Abstract
Accidental falls and major depressive disorder (MDD) are two common conditions associated with aging. Initial treatment of MDD often starts with administering antidepressants, followed by transcranial magnetic stimulation (TMS) for treatment-resistant individuals. The purpose of this case study was to determine the effect of repetitive TMS (rTMS) on postural control of an individual with MDD. A 44-year-old male with recurrent severe MDD was assessed for postural balance during eyes closed and eyes open conditions, pre and post three consecutive sessions receiving high-frequency rTMS (NeuroStar). Total excursion and velocity of sway significantly decreased following rTMS treatment when eyes were closed (p < 0.05). Power of the sway changed, but the changes were not statistically significant. The fractal dimension confidence circle area decreased significantly in eyes closed trials (p < 0.05). It appears that rTMS application can potentially impact postural steadiness in individuals with MDD. Our results warrant further studies with larger study samples.
Introduction
Accidental falls and major depressive disorder (MDD) are two common conditions associated with aging. 1 Falls are the leading cause of death due to unintentional injuries. 2 Approximately 25% of older adults over the age of 65 fall once or more per year. 3 In addition, nearly 10% of older adults suffer from MDD, 4 which is a prevalent and debilitating psychiatric condition that influences all aspects of life. Patients with MDD have been shown to have neuronal atrophy and a loss of synaptic connections in the cortical and limbic brain regions. 5
Studies have established a direct relationship between MDD and postural control deficits. 6 Initial treatment of MDD often starts with administering antidepressants. However, some antidepressants (e.g. fluoxetine, doxepin and amitriptyline) most likely debilitate psychomotor skills and postural control.7,8 An alternative solution for treatment-resistant MDD is transcranial magnetic stimulation (TMS). During TMS sessions, non-invasive brain neuromodulation is achieved via a magnetic pulse (induced through a magnetic coil) that is delivered to the brain cortex, which in turn creates an inverted electric charge and depolarizes neurons in brain tissue. 9
Recently, TMS has become a common modality to address symptoms of MDD when resistance to psychopharmacological treatments is experienced. Studies have shown that TMS treatment for MDD is highly effective.10,11 The effect of TMS treatment on postural control, however, is not yet well understood. A recent study on stroke patients reported that TMS treatment may improve static postural stability and postural control, as determined by the Berg Balance Scale. 12 To our knowledge, no other studies have explored the potential effects of TMS treatment on the postural steadiness of individuals with MDD.
Hypothesis
Although the dorsolateral prefrontal cortex (DLPFC) is primarily associated with executive functions, its role in the control of posture, particularly during cognitively demanding and complex sensory tasks, is well established.13–16 Cognitive impairments are shown to impact posture negatively by changing several variables of postural steadiness (e.g. sway velocity) and increasing the risk of accidental falls.17,18 At the same time, cognitive disorders in different domains are highly prevalent in individuals with depressive disorders,19–21 and often, symptoms of cognitive disorders persist even when symptoms of depression are in remission. 19 Considering this link between depression and poor postural balance, 22 a potential improvement of postural control may be anticipated when depressive symptoms are affected, particularly with treatment modalities that not only improve depressive but also cognitive symptoms as well. The standard high-frequency repetitive TMS (rTMS) protocol for individuals with depressive disorders is a non-invasive approach that aims to induce electrophysiological excitatory effects to improve DLPFC function by recruiting more neural resources and improving conflict resolution in cognitive control processing. 23 Thus, we anticipate that high-frequency rTMS, delivered under the standard protocol for MDD, may improve postural control via its antidepressant and precognitive effects as reflected by changes in postural sway variables. The purpose our case study is to identify whether high-frequency rTMS therapy can potentially affect postural steadiness as determined by time and frequency domain variables of postural sway.
Case report
Following approval of the Institutional Review Board at Northern Illinois University, a 44-year-old male with recurrent severe MDD (without psychotic features) and post-traumatic stress disorder was recruited to participate in this study in the summer of 2021. The participant scored 20 on the Patient Health Questionnaire-9 (PHQ-9) 24 and was taking bupropion HCL (150 mg), clonazepam (1 mg), prazosin HCL (5 mg), lamortrigine (200 mg), aripiprazole (15 mg), and benztropine (1 mg). Otherwise, the patient had no physical disability or visual or vestibular deficit that could potentially affect their balance, could ambulate without any assistance or use of an assistive device, and could stand upright independently for at least 10 min. The patient was not using any alcohol or recreational drugs, did not drink any caffeinated beverages at least 24 h prior to the tests and refrained from exercising heavily at least 8 h before the tests. The patient was scheduled for 36 rTMS sessions (one visit per day) and was tested before the first session and then after visits 15, 16, and 17. The patient was required to sign a consent form prior to participation in the study. In addition, written informed consent was obtained from his representative to use the anonymous data for publication.
The participant received high-frequency rTMS (NeuroStar) over the left DLPFC using a figure-8 coil. The stimulus was set at 120% of the motor threshold and delivered at a frequency of 10 pulses per second. Each 30-s cycle consisted of 4 s of active stimulation with 26 s of no stimulation. A total of 3000 pulses was delivered per session. A Kistler force platform (Kistler Co., Winterthur, Switzerland) was used to collect position data for the center of pressure (COP) at 100 Hz. A LabVIEW program (National Instrument, Austin, Texas) was developed to collect postural sway data.
At each visit, the participant was randomly assigned to two standing conditions: (a) eyes open and (b) eyes closed before and after (pre-post) the rTMS application. The condition of eyes closed was included to estimate changes in postural steadiness when the visual information required for postural control was challenged. Considering that vestibular, visual, and sensory information are typically used to maintain upright posture, deterioration of any of these sources of information may reveal information regarding an individual’s dependency on the lost source. These test conditions (i.e. eyes open, eyes closed) were repeated three times before and after treatment. Test orders (i.e. eyes open vs eyes closed) were block randomized (for pre and post treatment separately), with each condition presented once in each block.
During each session, the participant was instructed to stand straight and static with arms by their sides (bare feet, heels together, 5°–7° of toe-out) on the force platform. Data were collected for 45 s (Fs = 100). For the eyes closed condition, the participant was asked to close their eyes throughout the trial. While there were no specific resting periods implemented between trials, the participant was informed prior to and during the testing that they were welcome to request a rest time if needed. Anteroposterior and mediolateral time series data were filtered through a fourth-order zero phase Butterworth low-pass filter with a cutoff frequency of 5 Hz. The first 8 s and last 2 s of data were cut to remove any potential lead-in/lead-out effect. MATLAB and Toolbox Release 2012b (MathWorks, Inc., Natick, Massachusetts) were used to filter the postural sway data and to compute variables of interest. Time and frequency domain variables of postural sway were computed. A detailed explanation of the computation methods used for the variables and equations is available in the literature.25–30 Total excursion of sway, identified as the total distance that the COP travels while standing in a static position, was computed by summation of the distance between two consecutive data points
Velocity of sway was calculated by dividing total excursion over time
Power of sway signal was computed as the integrated area of the power spectrum, and 95% power frequency was determined as the point below which 95% of the total power was placed. 26
Although we only tested one participant, the repeated measures allowed us to quantify the within-subject variation in the outcome measures. Therfore, it is reasonable to assume that the measures are independent after controlling for the four variables: Factor (i.e. pre and post), Condition (i.e. eyes open and eyes closed), Factor × Condition, and Visit. We first calculated means and standard deviations (SDs) on several outcome measures (including mean velocity, resultant power, and sway velocity) across the four possible groups created by Factor and Condition. Then, we used the general linear model (GLM) to model each outcome measure after controlling for Factor, Condition, Factor × Condition, and Visit. All statistical tests were 2-sided with a p value less than 0.05 indicating a statistically significant effect. All models were fit in SAS (version 9.4) using PROC GLM.
Following completion of the 36 rTMS sessions, the participant’s PHQ-9 score was reduced from 20 (severe depression) on the first day of the treatment to 10 (mild depression). Comparison of mean velocity (MVELO) of sway showed that the participant swayed at a lower velocity following rTMS treatment (18.5 ± 3.19 mm/s pre and 17.51 ± 2.1 mm/s post). When postural control was challenged by eliminating visual input (eyes closed), changes in velocity of sway following rTMS application were more substantial when the participant’s eyes were closed as opposed to their eyes were open (33.38 ± 7.69 mm/s eyes closed—pre rTMS vs. 28.41 ± 2.45 mm/s eyes closed—post rTMS). Total excursion of postural sway also changed following rTMS application. The boxplots in Figure 1 also show the same pattern. After controlling for the visit effect, the GLM results (Table 1) show that no significant changes of mean velocity were observed pre and post rTMS for the eyes open condition, but mean velocity of sway was significantly reduced following rTMS sessions (p = 0.005). The R-square value indicates that the 83.7% within-subject variation was explained by Factor, Condition, and Visit. Figure 2 shows a trend that mean velocity of sway decreased over the three visits. At visit 3, the mean velocity was reduced to 14.37 (95% confidence interval (CI): 11.46–17.29) when the participant’s eyes were open and 25.27 (95% CI: 22.35–28.19) when their eyes were closed.

Figure depicts comparison of total excursion (a) and mean velocity (b) of sway pre and post TMS sessions. Two different conditions of eyes open (EO) and eyes closed (EC) are presented. Note the difference between eyes open and eyes closed conditions as well as constant changes following rTMS sessions.
General linear model (GLM) results: estimates and standard errors (SEs) for certain effects.
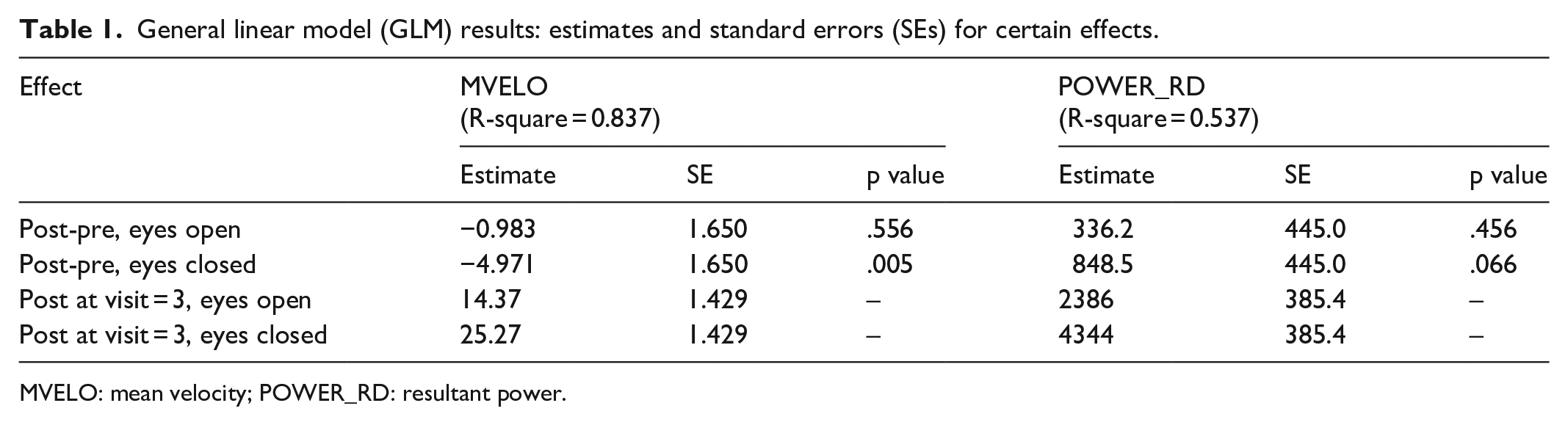
MVELO: mean velocity; POWER_RD: resultant power.

Figure illustrates fit for mean velocity of sway with 95% confidence limits and compares changes of postural velocity for different visits. Note significant and continued change of sway velocity following each visit.
The power of sway signal showed significant changes between the eyes open and eyes closed condition both before and after the rTMS sessions (p < 0.05). Comparison of sway signal power changes before and after rTMS sessions for each condition, however, did not show statistically significant changes (Figure 3).

Figure presents the resultant power spectral density of sway (a) and fractal dimension confidence circle area (b) of sway signal. The figure shows pre and post TMS sessions as well as eyes open (EO) and eyes closed (EC) conditions. Note that changes between pre and post TMS sessions are always augmented when eyes are closed.
The fractal dimension confidence circle area (FD_CC) was also compared between the two conditions and between pre and post rTMS sessions. Our results showed that FD_CC during the eyes open condition did not significantly change between pre and post rTMS session (p = 0.05). On the other hand, when eyes closed conditions were compared, the decrease of FD_CC following rTMS sessions was statistically significant (p < 0.05) (Figure 3).
Discussion
As expected, we noticed significant changes of postural sway between eyes open and eyes closed conditions. Although changes of sway following rTMS sessions during eyes open trials occurred, these changes were not statistically significant. On the other hand, when postural control was challenged with the eyes closed condition, all postural sway variables were changed significantly. Total excursion of sway represents the total length of the COP path. An increase in total excursion of sway has previously been reported in the literature as a result of aging or lower extremity amputation. 31 Total excursion of COP, which is an indicator of poor postural stability, was substantially decreased in our patient following rTMS sessions. As expected, we noticed a similar pattern of changes in sway velocity before and after rTMS sessions. Improvement of time domain variables of postural sway, particularly sway velocity, indicates a decrease in the risk of falling. Velocity of sway is reported as the main variable representing quality of postural control and the effect of aging.26,32
In our patient, we also assessed frequency domain variables of postural sway. Previous studies have suggested the use of frequency domain variables in conjunction with time domain variables (e.g. total excursion and sway velocity) to reveal more valuable information.25,26,33 Analysis of the frequency content of a signal may reveal underlying changes that are often not observed in a time series. 34 Our results indicated significant changes in the power of sway signal following rTMS sessions. Although these changes were not substantial during eyes open trials, they were significantly higher in eyes closed trials following the rTMS sessions. Such changes may indicate increased simultaneous muscle activities that may not be well reflected in a time domain series.
We also compared fractal dimension of sway signal pre and post rTMS sessions. Fractal dimension of postural sway is a unitless measure indicating the degree to which a curve fills the space it encompasses. 26 It is suggested that a fractal dimension, when compared with other characteristics of sway, more accurately detects changes of postural sway. 35 We found that fractal dimension of postural sway substantially reduced following rTMS sessions. The increased pattern of changes in fractal dimension is consistent with those reported by Prieto et al., 26 suggesting an increase of fractal dimension by aging. Therefore, rTMS treatment may have the potential to improve postural steadiness and reduce the risk of accidental falls.
Several limitations and confounders are associated with our findings. First, having a single subject prevents us from drawing general conclusions from our findings. The findings from this study are based on a single case and may be influenced by the patient’s wakefulness level. 36 Our study participant complied with the instructions to refrain from caffeine or alcohol consumption at least 24 h before the test and heavy exercise at least 8 h before the test. However, other physical and physiological variables, such as stress level and hours of sleep the night before each testing session, were not controlled, and this may have affected the results. In addition, since our findings are based on a single case, placebo effects cannot be ruled out due to the lack of a sham rTMS condition. Another limitation of the study is that the study assessments were only conducted in the middle of the patient’s 6-week rTMS course; future studies may have more significant findings when measuring postural control before the rTMS course and after the conclusion of 36 sessions of treatment. However, despite these limitations, our findings are interesting enough to warrant investigation in randomized controlled studies that can also examine the sustainability of rTMS-induced postural stability.
Conclusion
Overall, our results show substantial changes of time and frequency domain variables following high-frequency rTMS sessions over the left dorsolateral prefrontal cortex. Of note, we show an improvement of postural sway after each individual rTMS session and an overall continuous reduction following three consecutive rTMS sessions. Our findings provide initial evidence for use of the high frequency left DLPFC rTMS protocol in improving postural control. This case report justifies further studies with larger sample sizes to determine the benefit of rTMS on improving postural control and reducing the risk of falls.
Footnotes
Acknowledgements
The authors thank the school of Allied Health and Communicative Disorders at Northern Illinois University for their equipment support for the data collection of this study. The authors also thank Ms. Ashley E. Bartelt, Coordinator of Special Programs at Northern Illinois University, for her editorial help.
Author contributions
Hamid Bateni and Elham Soltani contributed to all aspects of the study, including design, data collection, data analysis, discussion and writing of the manuscript. Mujeeb U. Shad contributed to the design of the study, data analysis, discussion section and writing of the study. Kishwar Ali contributed to data collection and the discussion of the study. Haiming Zhou performed statistical analysis of the data and contributed to the discussion of the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from Northern Illinois University, DeKalb, IL
Informed consent
Written informed consent was obtained from the patient and the patient’s psychiatrist for his participation in the study and use of anonymized study data for publication. Participant had decisional capacity as confirm by his doctor. Written informed consent was obtained from participant’s legal representative to use anonymous data for publication.


