Abstract
Pulmonary air leak syndromes involve dissection of air out of the normal pulmonary airspaces and include pulmonary interstitial emphysema, pneumothorax, pneumomediastinum, pneumopericardium, pneumoperitoneum, subcutaneous emphysema and systemic air embolism. It presents as a spontaneous extension of dissecting air without a history of a procedure or penetrating injury. Pulmonary air leak syndromes are extremely rare complications of systemic autoimmune connective tissue diseases. Few cases were reported in the literature regarding rheumatoid arthritis patients. The purpose of this article is to emphasize on this rare pulmonary complication and discuss the physiopathology of the disease and the different risk factors for a better management of these patients. We report the case of a 45-year-old female, with a history of proven rheumatoid arthritis under methotrexate and steroids, who presented with a spontaneous dissecting subcutaneous emphysema, pneumothorax, pneumomediastinum and pneumoperitoneum. The patient’s condition improved after chest drainage and adjustment of her medical treatment.
Introduction
Rheumatoid arthritis (RA) is a systemic chronic inflammatory disease, primarily affecting joints, with respiratory complications involving different lung structures. 1 Pulmonary air leak is less frequent in systemic autoimmune connective tissue diseases (CTDs) and the exact physiopathology is not clear yet.
Imaging, especially computed tomography (CT) scan, is essential to assess the severity of the air leak, the presence of an underlying interstitial lung disease (ILD) or an infection. 2
Therapeutical management is, by and large, based on pulmonary care support, the control of the disease and the eviction of the risk factors.
Case representation and imaging features
A 45-year-old woman presented with symmetrical inflammatory polyarthritis affecting multiple peripheral joints with signs of cutaneous vasculopathy and ILD confirmed to be related to active RA with positive blood test results for rheumatoid factor and anticyclic citrullinated peptide (anti-CCPs), as well as a high level of C-reactive protein and erythrocyte sedimentation. Disease Activity Score28 (DAS28) was high (5.7), indicating a severe activity of the disease. She was put under prednisolone tablets (60 mg per day) and methotrexate (7.5 mg per week).
Three months later, she acutely presented with marked dyspnea and non-tender fluctuant swelling in the supraclavicular fossae with palpable and auscultated crepitations over it, gradually increasing in size, involving the neck with moderate back and abdominal pain.
High-resolution CT was performed showing dissecting subcutaneous axillary and neck emphysema, extending to chest and abdominal wall, with bilateral pneumothorax, moderate amount of pneumomediastinum and features consistent with ILD, such as nodular opacities with septal thickening due to fibrotic changes involving bilateral lower zones of the lung (Figures 1 and 2).

High resolution CT showing chest wall, axillary and neck subcutaneous emphysema with features consistent with interstitial lung disease.
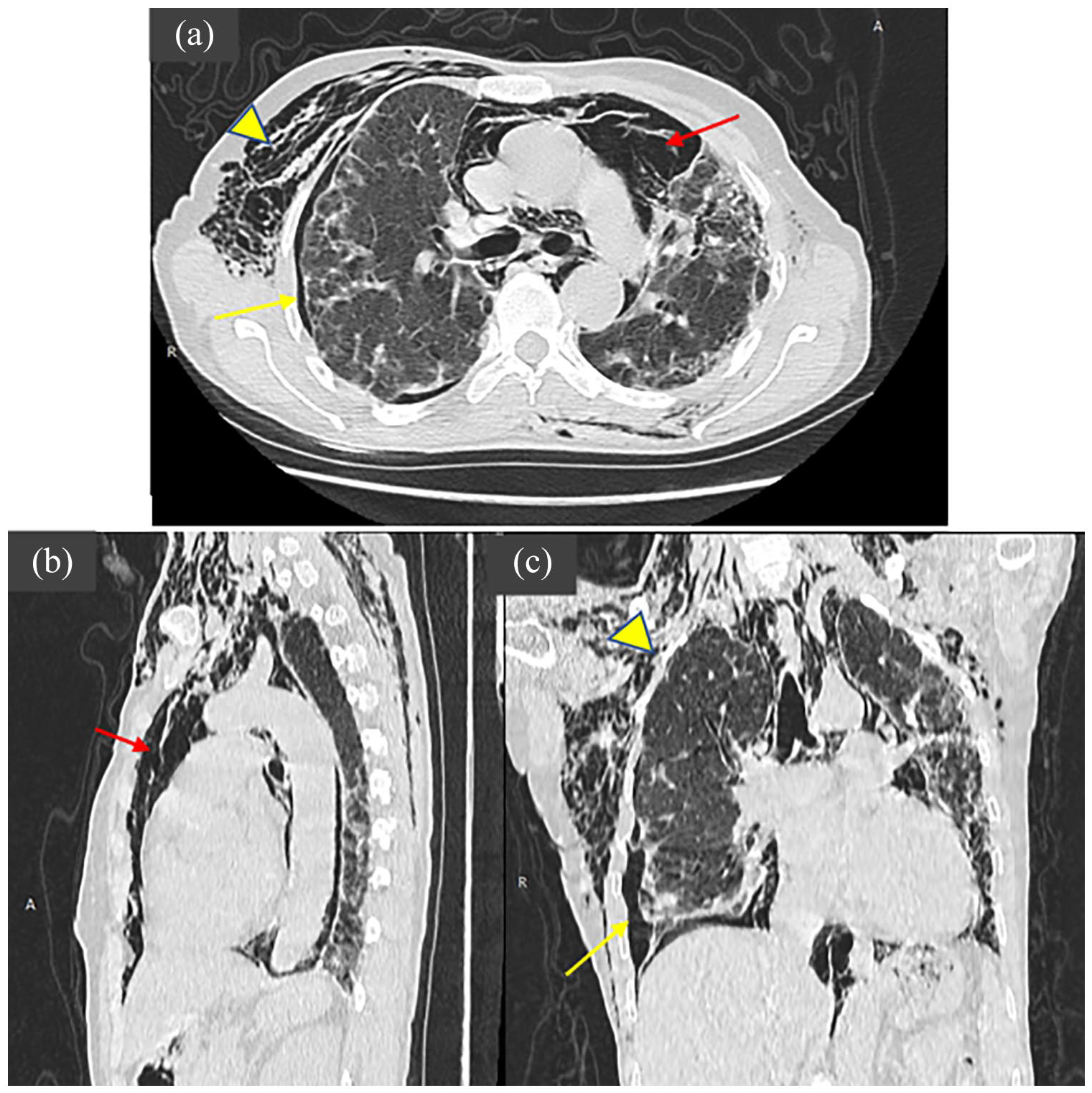
CT chest axial (a), sagittal (b), and coronal (c) images in the lung window showing diffuse subcutaneous surgical emphysema (yellow arrowhead), left pneumothorax (yellow arrow), moderate amount of pneumomediastinum (red arrow) with patterns of interstitial lung disease.
An air drainage was indicated considering the increasing size of the leakage and its important respiratory impact. Also, the air leak was attributed to the secondary effect of her treatment, prednisolone was consequently decreased to 40 mg and methotrexate was replaced by cyclosporine (3 mg/kg per day). The patient’s condition improved remarkably.
Discussion
RA is a systemic inflammatory disease characterized by destruction of joint synovium and extra-articular systemic lesions.
The most-known pulmonary manifestations of RA are ILD, rheumatoid nodules and pleural effusions.
However, pulmonary air leak syndrome is less common in RA and its presence should raise suspicion of bronchiolitis obliterans syndrome. 3
There is no exact prevalence of pulmonary air leak in the literature. However, research from database found series of 17 published cases in the past 30 years.3–19
It presents as an evolutionary manifestation of the disease but can also constitute a circumstance of discovery. 4
The risk factor includes ILD and cutaneous vasculopathy, and the accountability of immunosuppressive medication is yet to be proven. 5
Pulmonary air leak is the consequence of a barotrauma causing the rupture of a terminal bronchiole. The released air will drain along the bronchovascular structures towards the pulmonary hilum, the mediastinum and then towards the superficial cervical spaces or downwards to the abdomen. It can happen due to various factors causing thoracic hyper pressure, including coughing, an asthma attack, vomiting, childbirth or any closed glottis effort, which can overlap with a previously weakened lung making the leak easier. 20
A deleterious effect of corticosteroids on the pulmonary interstitial tissue and the possibility of the involvement of pathogenic organisms (virus, Pneumocystis carinii) have also been mentioned but not formally proven. The onset of air leak in CTDs coincides almost directly with the initiation or increasing dosages of corticosteroids.21,22
In a retrospective analysis, the clinical comparison between patients with CTD-related ILD who developed pneumomediastinum after the initiation of corticosteroid therapy and those who did not identified steroids as a risk factor for the development of pulmonary air leak especially if combined with major predictive factors such as low body mass index (BMI) and high serum lactate dehydrogenase (LDH) levels. 23
Probable lesions of pulmonary vasculitis and underlying interstitial lesions such as centrilobular and sub-pleural ‘pseudo blebs’ subsequent to cystic degeneration of fibrotic lesions or to excavation of rheumatoid nodules have also been incriminated. 3
Different modalities are used for air leak diagnosis. 24
Chest x-ray is the imaging of first intention; it demonstrates the presence of a radiolucent area of air in the lung field highly suggestive of a pneumothorax.
Pneumomediastinum can also be seen as column of air in the mediastinum. If abundant, it can produce the thymic sail sign.
Chest ultrasound is gaining popularity in the diagnosis of pneumothoraxes with 95% sensitivity and 100% specificity for the diagnosis of pneumothorax when compared with a CT scan.
It can be useful for the diagnosis of pneumothorax not seen on plain x-ray. The presence of subcutaneous emphysema can impair the accuracy of the ultrasound reading.
The sonographic sign of pneumothorax is the absence of lung sliding.
Chest CT scan is considered the gold standard in the diagnosis of air leak syndrome. It helps to differentiate other lung and pleural pathologies. This imaging modality can be potentially dangerous for critically ill patient on mechanical ventilation and vasoactive drugs because of the transporting risks.
There is no clear recommendation in the literature and a lack of expert consensus regarding best practice for pulmonary air leak syndrome management in RA. However, patients recovered conservatively in many reported cases.4–16 Furthermore, our hospital experience proved that this condition requires supportive care, the use of supplemental oxygen and control of the underlying inflammatory process.
Otherwise, if the air leak is abundant causing respiratory failure or, in case conservative treatment fails, chest drainage is then indicated. Reduction of prednisolone is often necessary, with adequate immunotherapy.
Conclusion
Pulmonary air leak syndrome is a rare manifestation that should be suspected in RA patients with acute breathing difficulties and requires close monitoring because of the possibility of a rapidly fatal evolution. The prognosis depends on several factors, including gender, use of steroids and level of air leak, which will eventually guide the therapeutical management.
Footnotes
Acknowledgements
I would like to express my gratitude to my professors and all the colleagues who participated in the completion of this work. The authors declare that they have no competing interests.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
