Abstract
Thymoma-associated multiorgan autoimmunity disease can be seen in patients with thymomas and presents with features of graft versus host disease. Here, we report a case of a 52-year-old woman with a complex medical history including malignant thymoma, myasthenia gravis, Good’s syndrome, and T cell large granular lymphocytic leukemia who presented with a diffuse pruritic rash ultimately found to be compatible with a cutaneous presentation of thymoma-associated multiorgan autoimmunity disease. The eruption heralded the recurrence of the malignant thymoma and the rash was ultimately found to be resistant to a multitude of therapies except for oral corticosteroids.
Keywords
Introduction
Thymoma is often associated with autoimmune conditions including myasthenia gravis, pure red cell aplasia, and thymoma-associated multiorgan autoimmunity (TAMA). 1 TAMA has much similarity to graft versus host disease (GVHD) including both clinical features such as involvement of skin, liver, and intestine, and histological features such as interface dermatitis with apoptotic keratinocytes; however, TAMA is distinguished as occurring in patients with a thymoma. 2 Here, we present a patient with primarily cutaneous TAMA with a lichen planus-like and morphea-like presentation. The patient was ultimately found to have a recurrent metastatic thymoma and the cutaneous eruption was resistant to a multitude of therapies except for oral corticosteroids.
Case report
A 52-year-old female presented to the dermatology service with a generalized pruritic rash. The patient had a complex past medical history including malignant thymoma status post-resection and secondary myasthenia gravis and Good’s syndrome. She was also diagnosed with T cell large granular lymphocytic leukemia. The patient underwent multiple resections for the malignant thymoma and was subsequently diagnosed with Good’s syndrome, for which she received intravenous immunoglobulin (70 g subcutaneous once per month). Due to her immunocompromised state, she suffered recurrent pulmonary infections, which was managed with oral azithromycin prophylaxis.
Initial skin examination revealed generalized linear violaceous lichenoid plaques with excoriations. No oral or genital involvement was appreciated. Given the clinical presentation, a diagnosis of lichen planus was favored versus lichen planus-like GVHD in the setting of TAMA. A skin biopsy was performed which showed exocytosis of lymphocytes, dyskeratosis, and pigment incontinence (Figure 1(a)). The lymphocytic infiltrate was a mixture of CD8-positive and CD4-positive populations with the CD8-positive lymphocytes outnumbering the CD4-positive cells in the epidermis (Figure 1(b)). In some areas, the lymphocytes were closely associated with the dyskeratotic cells with a satellite cell necrosis-like appearance. Unfortunately, these histologic findings were unable to narrow down our clinical differential diagnosis.
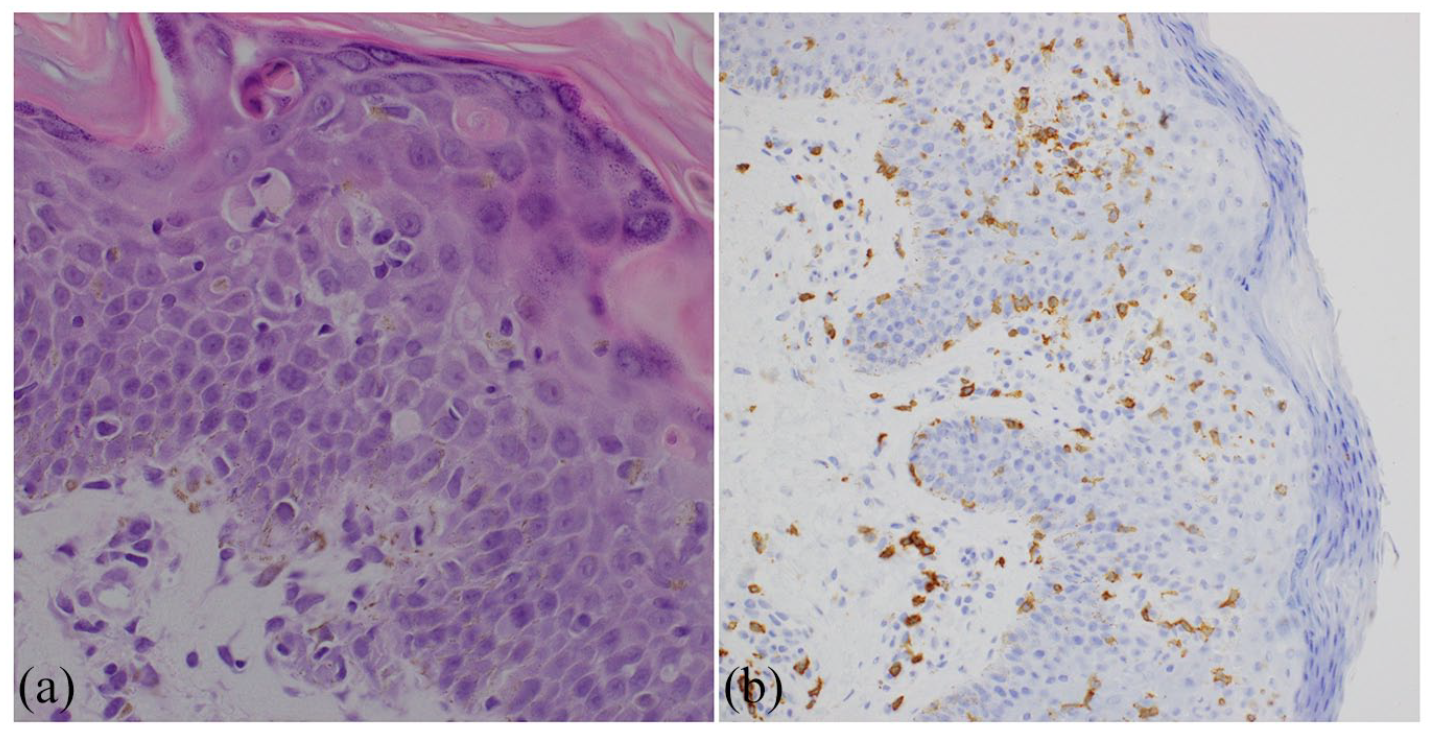
Skin biopsy showing exocytosis of lymphocytes, dyskeratosis, and pigment incontinence (a). The lymphocytic infiltrate was positive for CD8 (b).
Therapeutic attempts included topical steroids, topical tacrolimus (0.1%), topical glycolic acid (10%), oral methotrexate, azathioprine, acitretin, and narrowband UVB phototherapy were ultimately unsuccessful. Simultaneously, in attempt to control the patient’s severe pruritus, trials of hydroxyzine, cetirizine, doxepin, and naltrexone with escalating doses were tried with minimal improvement of her itch. The patient had temporary improvement with oral prednisone but due to her concomitant pulmonary infections and in conjunction with her respirologist and hematologist, oral prednisone was discontinued resulting in ultimate worsening of her cutaneous disease. The patient’s rash worsened to 90% of the patient’s body’s surface manifesting as lichenoid plaques with diffuse fibrosis and joint contractures (Figure 2).

On the upper back (a), upper chest (b), and abdominal area (c) were scaly annular hyperpigmented plaques with an erythematous border. Significant induration of the plaques was noted on the abdomen.
Given the patient’s worsening condition a biopsy was repeated, which similarly showed an interface pattern of inflammation. This time there was more prominent pigment incontinence with diffuse thickening of the collagen bundles in the dermis that extended into the subcutaneous septa (Figure 3). These changes can be seen in the sclerodermoid phase of chronic GVHD, lichen planus, or morphea in the setting TAMA. Given the patient’s past medical history of thymoma, the histopathologic findings of a sclerosing interface dermatitis, and the clinical presentation of diffuse indurated plaques, the most likely diagnosis was TAMA.

The subsequent skin biopsy demonstrated prominent pigment incontinence with diffuse thickening of the collagen bundles in the dermis.
Computed tomography (CT) angiography showed likely progression of thymoma with metastasis to pleural space and pericardium. Upon discussion with her surgical oncologist, further intervention was deferred given the risk of surgery and chemotherapy outweighed the benefits. The patient was actively followed by the oncology team to monitor for further progression. Given that TAMA is known to affect the gastrointestinal tract (enterocolitis) and the patient reported occasional right upper quadrant abdominal pain, a gastroscopy and colonoscopy was performed which showed candida gastritis but an otherwise normal colon. After treatment with oral antifungals, her gastrointestinal symptoms resolved.
Laboratory evaluation revealed a normal complete blood count, renal functions, and hepatic function. IgM and IgA were less than 0.05 g/L, and a normal IgG level was 11.89 g/L. Of note, there was no T cell clonality detected in the blood.
Discussion
TAMA is a paraneoplastic disease with clinical and pathological features resembling GVHD including involvement of the skin, liver, and gastrointestinal tract. Rarely, TAMA can affect only the skin as seen in our patient. 3 The disease was first named and described in 2007, by Wadhera et al. 2 Since then, few cases of TAMA have been documented and clinicopathological features are beginning to be elucidated.3–19 Here, we add to the literature by presenting a case of severe skin reaction in the setting of TAMA and to provide a brief review of these features.
It is first important to understand the role of the thymus in autoimmune disorders. Self-reactive lymphocytes are constantly produced but undergo central tolerance. For T cells, this occurs in the thymus where they undergo positive and negative selections. In the end, self-reactive T cells either undergo apoptosis, or become regulatory T cells, which suppress autoimmunity. Although not fully understood, it is suspected that a thymoma alters the physiology of the thymus consequently disrupting central tolerance, particularly negative selection. 8 Thymomas are associated with a variety of paraneoplastic and autoimmune diseases such as myasthenia gravis and rheumatoid arthritis. 1 It has been shown that the thymoma of patients with TAMA express decreased levels of autoimmune regulator genes (AIRE), which are responsible for the negative selection. 20 It is then hypothesized that self-reactive lymphocytes leave the thymus and attack the skin, liver, and colon in a manner that is almost histologically identical to GVHD. 20
TAMA features can present before or after thymoma is diagnosed. 8 Often TAMA is diagnosed years after initial diagnosis and resection of the tumor. 8 Usually, this is due to incomplete resection and tumor recurrence, as is the situation in this case report. 8 However, TAMA can occur years after resection of a thymoma, without recurrence of the thymoma itself. 8
Ideal treatment of TAMA is not well-established. Although often used for TAMA, systemic steroids have not proven to be effective in treating symptoms and predispose to infection. 8 Resection of the thymoma is thought to improve the course of the disease and should be preformed whenever possible. 8 In our patient, the location of the recurrent thymoma metastases posed a great surgical risk and active monitoring was opted. Whole body narrowband UVB phototherapy has also been proposed to be beneficial. 14
Our patient presented with a rare cutaneous only manifestation of TAMA, which was subsequently resistant to multiple therapies either than oral corticosteroids. Unfortunately, the patient ultimately contracted a viral illness resulting in respiratory failure. We hypothesize that her cutaneous disease could have potentially responded to plasmapheresis or antimalarials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The patient consent was obtained for the collection and publishing of the non-identifiable images and case details.
