Abstract
Coronavirus disease 2019 pneumonia in the newborn is a difficult-to-treat condition. Early clinical signs of pneumonia are nonspecific and present as respiratory distress of varying severity, and tachypnea is a predominant clinical sign. A 47-day-old, asymptomatic male newborn of coronavirus disease 2019 infected mother tested positive for coronavirus disease 2019 by reverse transcription polymerase chain reaction. During hospitalization, he developed progressive tachypnea, tachycardia, and chest radiography abnormalities, and was diagnosed as coronavirus disease 2019 pneumonia. He was treated with favipiravir, hydroxychloroquine, and lopinavir/ritonavir. A favipiravir-based regimen may be the drug of choice for coronavirus disease 2019 pneumonia in the newborn.
Keywords
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a novel coronavirus causing coronavirus disease 2019 (COVID-19), which emerged in Wuhan, Hubei Province, China, in mid-December 2019 to become pandemic disease.1,2 Adults may have asymptomatic to life-threatening disease, and older adults have greater risk of severe disease.3,4 Infection in infants aged under 1 year was recently reported, 5 and those without underlying diseases had mild COVID-19. 6 In Hubei, pediatric cases were treated with antiviral therapy (interferon, arbidol, oseltamivir, lopinavir/ritonavir (LPV/r), or empirical antibiotics), and a few critical cases also received invasive mechanical ventilation, systemic corticosteroids, and intravenous immunoglobulin. 7 No deaths were reported. 7 To date, no recommendations from randomized controlled trials (RCTs) have been published for any specific anti-SARS-CoV-2 treatment for patients with COVID-19. Favipiravir is a broad-spectrum oral antiviral agent with in vitro anti-SARS-CoV-2 activity. Data on its effectiveness and optimal dosage for COVID-19 are limited, especially in the newborn. 8
Case report
A 47-day-old, male newborn was asymptomatic but had household contact with his COVID-19 infected mother. He was hospitalized due to the household contact and tested positive for COVID-19 infection. He was transferred to the Bamrasnaradura Infectious Diseases Institute (BIDI) for isolation in negative pressure. He was the first child of consanguineous parents and was born by cesarean section with a birth weight of 2895 g. His mother had malaise, nausea, and vomiting. The newborn had no previous hospitalizations.
On admission, the newborn was alert, afebrile, and not tachypneic. His vital signs were a body temperature of 36.8°C, blood pressure of 120/70 mm Hg, pulse rate of 148 beats/min, a respiratory rate of 28 to 38 breaths/min, and a peripheral oxygen saturation of 99% on room air. The patient’s complete blood count results were a white blood cell count of 12,000/mL (lymphocytes 70%, neutrophil 25%), the neutrophil to lymphocyte ratio (NLR) of 0.36, hemoglobin of 10.7 g/dL, and platelet count of 326,000/mL.
On the day of admission, reverse transcription polymerase chain reaction (RT-PCR) for COVID-19 was positive, while a nasopharyngeal and throat swabs for influenza type A and B by rapid test, respiratory syncytial virus by RT-PCR, and hemoculture were all negative. Chest radiography showed no abnormalities (Figure 1). Azithromycin was given as empirical treatment.
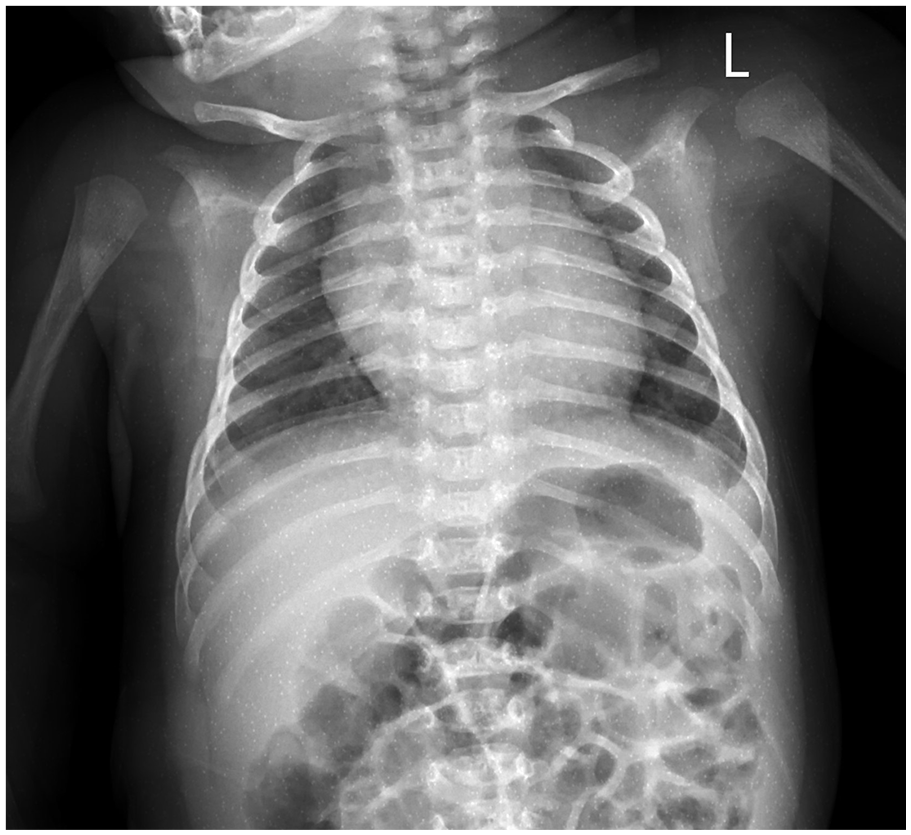
Day 1: The chest radiograph on admission shows no abnormalities.
On day 6 of admission, he was tachypneic (a respiratory rate of 40 breaths/min) and tachycardic, and his chest radiography showed abnormalities (Figure 2).

Day 6: The chest radiograph on day 6 of admission shows increased haziness with thickening of lung markings at both the perihilar regions and the lower lung fields.
According to the Thailand Ministry of Health guidelines for COVID-19, 9 a cocktail regimen of favipiravir (day 1: 30 mg/kg/dose BID, days 2–10: 10 mg/kg/dose BID), hydroxychloroquine (day 1: 10 mg/kg/dose BID, days 2–10: 6.5 mg/kg/dose BID), and LPV/r (days 1–10: 300/75 mg/m2/dose BID) were given for a total of 10 days.
On day 8 of admission, his clinical condition began to improve. On day 14, his chest radiography showed decreased haziness (Figure 3). On day 20 of admission, he was discharged from hospital in good general condition. He and his parents were quarantined and monitored for 14 days.

Day 14: The chest radiograph on day 14 of admission shows decreased haziness with thickening of the lung marking at both the perihilar regions and the lower lung fields.
Serial RT-PCR for COVID-19 was taken every 72 h. The results were positive on days 1, 4, and 7, and then negative on day 10 of quarantine and monitoring.
This study was reviewed and approved by the Institutional Review Board of the BIDI, Ministry of Public Health, Nonthaburi, Thailand. The reference approval letter codes are S019h/63_Exempt.
Discussion
Initial clinical studies revealed the promising therapeutic potential of several drugs, including favipiravir, a broad-spectrum antiviral drug that interferes with the viral replication, and hydroxychloroquine, a repurposed antimalarial drug that interferes with the virus endosomal entry pathway. 8 LPV/r is a pneumonia-associated adjuvant drug, which was used in the clinical treatment of patients with COVID-19, 10 and hydroxychloroquine showed good clinical outcomes among patients with COVID-19, which is thought to be due to the excellent efficiency of virus clearance after administration of this combination therapy. 11
The mechanism of actions of favipiravir is thought to relate to the selective inhibition of viral RNA-dependent RNA polymerase. 12 In February 2020, favipiravir was studied in China as an experimental treatment for COVID-19.13,14 A previous study showed it had a favorable safety profile regarding total and serious adverse events. However, safety concerns remain, including hyperuricemia, teratogenicity, and QTc prolongation. Favipiravir may be safe and tolerable for short-term use, but more evidence is needed to assess the effects of long-term treatment. 15
Chloroquine or hydroxychloroquine was reported to have potential broad-spectrum antiviral activity by increasing the endosomal pH required for virus/cell fusion and interfering with the glycosylation of cellular receptors of SARS-CoV.5,16 In early in vitro studies, chloroquine was found to block COVID-19 infection at low-micromolar concentrations, with a half-maximal effective concentration (EC50) of 1.13 µM and a half-cytotoxic concentration (CC50) greater than 100 µM. 17
Different radiographic patterns are seen as COVID-19 progresses. Typically, after the first to second week of the onset, lesions progress to bilateral diffuse pattern with consolidations. By contrast, both ground-glass opacification and consolidation were present relatively early in SARS. 18 Patients with COVID-19 developed dyspnea within a median (interquartile range (IQR)) of 5 days (1–10) after the onset of illness, while others did not develop any respiratory distress. 19 Pediatric patients with COVID-19 had a mild inflammatory infiltration, 20 which indicates that they are more likely to recover than adults after symptomatic treatment.
Conclusion
A favipiravir-based regimen may be the drug of choice for COVID-19 pneumonia in the newborn, safe and tolerable in short-term use, but more evidence is needed to assess the effects of long-term treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was reviewed and approved by the Institutional Review Board of the Bamrasnaradura Infectious Diseases Institute, Bamrasnaradura Infectious Diseases Institute, Ministry of Public Health, Nonthaburi 11000, Thailand. The reference approval letter codes are S019h/63_Exempt.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The patient’s mother provided written permission for publication of this case report and associated images. Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article (Thai language version).
