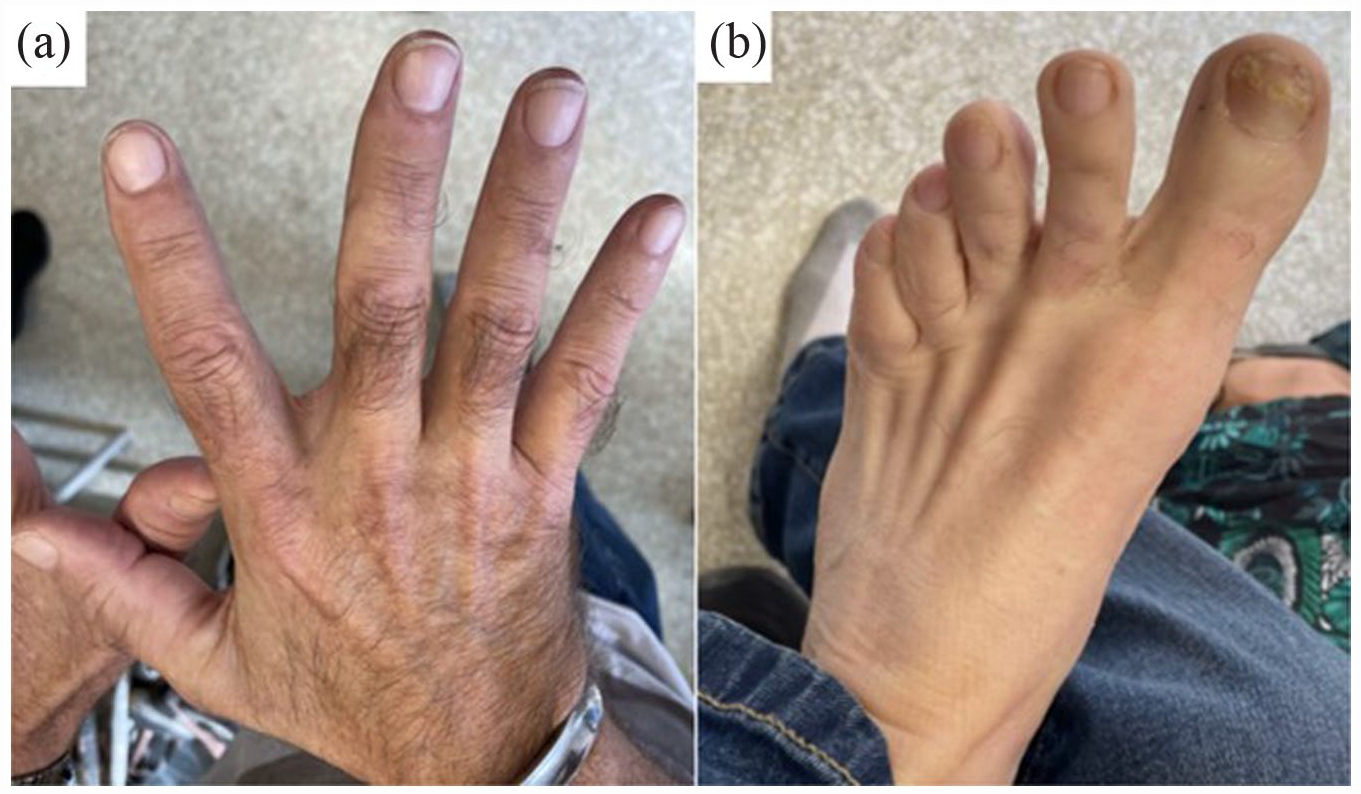
Clinical finding after > 1 year of treatment. (a) and (b) show complete remission of skin and nail disease.
Discussion
While rare, ACH is associated with severe pain and reduced quality of life. 3 It is usually recalcitrant to skin-directed therapies and conventional immunosuppressants. 4 Our patient was given methotrexate for joint disease and developed pancytopenia. Because of the underlying cancer, recurrent empyema, open chest wall (Clagett window), and concomitant psoriatic arthritis, acitretin and cyclosporine were not good options. Biologics have revolutionized the treatment of psoriasis and hence make a reasonable approach to try in special subtypes of the disease, including ACH. 2 Among the biologics, there are successful reports using tumor necrosis factor α (TNF-α) inhibitors, ustekinumab, guselkumab, and IL-17 inhibitors. 4 Japan’s Ministry of Health, Labour and Welfare (MHLW) recently approved guselkumab and ixekizumab for generalized pustular psoriasis. 4 Hence, There have been four published cases of ACH treated with IL-23 inhibitors (guselkumab and risankizumab). 5 These patients had failed previous therapies (including other biologic agents) and showed complete responses to the IL-23 inhibitors with no adverse events noted with a mean observation period of 10 months. 5
To the best of our knowledge, this is the first case describing the successful use of tildrakizumab for ACH. The clinical response to tildrakizumab has been maintained for over 1 year of therapy, with no adverse events noted. Our findings highlight the promising treatment of ACH with IL-23 inhibitors.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.N. has received investigator-initiated research support and/or acted as a consultant and/or speaker for Sun Pharma, Novartis, AbbVie, Beiersdorf, Leo Pharma, Eli Lilly, Sanofi, Bausch Health, BMS, Janssen, Mallinckrodt, UCB, Medexus, Boehringer Ingelheim, Eli Lilly, Galderma, Sandoz.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The patients in this manuscript have given written informed consent to the publication of their case details.
