Abstract
This case report describes a unique pattern of human epidermal growth factor receptor 2 expression in a patient with uterine carcinosarcoma. The endometrial tumor showed biphasic morphology composed of serous carcinoma and a heterologous high-grade sarcoma component. Human epidermal growth factor receptor 2 immunostaining showed positive (3+) expression in foci of myoinvasion, lymphovascular invasion, and lymph node metastasis but was negative in both the endometrial surface tumor and sarcomatous component. Fluorescent in situ hybridization testing for human epidermal growth factor receptor 2 confirmed no amplification within the endometrial surface carcinoma component and amplification of the lymphovascular invasion component. As the use of human epidermal growth factor receptor 2 immunohistochemical evaluation becomes more commonplace for therapeutic consideration in patients with uterine carcinosarcoma, interpretation of the immunohistochemical should be performed preferentially on large tissue sections including both a surface, myoinvasive portions, and suspected areas of lymphovascular invasion and lymph node metastasis.
Keywords
Introduction
Uterine carcinosarcoma, also known as malignant mixed Müllerian tumor, is a rare and highly aggressive cancer that accounts for <5% of reported uterine cancer cases. 1 Commonly, myometrial invasion and lymphovascular invasion (LVI) are found at the time of diagnosis. 1 Histologically, this tumor is characterized by a biphasic appearance and is composed of both a carcinoma and sarcoma component. Endometrial serous carcinoma (ESC), the most prevalent subtype of carcinoma seen in these tumors, has been known to preferentially metastasize, and therefore appears to influence the clinical behavior. 2
The use of HER2-targeted therapy has been well established in breast carcinoma and subsequently investigated in ESC since the early 1990s.3,4 The use of HER2-targeted therapy without adjunctive chemotherapy for the treatment of ESC has generally not shown beneficial clinical responses, 5 but a more recent phase II clinical trial for patients with ESC revealed that the addition of trastuzumab, a HER2-targeted monoclonal antibody, to carboplatin–paclitaxel chemotherapy showed an increase in progression-free survival. 6 Patients with uterine carcinosarcoma were not included in this most recent trial; however, the use of HER2-targeted therapy in this context continues to be an area of active clinical inquiry with recent studies showing both in vivo and in vitro anti-tumor activity in ovarian and uterine carcinosarcomas with the use of HER2-targeted therapy.2,7,8
Case report
A 67-year-old woman presented with postmenopausal bleeding, abdominal pain, and >30 lbs weight loss. Initial computed tomography (CT) imaging of the abdomen and pelvis showed an enlarged uterus with a widened endometrial canal and enlarged retroperitoneal lymph nodes consistent with metastatic disease. A dilation and curettage was performed and showed endometrial carcinosarcoma. Subsequently, the patient underwent a total abdominal hysterectomy, bilateral salpingo-oophorectomy, omentectomy, and lymph node dissection. Gross examination was significant for a 5.8-cm polypoid mass occupying the endometrial cavity. Histologic evaluation confirmed the presence of endometrial carcinosarcoma with 86% myometrial invasion and extensive LVI within the myometrium. The tumor was composed of a predominantly carcinomatous component with high-grade cytology with solid, nested to glandular morphology, frequent mitoses, and P53 overexpression consistent with endometrial serous subtype. No precursor endometrial lesion was identified. A minor sarcomatous component was also present with a myxoid and cartilaginous appearance associated with pleomorphic cells supporting the diagnosis of a heterologous chondrosarcoma. Evaluation for staging showed extension into pelvic peritoneum, omentum, and three pelvic lymph nodes consistent with International Federation of Gynecology and Obstetrics (FIGO) stage (2018 FIGO Cancer Report) IVB or pTNM (AJCC, 8th Edition) pT3a N1a M1 disease.
Immunohistochemical (IHC) staining was performed on Ventana Ultra using the Ventana anti-Her2/neu (4B5) clone on histologic sections of the tumor according to the manufacturer’s protocols. Stained sections included carcinoma and sarcoma components, LVI of the myometrium, and pelvic lymph node metastasis (LNM). HER2 staining was evaluated using both the breast ASCO/CAP 2018 guidelines 9 and proposed ESC guidelines. 4 The percent of HER2 positivity was calculated per slide by determining the number of HER2 positive tumor cells out of the total number of tumor cells in the section, including surface tumor, invasive tumor, and tumor within LVI.
The surface endometrial tumor, including the carcinoma and sarcomatous components, had a HER2 staining score of 0, using both breast and endometrial guidelines (Figure 1) no tumor staining was seen. In contrast, staining performed on sections with myometrial invasion, myometrial LVI, and pelvic LNM (Figure 2) were positive. Using breast criteria, the cells were scored as 3+ with a range of positive staining from 30% in sections with extensive LVI to up to 80% in sections with prominent myometrial invasion (Figure 2(a) and (d)). Where positive, HER2 exhibited uniform intense, complete membrane staining. Applying the proposed uterine criteria, the cells were also considered 3+ with similar findings of 30%–80% staining demonstrating intense complete and basolateral/lateral staining.
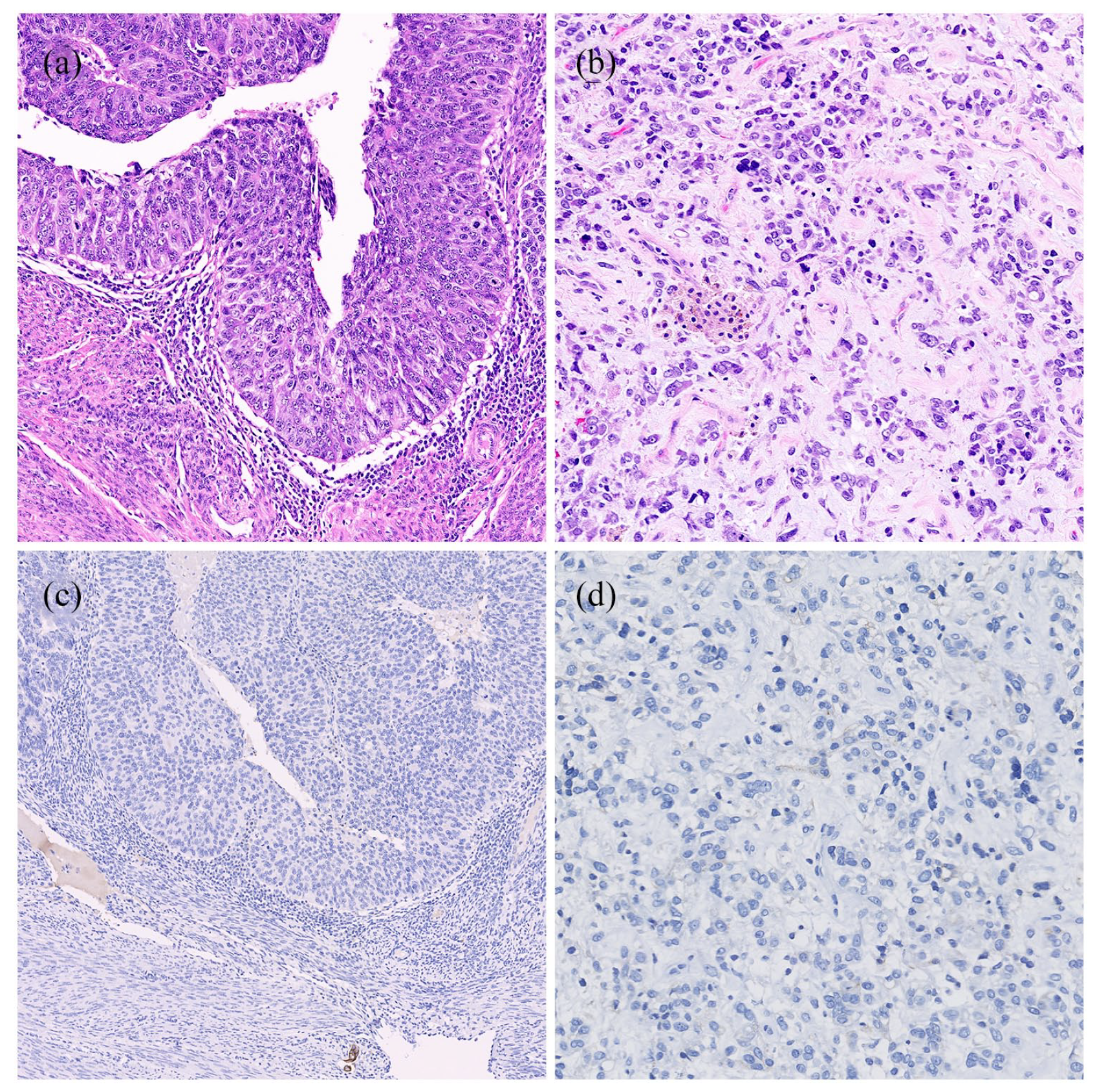
Uterine carcinosarcoma with (a) surface high-grade carcinoma component (20×) and (b) sarcoma component (20×). HER2 IHC negative in both surface carcinoma (c) (20×) and sarcoma component (d) (20×).
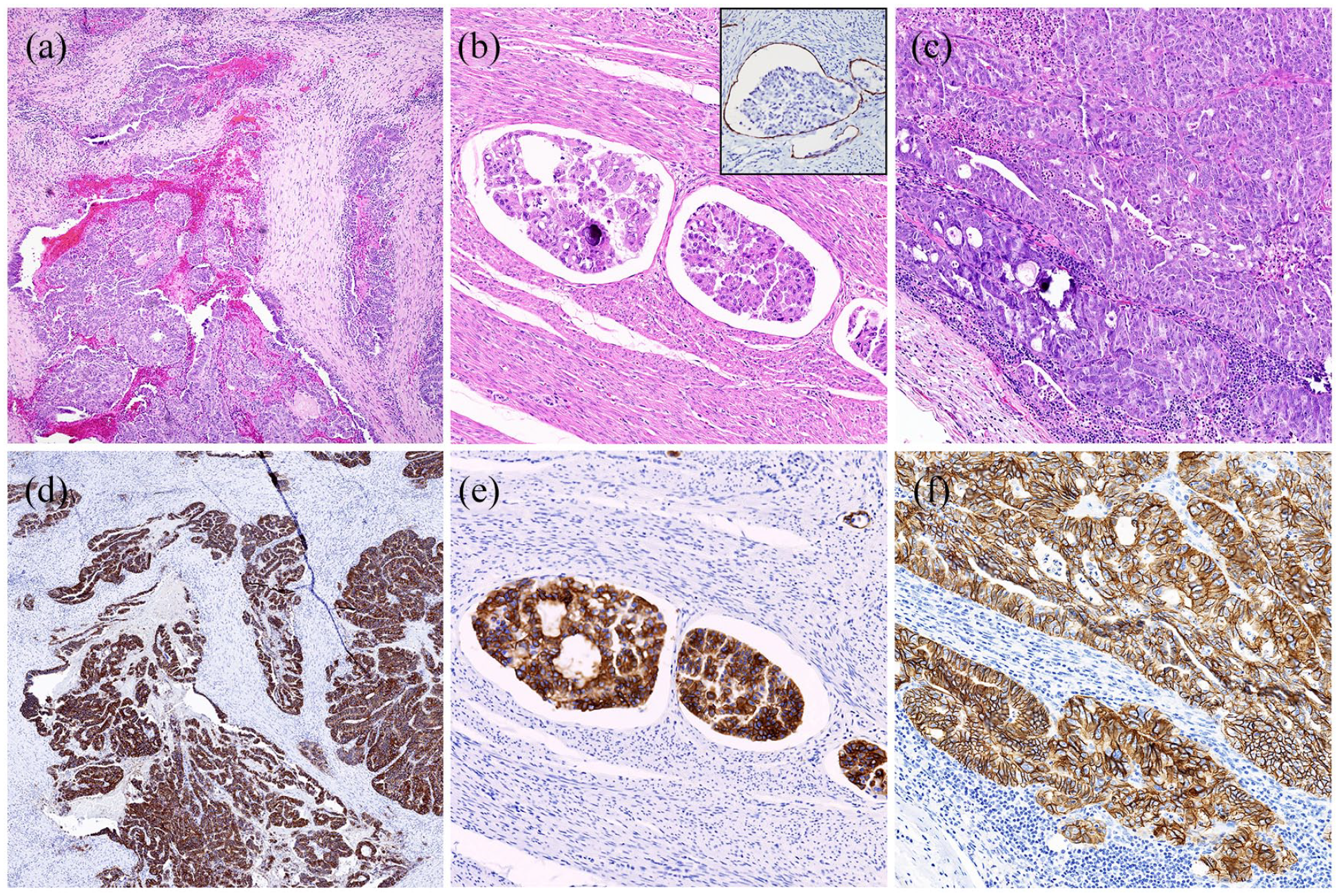
(a) Myoinvasive tumor (4×), (b) myometrial lymphovascular invasion (LVI) with prominent retraction artifact (10×), D2-40 IHC highlights LVI (inset 20×), and (c) pelvic lymph node metastasis (20×). HER2 IHC positive in myometrial invasion (d) (4×), myometrial LVI (e) (10×), and pelvic lymph node metastasis (f) (20×).
Confirmatory HER 2 fluorescence in situ hybridization (FISH) was performed on sections of the uterine tumor, including surface tumor, myometrial invasion and myometrial LVI (Figure 3) using a test probe localized to the ERBB2 locus (also called HER2), along with a control probe that is specific for the pericentromeric region of chromosome 17 (locus D17Z1) (Abbott Molecular; Abbott Park, IL, USA). Following hybridization, at least 60 interphase nuclei identified as having tumor-like morphology (area identified by the pathologist on hematoxylin and eosin) were evaluated for each probe. A ratio ⩾2, with an average of ⩾4 HER2 signals per cell, indicates amplification for this locus. A ratio <2, with an average number of <4 HER2 signals per cell, is categorized as negative for amplification. FISH demonstrated negative results in the endometrial tumor but amplification within myometrial LVI. Subsequent next generation sequencing (NGS) was performed on a representative section of the endometrial tumor (including surface tumor, myoinvasive tumor, and myometrial LVI) for identification of protentional additional therapeutic targets (Foundation Medicine, Cambridge, MA, USA) showed amplification of AKT2, AURKA, CCNE1, and TP53. Ultimately, these results did not provide any additional therapeutic options for patient management.

HER2 FISH (a) endometrial surface tumor with normal expression and (b) focus of myometrial lymphovascular invasion with signal amplification.
Post-surgery the patient completed six cycles of carboplatin and paclitaxel. CA-125 tumor marker showed a steady decline following surgery. Four months after completion of chemotherapy, she was noted to have an aortocaval recurrence of disease with a rising CA-125 but was asymptomatic. At this time, she was worked up for clinical trial consideration, however, due to intellectual disability and inability to consent or communicate symptoms, was unable to be enrolled. Multiple options for therapy were discussed with the patient and her power of attorney and they elected for single-agent trastuzumab given their primary goals for comfort, reduced visits, and toxicity. This therapy was offered as the lymphatic disease was the only site of disease. Her CA-125 at the start of therapy was 437 U/mL and fell to 405 with cycle 2 which was delayed a month due to poor patient compliance. She remains asymptomatic on therapy with no toxicity currently.
Discussion
Advanced endometrial cancer, especially high-risk histologies such as uterine carcinosarcomas, portend a poor prognosis for patients. In advanced or recurrent cases, treatment choice is directed at the epithelial component. 10 Recent data describe improved progression-free survival when trastuzumab is added to traditional platinum-based chemotherapy for advanced or recurrent ESC6,11 that overexpress HER2/neu at 2+ or 3+ by IHC and positive amplification by FISH. 11 HER2 expression rates in carcinosarcoma range from 17% to 43% with higher expression rates in the carcinomatous component (up to 56%) when compared to the sarcomatous component (6%).12–15
Compared with breast carcinomas, the pattern of HER2 expression by IHC in ESC has been described as having higher intratumoral heterogeneity 3 defined as at least 2 degrees of difference in staining intensity involving at least 5% of tumor cells. 4 In addition, the distribution of staining may differ in ESC samples with recently proposed criteria suggesting HER2 IHC score of 3+ should be given when >30% strong, complete, or basolateral/lateral staining is present.4,6 These findings support recent recommendations to submit large tumor sections when evaluating ESC for HER2 IHC. 4 Furthermore, when comparing HER2 in primary versus metastatic endometrial serous cancers in tissue microarrays, discrepant HER2 scores were observed in 23% of cases. 16 Typically, the primary tumor was HER2 positive and the metastasis was HER2 negative. 16 Furthermore, when selecting a HER2 assessment method, the intended therapeutic agent and its performance in clinical trials, rather than primary disease site (breast vs gynecologic tract), should also be considered.
Our case demonstrated that HER2 expression was restricted to the carcinomatous component (seen in myoinvasion, LVI, and LNM) but not the sarcomatous component, consistent with prior reports. A recent investigation into differences in primary versus metastatic lesions of carcinosarcoma was limited (five samples) but showed discrepant HER2 by FISH in only one ovarian carcinosarcoma case where the primary was negative and metastasis was positive. 2 While there is evidence of intratumoral heterogeneity in endometrial carcinoma, including ESC from biopsy to primary tumor with HER2 testing,17,18 there is no data to our knowledge quantifying HER2 expression heterogeneity in primary tumor versus recurrent tumor for uterine carcinosarcoma. Given known tumor heterogeneity, we expect that driver mutations can be explored independent from the initial tumor to guide clinical decisions. A recent proposal toward standardization of reporting of HER2 in carcinosarcomas suggests that as with ESC, large tissue sections be evaluated to account for tumor heterogeneity and if possible metastatic lesions also be included. 15 Scoring including lateral/basolateral membranous staining was also recommended. 15 To date, no large-scale studies have been conducted to evaluate HER2 expression specifically in LVI of carcinosarcoma.
Conclusion
In summary, differential staining of HER2 and FISH amplification of HER2 in this patient was seen in myometrial invasion, foci of myometrial LVI, and within LNM. Unlike prior reported findings in ESC, the present case was negative for HER2 staining in the primary surface tumor but positive in the invasive, metastatic, and LVI components. This finding suggests the inclusion of histologic sections with invasion, LVI, and LNM when evaluating HER2 expression in uterine carcinosarcoma, with reflex FISH testing when indicated, may be helpful to reduce the false-negative results and provide additional treatment opportunities for patients. In addition, our patient had a nodal-only recurrence which has a biomarker response to single-agent therapy supporting the importance of diligent testing and additional investigation for this amplification. Further studies need to be performed to determine if this finding is generalizable to carcinosarcoma and ESC samples.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from Virginia Commonwealth University Health IRB HM20012019.
Informed consent
The patient’s legal representative gave both verbal and written consent for this publication, specifically a case report with patient information. Pathology images were submitted only using existing tissue submitted for pathologic assessment. The clinical information has been deidentified.
