Abstract
The late type Ia endoleak after endovascular aortic repair could be a challenging issue. Over the last years, in case of short or enlarged neck, fenestrated and branched stent grafts have been increasingly employed with improving results. However, these devices have limited use in urgent/emergent cases as custom graft manufacturing takes long time, and may not be fit in patients with particular anatomic features. In this setting, chimney and relining remain an alternative but sometimes may not be adequate. According to literature, the use of the endoanchors associated to chimney technique can improve the procedure results in primary endovascular aortic repair. We treated two patients with a late type Ia endoleak after endovascular aortic repair with a simultaneous relining, single renal chimney, and endoanchors implant. These patients were valuated unfit for open repair with neck configuration unadapt for a simple relining, ballooning, or stenting. The patient conditions were unfavorable for an endovascular repair with branched endovascular aortic repair–fenestrated endovascular aortic repair. The same procedure was performed in both patients. Postoperative angio-computed tomographic scan demonstrated the resolution of the endoleak with patency of renal graft. Our preliminary experience, in these selected cases, demonstrate the feasibility of the technique in late type Ia endoleak.
Introduction
The late type Ia endoleak (IaEL) in endovascular aortic repair (EVAR) presents an incidence of 3.5%–11%, decreasing in recent years due to device and technique improving.1,2 It is validated that IaEL should be treated promptly, even though late and asymptomatic, with the aim of excluding the aneurysm from pressurized circulation. 3 An open conversion could be performed with acceptable results in patient fit for open repair (OR). However, most of the patients had already been considered unfit for open surgery. Landing zone extension is required with proximal cuff insertion in case of wrong sizing and deployment or distal graft migration. Other endovascular options include graft balloon dilation or insertion of a bare metal stent. 3
Over the last years, in case of short or enlarged neck, fenestrated and branched stent grafts have been increasingly employed with improving results. However, these devices have limited use in urgent/emergent cases as custom graft manufacturing takes long time, and may not be fit in patients with particular anatomic features. In this setting, chimney graft (CG) and relining remain an acceptable alternative to treat selected cases. 4 According to experiences shown in literature, the use of the endoanchors with chimney technique can improve the procedure results in primary EVAR, especially in distal migration risk cases.5,6 We show a proximal relining with mono renal chimney and simultaneous endoanchors implant in treatment of late IaEL in two cases.
Case report
Two male patients with a IaEL, previously undergone EVAR with Medtronic Endurant II bifurcated endograft implant, 5 and 7 years early, respectively, referred to our department.
The first patient was an 86-year-old man, regularly controlled in semestral ultrasound (US) follow-up, referred to our emergency room for abdominal pain. An US and computed tomographic (CT) scan showed an IaEL in a 64 mm aneurysm diameter (Figure 1). Associated comorbidities included a coronary artery disease, treated by coronary artery bypass grafting (CABG) and percutaneous transluminal coronary angioplasty (PTCA), severe mitral regurgitation complicated by permanent atrial fibrillation in anticoagulant oral therapy (AOC).
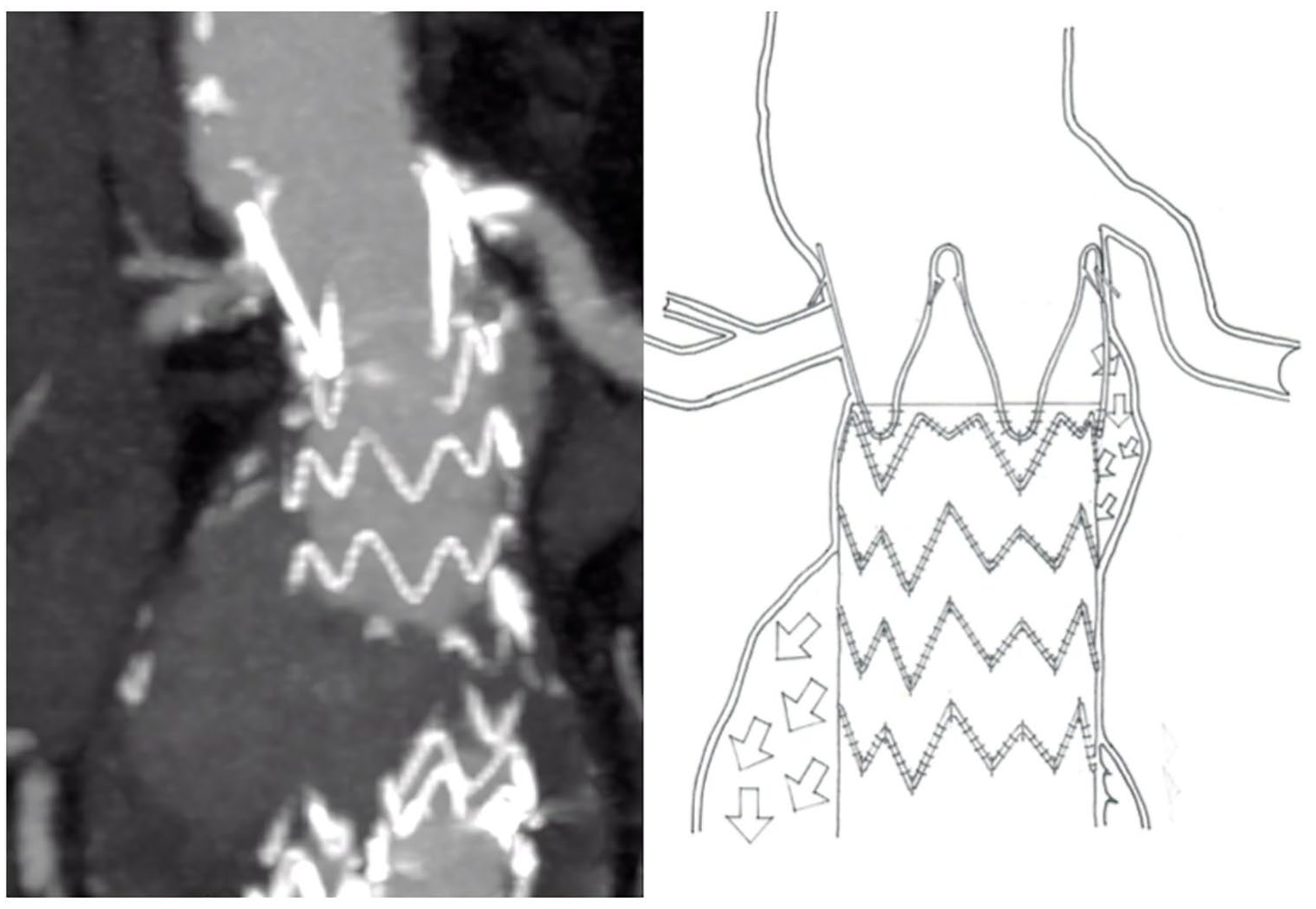
Preoperative angio-CT—drawing patient 1 showing the type 1a endoleak.
The second patient was a 79-year-old man, with associated comorbidities included obesity (BMI 32) moderate hypertension, diabetes mellitus, dyslipidemia, and OSAS treated by C-PAP machine at night and moderate chronic obstructive pulmonary disease; previous EVAR with embolization of the left hypogastric artery and following embolization of type II endoleak from lumbar arteries. A regular follow-up by US showed a suspect of IaEL with increase of the aortic diameter (55–61 mm). A followed CT scan revealed an IaEL with inflow close to left renal artery (Figure 4(a)), confirming the aortic diameter increase. In both cases, a graft distal migration and distal enlargment were detected.
The same procedure was performed in both patients.
Under local anesthesia, right surgical femoral access and percutaneous left brachial artery (LBA) were obtained. A preliminary angiogram confirmed the IaEL (Figure 2(a)). After systemic heparinization, selective catheterization of the distal renal artery was done by the brachial access, using Terumo Radifocus® Guidewire Stiff. After placing an Advanta V12 covered stent (Atrium, USA) in the renal artery, with controlled hypotension, an Endurant (Medtronic, USA) cuff was deployed just below the higher renal artery, supported by an Amplatz Stiff (Boston Scientific, USA) guidewire gently curved to optimize the deployment.

Intraoperative image: (a) preliminary angiogram; (b) post relining-chimney; (c) endoanchors implant; (d) final angiogram (red arrow: endoleak).
Afterward, the renal stent graft was delivered by overlapping few millimeters with the previous endograft. Simultaneously, ballooning of the aortic cuff was performed with compliant balloon (Reliant—Medtronic, USA). An angiogram check showed persistent endoleak, even though with flow reduction (Figure 2(b)). Therefore, implant of Heli-FX (Medtronic, USA) endoanchors was performed using multiple devices on inflow endoleak side (Figure 2(c)). The final angiogram showed the endoleak resolution (Figure 2(d)).
Postoperative course was uneventful, and the patients were both discharged on postoperative day 3.
Postoperative 6 months CT scan showed a complete exclusion of aneurysms and renal stent graft patency (Figures 3 and 4(b))

Postoperative angio-CT—drawing patient 1 scan showing the endoleak exclusion.
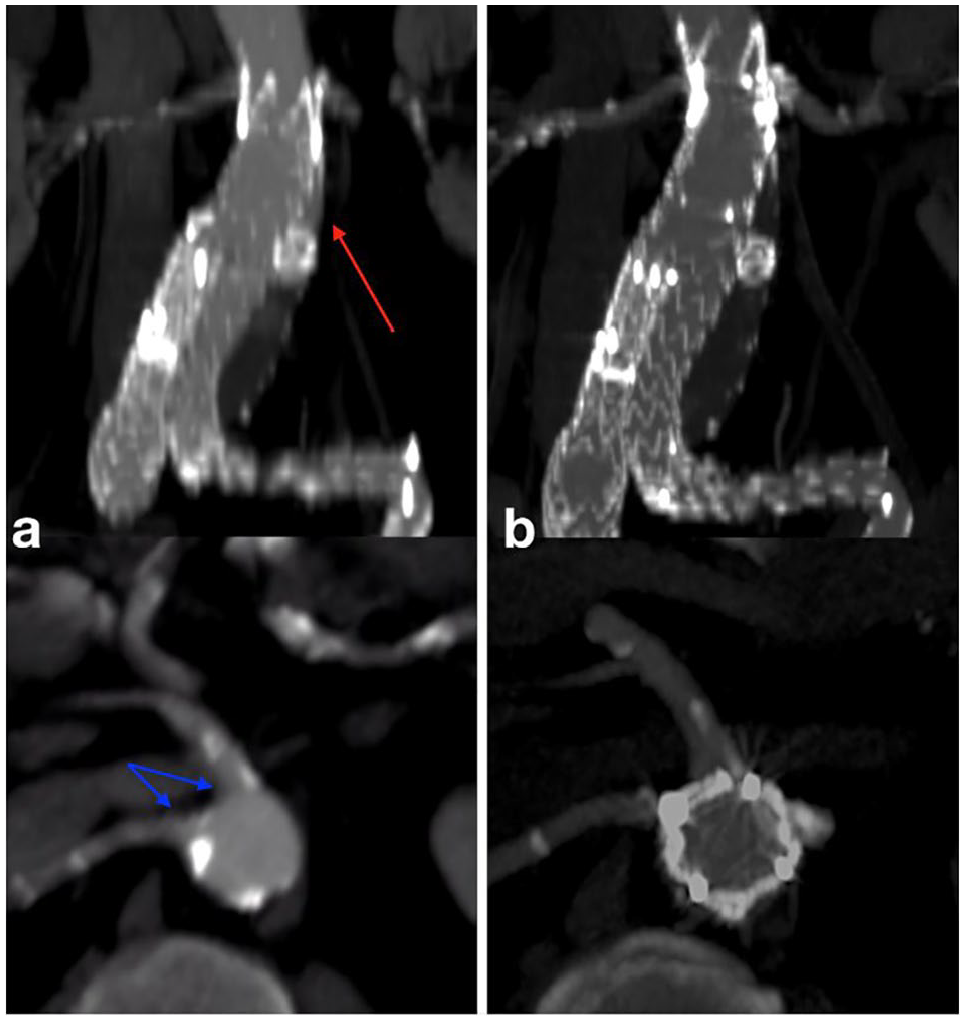
Preoperative (a) and postoperative (b) angio-CT patient 2 (red arrow: endoleak, blue arrows: closeness visceral vessels).
Discussion
Patients with IaEL could have complex aortic anatomy that limits their endovascular treatment options.
Moreover, many of these patients are not candidates for surgical repair, which poses risks for morbidity and death. In these reported cases, open surgery was excluded due to patient comorbidities.
We strongly agree, as supported in recent authoritative literature, 3 that in type I endoleaks, a fast procedure is recommended, especially in symptomatic case (patient 1).
Custom-made FEVAR and BEVAR could be a valid alternative, but all of these devices require a manufacturing time of 3–8 weeks, or even more, limiting their use in urgency setting. Some significant anatomical circumstances, regarding to different aspects of current available endografts, could limit FEVAR–BEVAR construction and utilization. 7
In our case (patient 2, Figure 4(a)), the closeness of target vessels could increase the complexity of the procedure, since four fenestrations would be necessary.
The aortic diameter and vessel disposition contraindicated a BEVAR off-the-shelf device implant.
We also considered a higher risk of spinal cord ischemia due to long aorta covering.
The neck was too short in both cases for a relining without a renal artery covering. For the above reason, it was necessary to cover the distal renal artery ostium landing immediately below the proximal renal artery.
In these selected patients, a simultaneous relining with single renal stenting was offered. Moreover the endoanchors implant was previously planned to avoid a new device migration and to improve the proximal sealing, specially in the stretch where the neck was enlarged. The angiogram check (Figure 2(b)), after aortic and renal graft deployment, confirmed that the relining and chimney were not enough for the leak resolution.
As recently Qamhavi et al. 8 showed, in an interesting review, the use of the endoanchors in secondary fixation is encouraged by technical success rate of 91.8% and interesting results in follow-up.
Other authors describe the endoachors use in primary ch-EVAR to close gutters, deploying them sorrounding visceral stents with techincal success. 9 These data supported our choice using this unique combined technique.
Conclusion
Our preliminary experience, in these selected cases, demonstrates the feasibility of the technique in IaEL.
In our experience, this technique was choosen to optimize the very short sealing below the proximal renal artery avoiding a higher operative risk.
We are well aware that a longer follow-up and more data are required to demonstrate safety and efficacy of this approach in cases where the other established approaches would be contraindicated.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
