Abstract
A 38-year-old Asian male with severe mitral valve regurgitation underwent elective minimally invasive mitral valve repair with artificial chordae and concomitant Cox-Maze procedure. Cardiopulmonary bypass required large peripheral cannulas due to the patient’s increased body surface area with a total bypass time of 216 min. At 10 h, the patient reported progressive right lower extremity pain with evidence of swelling, diffuse paresthesias, and weak peripheral pulses. The patient underwent double-incision lower leg fasciotomies, revealing significant interstitial fluid and bulging muscle chambers. Compartment syndrome demonstrates non-traumatic etiologies. Elevated body mass index, Kawashima Type D femoral artery classification, prolonged bypass times, driven partially due to concomitant Cox-Maze, and larger cannula sizes should increase the index of suspicion.
Keywords
Introduction
Compartment syndrome (CS) is defined as blood perfusion insufficient for tissue viability secondary to high pressure within a closed fascial space. 1 Classically, CS of the lower extremity is associated with high-energy injuries, presenting with some combination of the “6 P’s”: pain out of proportion to injury (or pain with passive stretch), pallor, paresthesia, pulselessness, paralysis, and poikilothermia. 2 CS should remain on the differential diagnosis in any intervention with possible local ischemic compromise. In recent decades, minimally invasive cardiac surgery (MICS) through a small 4 cm incision is increasingly being utilized in the cardiovascular surgical arena. Cardiopulmonary bypass (CPB) established in MICS is performed peripherally through femoral vessel cannulation. However, this increases the risk of ischemia in the lower extremities. 3 Elevated body mass index (BMI), large cannula size, Kawashima Type D femoral artery classification, 4 and extended CPB times can be risk factors relevant to MICS.
Case report
A 38-year-old Asian man presented for elective MICS. Symptoms included fatigue and dyspnea on exertion, classified as New York Heart Association class II. Workup revealed atrial fibrillation on electrocardiogram and a Carpentier type II mitral regurgitation with multi-segmental prolapse of A2, A3, and posterior commissure segments on transesophageal echocardiography. Pulmonary artery pressure was 27 mmHg. Of note, the patient had an elevated BMI of 28.7 kg/m2 (overweight when using adjusted Asian Pacific-Islander ranges) 5 and an elevated body surface area of 2.16 m2. The preoperative European System for Cardiac Operative Risk Evaluation II (EuroSCORE II), which predicts the risk of in-hospital mortality after cardiac surgery, was 0.75%, indicating a minimal risk of mortality. 6
The patient underwent minimally invasive mitral valve repair including a concomitant Cox-Maze procedure. The procedure is accomplished through a 4 cm incision at the right anterior fourth intercostal space. CPB was established peripherally with cannulation by open Seldinger-guided technique 7 with the following cannulas & locations: 22-Fr right femoral artery, 23/25-Fr right femoral vein, and 16-Fr right internal jugular vein. Due to intraoperative rapidly rising A-line pressures (260 to 270 mmHg) and increasing lactate levels (3.8 to 7.9 mmol/L), an additional 22-Fr left femoral artery was inserted to optimize arterial perfusion during CPB. Perioperatively, multi-segmental prolapse of A2, A3, and posterior commissure segments were identified, and artificial chordae loops were required to secure the A2-A3 as well as cusp commissuroplasty. After the mitral valve repair was confirmed by transesophageal echocardiography, we began to wean off the CPB. Femoral venous and arterial cannulas were removed and cannulation sites were sutured. The total CPB time was 216 min.
The patient remained intubated for 6 h postoperatively and extubated without issue. At 8 h, he reported right lower extremity discomfort with clinical evidence of mild swelling; however, overall patient grogginess made symptoms difficult to interpret. On examination, the right groin appeared grossly normal with no evidence of hematoma and no femoral arterial lesions evident on Doppler ultrasound. At 10 h, the patient noted rapidly progressive right lower extremity pain with evidence of cold, tenderness on palpation, diffuse paresthesias below the knee, and weak peripheral pulses.
A working CS diagnosis was made, and the patient underwent a double-incision fasciotomy. Upon surgical decompression of all four compartments, significant interstitial fluid was observed with healthy-appearing and contractile musculature without evidence of necrotic tissue. Visual bulging of muscle chambers was noted, cinching the CS diagnosis (Figure 1).

Perioperative photograph, showing fasciotomy procedure. (a, b) Medial incision fasciotomy on the right lower limb reveals healthy-appearing and contractile musculature without evidence of necrotic tissue and visual bulging of muscle chambers. (c) Anterior view on incisions closure.
Within an hour after fasciotomy, the patient endorsed a resolution of preoperative numbness and pain. The wounds were closed after 5 days. The patient was discharged within 10 days of initial admission without 30- and 90-day complications.
Discussion
This reported case presented a number of obstacles surgically. The standard approach for CPB is to supply 2.4 L/m2/min of blood flow. 8 This patient’s elevated 2.16 m2 body surface area necessitated larger cannula sizes (i.e. 20-Fr or greater), subsequently requiring appropriately large patient arterial diameters of at least 6.5 mm to achieve proper perfusion. Generally, a 20 to 22-Fr femoral arterial cannula is sufficient in the Asian population, and the 22-Fr cannulation was the majority. 9 Kitahara et al. 10 remarked that alternative perfusion access should be considered when the patient’s body surface area is over 2.00 m2 (which 20-Fr cannula cannot be adequate) and when the initial arterial cannula pressure is elevated (approximately 300 mmHg). Several alternative perfusion sites can be documented, including bilateral femoral artery, additional right axillary artery, side-arm graft for femoral artery, and traditional ascending aorta site. Nevertheless, a bilateral femoral arterial cannulation approach was the most regular alternative perfusion access.9–11 Femoral artery sizes on the right limb are summarized in Figure 2. The patient’s right deep femoral artery diameter measured smaller than the superficial femoral artery (5.68 and 6.65 mm, respectively) without additional side-perforators at the bifurcation (Figure 3). In addition, this patient presented with Kawashima Type D (deep femoral artery diameter < superficial femoral artery diameter without an ectopic side branch), which is associated with elevated risk for limb ischemia. 4 This combination of elevated body surface area with relatively inadequate femoral vessel size and type D anatomical classification made balancing optimal perfusion during CPB without encouraging a dangerously elevated arterial line pressure difficult. Overall, the patient was discharged at 10 days, with typical MICS procedures requiring 5–7 day admissions.

Right femoral artery diameter (mm), sizing with OsiriX/Horos imaging software. (a) CFA and SFA at A (9.18 × 9.48), B (6.42 × 6.87), and C (6.84 × 6.46). (b) CFA and DFA at A (9.02 × 8.89), B (5.59 × 5.69), and C (5.78 × 5.64).
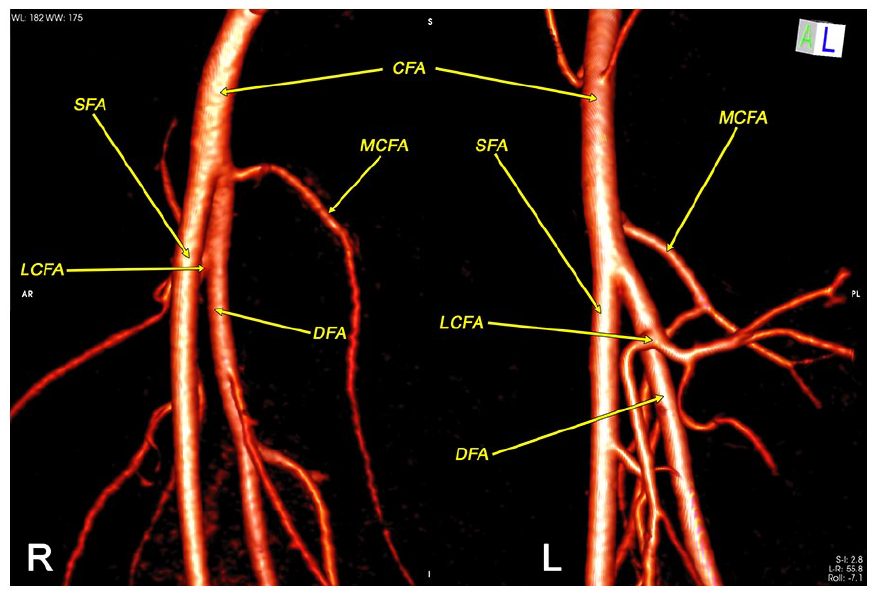
Bilateral femoral vessels via preoperative computed tomography angiography, 3D sizing with OsiriX/Horos imaging software. R: right femoral artery with missing vessels at CFA and the bifurcation position. L: left femoral artery with MCFA branch at CFA above the bifurcation position.
Of note, MICS are increasingly done concomitantly with Cox-Maze procedures. A risk-benefit analysis must be done as this joint procedure elongates operation time. Cases with prolonged extracorporeal circuit (i.e. >4–6 h) should elevate the index of suspicion for acute CS. In addition, laboratory diagnostic studies can aid in risk stratification. This patient demonstrated a significantly elevated creatinine phosphokinase of 76,947.4 U/L on post-op day 1 and 70,467.5 U/L the following day (normal range, 22–198 U/L). Valdez et al. 12 found a creatinine kinase level > 4000 U/L may suggest CS. Lactate concentrations can also be informative. Mitas et al. 13 demonstrated that lactate gaps of over 3 mmol/L might be predictive of CS. Nevertheless, should clinicians suspect a diagnosis of CS, no paraclinical findings should postpone notification of appropriate personnel to conduct the urgent fasciotomy.
Conclusion
In MICS, decreased limb perfusion during CPB can provoke limb ischemia and lead to CS. Patients presenting with high BMIs, requiring large cannula size, Kawashima Type D vasculature, and extended operation times are risk factors for limb ischemia.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from Ethics Committee of the University of Medicine and Pharmacy at Ho Chi Minh City number 166/HDDD-DHYD.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Vinh D.A. Bui was funded by Vingroup JSC and supported by the Master, Ph.D. Scholarship Programme of Vingroup Innovation Foundation (VINIF), Institute of Big Data, code VINIF.2021.TS.109.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
