Abstract
Pyroglutamic acid is an endogenous organic acid and a metabolite in the γ-glutamyl cycle, involved in glutathione metabolism. Accumulation of pyroglutamic acid is a rare cause of high anion gap metabolic acidosis. There are multiple risk factors for pyroglutamic acid accumulation, such as chronic paracetamol use and sepsis. In this case report, we discuss how we came to this diagnosis, how it was subsequently managed and why it is an important consideration for critically ill patients with risk factors who are likely to end up in an intensive care setting. Pyroglutamic acid recognition and treatment could benefit patients in the critically ill population as pyroglutamic acid is a rare cause of high anion gap metabolic acidosis, which is likely under-recognised and easily treated. Inappropriate management of metabolic disorders can contribute to patient morbidity and mortality. Therefore, the recognition and appropriate management of pyroglutamic acidaemia could benefit patients with risk factors for its development in a critical care setting.
Introduction
Here, we discuss a case of a patient who developed pyroglutamic acidaemia and our management strategy, with reference to previous case reports. Pyroglutamic acid (PGA; called 5-oxoproline alternatively) is an endogenous organic acid and metabolite in the γ-glutamyl cycle, which is involved in glutathione metabolism and production. Accumulation of PGA is a rare cause of high anion gap metabolic acidosis (HAGMA). 1 However, there are various risk factors which have been previously established in the literature. 2 It is important to note that these risk factors do not completely explain the underlying pathogenesis of PGA accumulation, as the exact biochemical mechanisms underlying PGA accumulation are unknown. Most commonly, it is associated with glutathione depletion, 3 which indirectly leads to excess PGA production. Risk factors for glutathione depletion include chronic malnutrition, alcohol misuse, sepsis and paracetamol use. 3 Alternate risks derive from the reduced clearance of PGA. This is most commonly associated with the medications, such as vigabatrin 4 and flucloxacillin. 5 These inhibit the enzyme, 5-oxoprolinase, which breaks down PGA. Renal failure can also exacerbate PGA accumulation, as it is renally cleared. Rarely, it can be associated with inborn errors of metabolism related to the γ-glutamyl cycle. 6 As chronic paracetamol use is a risk factor for PGA accumulation 7 and paracetamol is a commonly used medication, especially in the management of pyrexia from sepsis, it is likely an underdiagnosed cause of HAGMA in patients with sepsis. Inappropriate management of metabolic disorders can be a contributing factor to patient morbidity and mortality, 8 which is important to recognise and easy to manage. Therefore, the recognition and appropriate management of pyroglutamic acidaemia could benefit patients with risk factors for its development.
Here, we report a case of a 54-year-old gentleman who presented with sepsis and HAGMA, of which PGA was suspected early in admission and his subsequent clinical workup and management.
Case presentation
A 54-year-old gentleman presented to the emergency department with a 5-day history of fevers, vomiting, non-productive cough, shortness of breath, lethargy and two episodes of collapsing while walking to the bathroom. He also complained of some left shoulder swelling and redness from a graze on his elbow he sustained from falling against a wall 2 weeks prior. His medical history consisted of chronic shoulder pain secondary to recurrent bilateral shoulder dislocations, a right humeral arthrodesis and alcohol misuse in the past. He took regular paracetamol 1 g QDS (four times daily) and ibuprofen 400 mg TDS (three times daily). He denied taking excessive doses of these.
On examination, he looked unwell and pale with Kussmaul breathing. His initial observations were a heart rate of 115, which was sinus rhythm, a blood pressure of 178/105, a respiratory rate of 32 with oxygen saturations of 98% on room air. He was afebrile on presentation. He had an elevated body mass index (BMI) of 30. The main clinical findings on exam were increased work of breathing, left renal angle tenderness, a warm erythematous and fluctuant swelling to his left elbow and left shoulder, and a purulent draining sinus in his left axilla. Interestingly, he did not complain of any elbow, shoulder or axillary pain and was unaware of the discharging sinus.
Initial blood investigations revealed a bedside glucose of 18.7 mmol/L and ketones of 0.5 mmol/L, an acute kidney injury (AKI) with urea of 10.1 mmol/L (3–8 mmol/L) and creatinine of 186 mmol/L (60–110 mmol/L). He also had a thrombocytosis and leukocytosis with a leftward shift, 862 × 109 (135–400 × 109/L) and 30.9 × 109 (4–11 × 109/L), respectively, and a CRP (C-reactive protein) of 424 mg/L (<5 mg/L). His liver function tests (LFTs) were deranged in a cholestatic pattern with an alkaline phosphatase (ALP) of 204 U/L (30–110 U/L) and GGT (gamma-glutamyl transferase) of 345 U/L (< 51 U/L). His ALT (alanine aminotransferase) and bilirubin were normal. This was associated with an elevated INR (international normalised ratio) of 1.9, which was likely multifactorial and possibly due to underlying sepsis and prolonged fasting from his anorexia. He also likely had some elements of underlying liver disease due to his past alcohol misuse, or non-alcoholic fatty liver disease from long-standing undiagnosed type 2 diabetes mellitus (T2DM). His initial venous blood gas (VBG) demonstrated a mixed metabolic acidosis, with the results as follows (see Table 1):
Venous/arterial blood gas values on admission and over the following 96 h.

The first VBG demonstrated a mixed HAGMA with a hyperchloremic metabolic acidosis with partial respiratory compensation. The anion gap was calculated to be 20 mmol/L using the formula Na+ + K+–(Cl− + HCO3−), with that only partially being explained by the lactate of 3.4 mmol/L which quickly resolved, with a serum albumin of 34 g/L and normal ketones. His urea was only mildly elevated and his AKI resolved quickly. The normal anion gap is 12 +/− 4 (8–16) mmol/L. When hypoalbuminaemia is present, the normal anion gap range is reduced 9 by 1 mmol/L; for every 4 g/L, the albumin is below the normal range (38–50 g/L). Using this, our patient’s initial normal anion gap would be 11 +/− 4 (7–15) mmol/L, as his serum albumin was 34 g/L. The hyperchloremic acidosis is demonstrated with a delta ratio of 0.42 or a decreased abbreviated strong ion difference 10 of 23. This was prior to the administration of intravenous (IV) fluids, such as normal saline. Interestingly, his serum potassium was low, despite the fact that he was acidaemic. Hypokalaemia has been shown to be a feature of PGA accumulation 11 for unknown reasons. Analgesic nephropathy-induced renal tubular acidosis was thought to be a contributing factor to the hyperchloremic acidosis as demonstrated later on urinary biochemistry, and given the history of chronic non-steroidal anti-inflammatory drug (NSAID) usage. Again, this could also be another contributing factor to the low potassium. The unexplained HAGMA was postulated to be related to the chronic paracetamol usage, sepsis and poor nutritional state and possibly underlying chronic hepatic impairment contributing to PGA accumulation. He denied ingestion of any other potential substances with a directed toxicology history. This was later confirmed on urinary biochemistry with a markedly elevated PGA level of 9 mmol/mmol of creatinine (normal range is < 0.1 mmol/mmol of creatinine). His suspected pyroglutamic acidosis was treated with IV n-acetylcysteine (NAC) prior to laboratory confirmation. In our case, we used the same dose of NAC that is used in acute paracetamol toxicity. The raised anion gap persisted and eventually resolved over time with NAC administration and improvements in renal function as you can see from the trend in the blood gases above. His hyperchloraemic acidosis was treated with IV sodium bicarbonate and liberal potassium supplementation. This also resolved and he eventually went on to develop a mild metabolic alkalosis which was likely due to iatrogenic sodium bicarbonate administration coinciding with the resolution of his metabolic acidosis. He was also newly diagnosed with T2DM, with an elevated haemoglobin A1c (HBa1c) of 10.9%. We suspect that he had peripheral neuropathy from his likely long-standing T2DM, explaining his lack of elbow or axillary pain on initial examination.
He was admitted to the high dependency unit for further management. Initial management consisted of IV fluid resuscitation, IV piperacillin/tazobactam, which was soon escalated to meropenem, vancomycin and lincomycin, the aforementioned sodium bicarbonate, potassium supplementation and NAC, and a peripherally inserted central catheter (PICC) was also inserted to facilitate the multiple infusions and predicted long-term administration of antibiotics. He had a computed tomography (CT) of his left upper limb and abdomen/pelvis, which demonstrated collections in the left elbow, biceps and shoulder and features of left-sided pyelonephritis. He was taken to theatre twice over the next couple of days by the plastic surgery team for incision, drainage and washout out of his multiple collections.
Later, the patient returned positive blood cultures demonstrating methicillin-sensitive staphylococcus aureus (MSSA) bacteraemia, with disseminated infection seeding multiple sites, including the cutaneous tissues of the left shoulder, left kidney, left inguinal lymph nodes and lungs, which were confirmed on a total body gallium scan. He had a transthoracic and transoesophageal echocardiogram which did not show any infectious valvulopathy, ruling out infective endocarditis. The initial inoculation site was thought to be his left elbow abrasion. He briefly required non-invasive ventilation (NIV) for type 1 respiratory failure, secondary to metastatic staphylococcal pneumonia. Ironically, his MSSA was flucloxacillin sensitive and the preferred agent to manage MSSA infection by our clinical microbiologists. However, given his susceptibility to PGA, he was preferentially treated with a prolonged course (6 weeks) of IV cefazolin, as flucloxacillin 5 is a risk factor for PGA accumulation. This demonstrates that the early recognition and suspicion of pyroglutamic acidosis played an important role in altering this patient’s ongoing management, and this prevented the potential recurrence of his PGA-related metabolic acidaemia. Namely, we made alterations to our management based on a high index of clinical suspicion. The PGA was only confirmed on a urinary specimen weeks later.
The patient spent a total of 4 weeks in hospital to manage his HAGMA, sepsis and AKI which all ultimately resolved. He received his last 2 weeks of IV antibiotics (IVABs) through home nursing services. He was commenced on metformin and gliclazide to manage his T2DM, with outpatient management through his general practitioner. He had one representation to hospital after discharge with concerns of recurrent wound infection. This was found to be delayed wound healing due to his underlying T2DM and was managed conservatively by the plastic surgery team.
Discussion
PGA is an endogenous organic acid that is a metabolite involved in the γ-glutamyl cycle. The γ-glutamyl cycle 12 is involved in the synthesis and metabolism of glutathione. Glutathione is a tripeptide consisting of cysteine, glutamic acid and glycine that plays a pivotal role in immunomodulation, amino acid transport and detoxification of free radicals in oxidative stress. 13 It brings amino acids intracellularly, replenishes glutathione and recycles it. The various biochemical processes involved in this cycle are demonstrated in Figure 1.
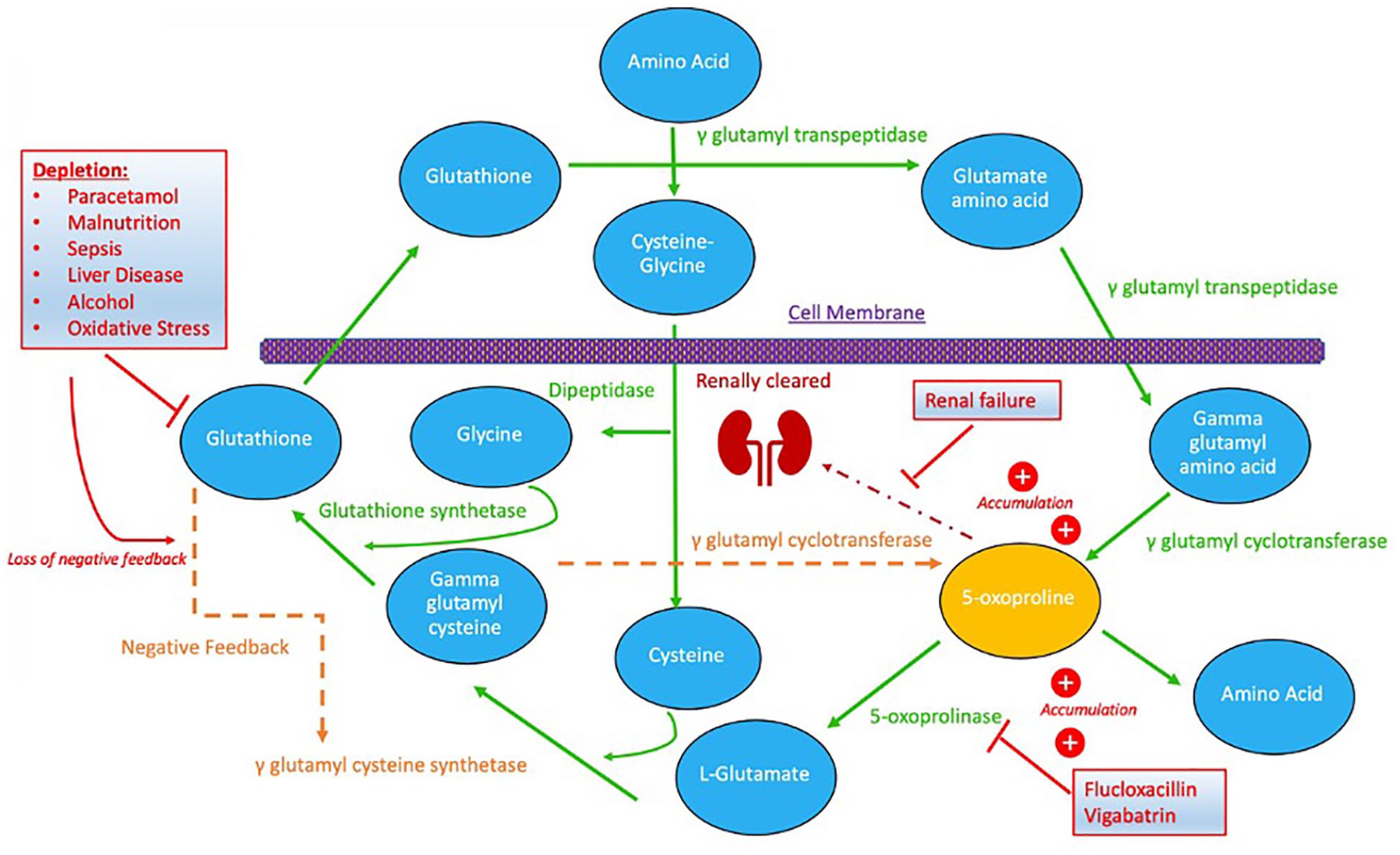
The γ-glutamyl cycle and its various biochemical processes. 12 Outlining processes which deplete glutathione and removes the negative feedback of γ-glutamyl cysteine synthetase, medications which inhibit 5-oxoprolinase and how PGA is renally cleared. All ultimately compounding to increase PGA levels.
Glutathione is synthesised by the enzymes glutathione synthetase and γ-glutamyl cysteine synthetase. The activity of γ-glutamyl cysteine synthetase is regulated by the negative feedback of increased glutathione levels. 14 Glutathione depletion results in a loss of negative feedback on γ-glutamyl cysteine synthetase, therefore potentiating its action and increasing γ-glutamyl cysteine production. This can then be converted into glutathione by glutathione synthetase. However, with high levels of γ-glutamyl cysteine, γ-glutamyl cyclotransferase converts it directly to PGA. PGA undergoes its own metabolic pathway, oxidised to glutamate by 5-oxoprolinase. This is a rate limiting step in this enzymatic pathway. 15 High PGA levels saturate 5-oxoprolinase, resulting in PGA accumulation. 16
For further reference, Hunter et al. 11 also provide a good explanation of the complex biochemistry discussed here.
There are multiple risk factors for the development of PGA accumulation, through two main biochemical mechanisms. Those associated with glutathione depletion or inhibition of 5-oxoprolinase. Most commonly, PGA accumulation is associated with the depletion of glutathione stores and loss of the negative feedback of γ-glutamyl cysteine synthetase. Risk factors for this include paracetamol use, sepsis, liver disease, alcoholism and malnutrition. 17 Alternate risks derive from the reduced clearance of PGA, which is associated with the medications vigabatrin 4 and flucloxacillin. 5 These inhibit the enzyme 5-oxoprolinase, which breaks down PGA. PGA is renally cleared 18 ; therefore, renal failure also plays a role in its accumulation. Rarely, it can be associated with inborn errors of metabolism related to the γ-glutamyl cycle. 6 Female sex is also a risk factor, possibly relating to differences in the enzymatic activity of the γ-glutamyl cycle between sexes. 19
In this case, our patient was chronically taking paracetamol at therapeutic quantities, had probable chronic hepatic disease from previous alcohol misuse, was acutely unwell with sepsis and had renal impairment. All combining to give rise to his pyroglutamic acidaemia.
The main steps in management for our patient’s PGA was addressing his underlying sepsis, including directed antimicrobial therapy with cefazolin rather than flucloxacillin, supportive care for his AKI and replenishing glutathione stores with an NAC infusion and avoidance/cessation of medications implicated in PGA, in this case flucloxacillin and paracetamol. There have been some case reports which demonstrated the resolution of HAGMA with NAC infusions. One such case report 20 demonstrated that a patient who had refractory HAGMA due to PGA accumulation had rapid resolution of their acidaemia when they were administered an NAC infusion, similar to our case.
In routine practice, many critically unwell patients who are admitted to the intensive care or high dependency unit with sepsis syndrome develop fever and are placed on regular paracetamol. 18 The 2015 ‘HEAT’ trial by Young et al. 21 demonstrated no difference in 28- and 90-day mortality in patients with sepsis whose fevers were managed with paracetamol versus placebo. This cohort of patients also have high incidence of AKI, chronic liver disease, malnutrition and may be on anti-staphylococcal antibiotics or anti-epileptic drugs. As a result, there may be many more critically ill patients than we realise who develop PGA acidaemia, which is likely undiagnosed. PGA accumulation can be confirmed on either serum or urinary biochemical studies. 22 As it is a cause of HAGMA that is simple to treat and has sequelae that may affect subsequent antibiotic and anti-epileptic drug choice, it is worth considering in patients with risk factors. It is also important to reiterate that when PGA accumulation is suspected early it should be treated prior to confirmation on laboratory testing. In our case, it took a couple of weeks for the initial urine sample to be processed and the results to be available, so we opted to treat our patient based on a high clinical suspicion.
A 2020 prospective cohort study by Gueta et al. 23 investigated whether there was an association between elevated PGA levels and in-hospital mortality rates in patients presenting to hospital with sepsis. In the multivariate analysis of this study, there was no association between elevated PGA levels and in-hospital mortality. It is important to note that the main limitation of this study was that there were only 100 patients included in this study. However, there were a couple of interesting findings from this study. First, it did show an association between elevated PGA levels and in-hospital mortality rates in the univariate analysis, but this was not statistically significant. Sub-analysis showed that patients who initially presented with sepsis and a qSofa score of < 1 (i.e. a lower illness severity) had higher in-hospital mortality rates associated with elevated PGA levels at presentation. Perhaps, this shows that elevated PGA levels may prognostically indicate a higher disease severity, prior to clinical signs of more severe sepsis, but obviously future studies with larger cohort numbers would be needed to investigate this. Interestingly, patients with lower haemoglobin, serum albumin and systolic blood pressure also had increased PGA levels, which suggest an association with higher illness severity.
A 2019 study 24 looking at oxidative stress due to glutathione depletion in patients with septic shock demonstrated that these patients had higher serum and urine 5-oxoproline levels. This study discussed how PGA accumulation is potentially an indirect marker of glutathione depletion and oxidative stress. Overwhelming sepsis causes indirect consumption of glutathione stores by detoxification of free radicals generated by oxidative stress. NAC has also been used as an adjuvant therapy in sepsis due to its role in inflammation and oxidative stress. 25 A 2003 study demonstrated reduction in inflammatory mediators in sepsis syndrome patients after administration of NAC. 26 A 2011 literature review also demonstrated that NAC plays an important role in free radical formation and the response to inflammation in sepsis. 27 There has been conflicting data behind the administration of NAC in sepsis, 28 and its role has not been fully established. However, it is worth considering in patients with PGA risk factors who have sepsis as it has been demonstrated to be beneficial in multiple case reports referenced previously in this report. Ultimately, more work needs to be conducted in the future to provide evidence relating to this.
Conclusion
In summary, pyroglutamic acidaemia is a rare, but easily treatable, cause of HAGMA which is likely underdiagnosed in the critically ill population. Therefore, recognising it early in patients could lead to improved patient care, which was the case with our patient. Poorly managed metabolic acidosis can be a contributing factor to mortality, and some recent studies suggest that PGA levels could potentially be an adverse prognostication; therefore, PGA acidaemia should be considered early while managing patients with any of the aforementioned risk factors, in particular sepsis, and treated empirically prior to potentially delayed laboratory confirmation.
Footnotes
Acknowledgements
The authors thank all the staff at Joondalup Health Campus intensive care unit for their help with this patient’s care.
Author contributions
All authors of this article contributed to the creation, design and review of this case report.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymised information to be published in this article.
