Abstract
In this study, we aimed to present a huge mandibular tumor. In this study, we presented a 29-year-old patient with mandibular tumor. Osteosarcoma is the most common malignant tumor of bone that occurs mostly in the young age group. It is most common in the second decade. The most important areas of bone involvement are the metaphysis of the load-bearing joints. Osteosarcoma is rarely seen in the head and neck region. The most affected area in the head and neck region is the mandible. No matter how large jaw tumors are, they can be operated on with an experienced surgeon and adequate repair.
Introduction
Sarcomas are malignant tumors originating from mesenchymal cells such as bone, cartilage, muscle, fat, and nerve. Osteosarcoma (OS) generally occurs in the long bones of adolescent patients in the skeletal system. In contrast, jaw OSs are most common in the third and fourth decades. After the femur, tibia, and humerus, the fourth most common location is the jaw bones. 1 It most commonly affects the mandible, maxilla, and calvarium in the head and neck region, respectively. 2 In the our case, the mentioned lesion occurred in an adult patient, which corroborates the majority of reports in the literature, which indicate the greater prevalence of the lesion in individuals in the first and second decade of life. 3 OS is a malignant neoplasm characterized by the formation of osteoid matrix by neoplastic cells. It is the most common primary malignant bone tumor accounting for 20% of all sarcomas, although its occurrence in the jaw is rare. It shows typical clinical behavior but varied radiological and histopathological features. 4 In this report, we presented the clinical, radiological, and pathological findings of a young male patient with severely enlarged OS in the mandible.
Case report
A 29-year-old male patient applied to us with the complaint of swelling on his face growing for 7 months. No surgical or medical treatment was given to the patient until he was admitted to us. In the examination, a lesion starting from the right mandible mentum and extending to the condyle and invading the floor of the mouth was observed. A preliminary diagnosis was made through anamnesis, physical examination, and imaging methods, and a definitive diagnosis was made after the operation. No biochemical examination was applied to the patient preoperatively, except for anesthesia preparation. A definitive diagnosis was made by pathological examination. No systemic disease was detected in the patient. No metastasis was detected in the patient (Figure 1).
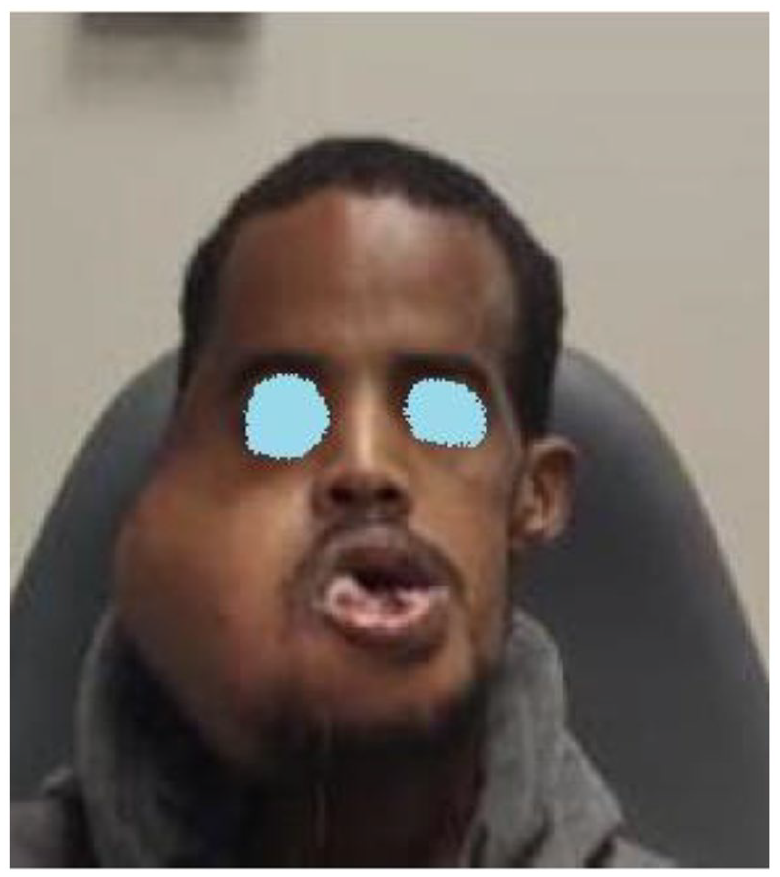
On physical examination, mass starting from the right mandible mentum and extending to the condyle.
In the neck, (CT) computed tomography imaging was observed that a heterogeneous central calcific mass lesion measuring 10 cm at its widest point invading the mandible and maxilla on the right half of the face, leading to destruction in the right zygomatic arch. This mass also extended to the level of the vascular space and continued up to the medial pterygopalatine fossa (Figure 2).

(a) CT axial section and (b) CT coronal section.
On neck magnetic resonance imaging (MRI), a heterogeneous mass lesion of 18 cm × 15 cm in size of mandibular origin was observed in the right face region. It had necrosis areas and partially occupied the maxillary sinus, partially involving the pterygopalatine fossa and muscles and extending superiorly to the temporal level, periorbital and intraorbital areas. In addition, it was observed that the mass extended to the base of the tongue and oral cavity, invading the oropharynx, nasopharynx, and partially inferiorly to the hypopharynx (Figure 3).

Neck MRI (magnetic resonance imaging).
In fluorodeoxyglucose (FDG) positron emission tomography (PET), there was an intense hypermetabolic mass lesion in the right half of the face, but no metastasis was detected in other parts of the body (Figure 4).
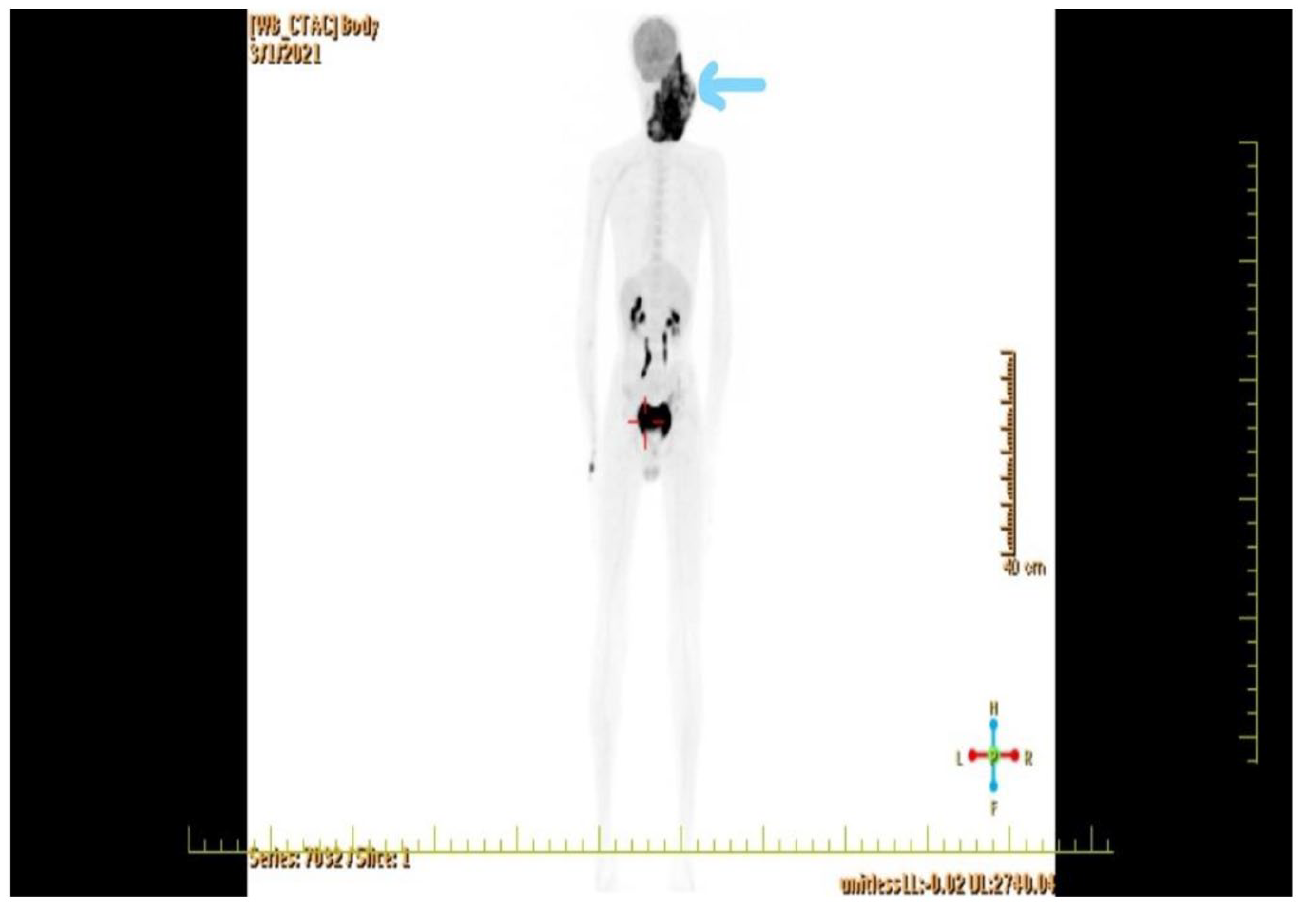
Positron emission tomography (PET).
Surgical procedure
Under general anesthesia, the Y incision extending from the mastoid type to the middle of the lower lip was extended to the clavicle. The subplatysmal flap was extended to the zygomatic arch and elevated. The mass was dissected from the tragus and reached to the tragal pointer. The mass was retracted from the tragus to the anterior area, the facial nerve trunk was found, and the nerve was sacrificed because the nerve was surrounded by the tumor 360 degrees. It was observed that the coronoid process and the zygomatic arch were destroyed. The temporomandibular joint was disarticulated from the glenoid fossa and the mass was released inferiorly. The mandible was cut from the left lateral part of the midline by performing a full-thickness osteotomy. The tumor had invaded the right part of the floor of the mouth up to the base of the tongue and anterior fold. The mass was lateralized by making an anterior to posterior incision on the floor of the mouth. Infero-lateral maxillectomy was performed due to tumor invasion in the postero-superior and tubel region of the maxilla. The tumor was resected en-bloc, together with the ramus and corpus of the mandible. The defect was repaired with a pectoralis major myocutaneous flap (Figure 5(a) and (b)).

(a) Tumoral lesion and (b) myocutaneous flap.
The patient received nutrition and wound care in our service for 2 months in the postoperative period. On histopathological examination, a high-grade malignant neoplasm producing osteoid matrix and showing extensive areas of chondroblastic differentiation was revealed. Neoplastic cells were high-grade in morphology by being possessed of hyperchromatic, pleomorphic large nuclei with increased nucleo-cytoplasmic ratio and eosinophilic cytoplasms. Mitotic activity was brisk (15 per 10 hpf) and necrosis was present (15% of the tumor). Pathological study of the lesion (Figure 6).

(a) Gross pathology photos of the surgical specimen, the cut-surface of the resected tumor. (b) Replacement of bone marrow by the tumor cells and the lace-like osteoid deposition. Note the tumor component with chondroblastic differentiation and the high-grade atypia, H&E, ×100. (c) Atypical osteoblasts with plasmacytoid morphology producing both osteoid and chondrogenic matrices, H&E, ×200. (d) Increased mitotic activity (arrows) and the high-grade atypia of the tumor cells, H&E, ×400.
Discussion
The clinical and biological behavior of long-bone and jaw OSs slightly differs. Head and neck OSs have a tendency to recure locally and frequency symptoms are swelling site of disease facial dysesthesia and loosening of the teeth. 5 There were clinical findings in our case, caused by a malignant lesion such as facial anesthesia, pain, and swelling. Although histological subtypes have no effect on treatment and prognosis in conventional OSs, they are important in the differential diagnosis of OSs. The differential diagnosis of OS includes benign lesions such as osteoblastoma, chondroblastoma, and fibrous dysplasia, as well as some other malignancies invading bone tissue such as chondrosarcoma and Ewing’s sarcoma. The presence of cellular atypia and surrounding tissue invasion is important in the differential diagnosis from benign lesions. In cases of OS containing chondroblastic areas, it is necessary to see the osteoid matrix structure in the tumor cells in order to make the differential diagnosis of chondrosarcoma. Chondrosarcoma, which is almost always seen in advanced ages, but OS shows bimodal age distribution. Chondrosarcoma of the jaw bones has been reported very rarely. 6 The age of our patient is compatible with the literature. Histologically, OS is divided into the central and peripheral subtypes. The main type of central OS is the conventional OS, which is represented by a wide spectrum of morphologies. Depending on the predominant type of extracellular matrix present, conventional OS is classified histopathologically in osteoblastic, chondroblastic, and fibroblastic subtypes. The osteoblastic subtype consists of osteoid or immature bone surrounded by haphazardly arranged fibroblast-like or epithelioid cells. The chondroblastic variant shows areas of atypical hyaline chondroid tissue. 7 In our case, high-grade conventional OS was detected. The tumor contained chondroblastic and osteoblastic areas. Difficulties may be encountered when diagnosing chondroblastic OS and can be mistaken as chondrosarcoma especially in the cases when the osteoid is absent in incisional biopsy and/or in unrepresentative tissue samples. Chondrosarcomas are relatively rare in jaw bones and show better prognosis than osteosarkoma. 8 Jaw osteosarcomas is a rare disease and the majority of them arise de novo, but some of them may develop in bone affected by Paget’s disease, fibrous dysplasia, trauma, viral infection, exposure to high-dose radiation, joint prostheses, and genetic syndromes such as Li-Fraumeni syndrome and hereditary retinoblastoma. 9 No specific risk factor was found in the history of our case. In a study by Paparella et al., 10 the histological subtype with the highest prevalence in jaw tumors is chondroblastic OS, followed by osteoblastic OS. In some studies, it has been shown that the osteoblastic pattern is more dominant, followed by the chondroblastic pattern. 11 There is no consensus on differentiation patterns in jaw tumors and more often it has heterogeneous histology. 12 Rare but important histological subtype of central jaw OS is the low-grade central osteosarcoma (LGCO). High-grade, grade 3 tumor was detected in our case. This is a well-differentiated OS consisting of spindle cell fibroblastic proliferation with low cellularity, no significant atypia, low mitotic figures, and a variable osteoid production. The most important feature of LGCO in long bones, and also in the jaw, is its similarities with benign lesions, first with fibrous dysplasia. 13 Yoshida et al. 14 reported that immunohistochemical analysis shows 100% sensitivity and 97.5% specificity for the diagnosis of low-grade OS. However, Tabareau-Delalande et al. 15 noted discordant results, with 33% of ossifying fibromas and 12% of fibrous dysplasias exhibiting MDM2 amplification by quantitative reverse transcription polymerase chain reaction (qRT-PCR) but no cases exhibiting MDM2 (Mouse double minute 2 homolog) overexpression by immuno-histochemistry. These investigators also showed amplification of an MDM2 neighbor, RASAL1 (RAS Protein Activator Like 1), in all the fibro-osseous lesions with MDM2 amplification but in none of the low-grade OSs studied. 15 We did not perform an immunohistochemical study in our case. After the anamnesis and physical examination, the imaging method should first be a direct X-ray. Later, whole body bone scintigraphy, MRI, computed tomography (CT), and PET may be requested. In scintigraphy, osteoblastic activity of the lesion on direct X-ray, skip metastases in the same bone, and bone metastases in the skeleton can be revealed. CT can be used to obtain detailed information about cortex invasion in OS. PET-CT is valuable in the treatment of OS in terms of evaluating the response to chemotherapy and showing whole body metastases. 16 CT scanning is rarely used as a diagnostic tool in patients with chondroblastoma. This imaging modality is usually reserved for more severe or recurrent tumors. CT scans can depict matrix mineralization, an extension of the tumor into the soft tissue, and erosion of cortical bone. Coronal and sagittal reconstructions can be used to assess the extension of the tumor across the epiphyseal plate into the metaphysis of the bone. 17 The diagnosis of OS can be made by fine needle aspiration biopsy and incisional biopsy after anamnesis, clinical examination, and imaging. 18 As a result of anamnesis, physical examination, and imaging methods, we operated on the patient and confirmed the diagnosis by performing an excisional biopsy. The first step in the treatment of OS is chemotherapy. After induction chemotherapy, the operation is completed with wide excision margins and negative surgical margins. 19 We applied the treatment plan as stated in the literature. OSs in the head and neck region have high morbidity and mortality due to the complex anatomy and difficulty of complete resection. The best treatment results can be achieved with neoadjuvant chemotherapy and adjuvant chemotherapy after surgical resection. The effectiveness of chemotherapy in the treatment of OS in the jaw bones is not clearly known. Although radiotherapy is recommended in inoperable cases, it is not effective in OS of the jaw bone. Elective neck dissection is not recommended because lymph node metastasis is rare. Tumor grade and surgical margin are the most important parameters in determining prognosis in OS. While survival reaches 75% in patients with a negative surgical margin, this rate regresses to 32% in the presence of residual tumor. Five-year survival is 74% for low-grade tumors but 42% for high-grade tumors. In the jaw bones, the anterior mandible has the best prognosis, while the anterior maxilla has the worst prognosis because of its difficult of resection. 20 The two main prognostic criteria of JOS (Osteosarcomas of the jaw) are tumor size and resectability at presentation. 21 In the high-grade OSs, the best curative option is represented by a multimodal treatment. However, the treatment of low-grade central and parosteal OSs can rely on surgery alone, provided a complete assessment of their metastatic potential. 22 Wide surgical resection is an effective treatment because it provides a clean surgical margin, a lower risk of local recurrence, and a higher overall survival. 23 Although adequate margins being the first goal of head and neck surgery in OS, it requires a careful balance between effective surgery and function-saving procedures. Surgical planning and the technical execution should be based on the expectation of performing a functionally effective reconstructive surgery. 24 In a study by Bertoni et al., 25 it was reported that poor results were probably due to inadequate surgery, the presence of positive surgical margins, and the inefficiency of surgery. Death is usually secondary to local tumor extension with neural and vascular infiltration. 26 Our case had lymphovascular invasion and a negative surgical margin. Preoperative and adjuvant chemotherapy has become the standard of care in long-bone OSs, and its role in jaw tumors is still controversial. 27 Excellent results have been obtained with the application of surgery, neo-adjuvant chemotherapy, and adjuvant chemotherapy. 28 In this study, following the surgical treatment, the patient received chemotherapy and radiotherapy. Adding CT or radiotherapy (RT) to surgery has demonstrated improved survival in locoregionally advanced head and neck cancer. The aim of chemotherapy is to reduce tumor size ameliorating surgical outcome, improve local control, and reduce distant metastases. RT is usually employed in the adjuvant setting and has the fundamental role of decreasing locoregional relapse. 29
Conclusion
In conclusion, the achievement of OS treatment is directly related to the grade and surgical margin of the tumor. In places such as the head and neck region, the morbidity and mortality of the operation result are high. However, no matter how large the tumor is in this region, the operation can be performed successfully with an experienced surgeon and adequate reconstruction.
Footnotes
Author contributions
The first author (A.B.), corresponding author (A.B.), and the coauthor (Ş.Ö., F.M.D., and S.T.) contributed to the writing and preparation of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were by the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article. Routine informed consent was obtained from the patient before the hospitalization and also informed consent from the patient was obtained in the study. There is no supported source that requires confirmation.
