Abstract
In rare cases, the celiac artery and the superior mesenteric artery arise from a common origin known as a common celiacomesenteric trunk. Celiac trunk stenosis or occlusion has been reported to accompany this anatomical aberrancy. Even rarer, are aneurysms associated with this common celiacomesenteric trunk. In general, visceral artery aneurysms are uncommon. We hereby present a 39-year-old female patient with a 1-month history of mild diffuse abdominal pain, with an incidental finding of superior mesenteric artery aneurysm on abdominal ultrasound. Subsequent contrast-enhanced computed tomography revealed severe narrowing of the celiac trunk and saccular aneurysmal dilatation of the superior mesenteric artery. Coil embolization of the aneurysm was performed, while maintaining persistent flow in the superior mesenteric artery and celiacomesenteric trunk. Visceral artery aneurysms are increasingly being identified incidentally with improvement in imaging techniques. The question lies whether to treat these aneurysms or observe. No universal guidelines exist regarding that matter, but the decision to intervene is made based on aneurysm location, size, and patient characteristics.
Keywords
Introduction
Mesenteric vascular structures are highly variable, as celiac and superior mesenteric arteries (SMAs) can present with anatomical variations. In roughly 1.5% of cases, the SMA originates from the celiac artery as a common celiacomesenteric trunk 1 (Figure 1).

(a) Upper figure: Embryology of normal vascular anatomy and variant celiac-SMA trunk. In the primitive vasculature, the 10th to 13th vitelline arteries connect the aorta with a ventral longitudinal anastomosis. In a normal anatomy, the 10th and 13th persist to individually form the celiac axis and superior mesenteric artery; the remaining segments regress before birth. If the 10th to 12th vitelline arteries regress but there is abnormal persistence of ventral anastomosis, a celiacomesenteric trunk occurs 5 and (b) lower figure: stenosis of the celiac trunk arising from the CMT, with aneurysm in the SMA. Note the retrograde flow through the inferior pancreaticoduodenal artery (PDA), which functions like a collateral as compensation for the stenosed celiac axis.
Representing an incidence rate of just over 0.1%–2% in the general population, visceral artery aneurysms (VAAs) are rare. 2 Although rare, mortality still ranges from 10% to 90% after rupture depending on the location and size of the aneurysm.2,3 Most VAAs are splenic artery aneurysms (SAAs) accounting for 60%, while superior mesenteric artery aneurysms (SMAAs) and celiac artery aneurysms (CAAs) represent 5% and 4%, respectively, of VAAs. 2 Aneurysms involving the celiacomesenteric trunk are even rarer with only 20 cases having been reported in literature. 4
In this case study, we present a 39-year-old female with an incidental finding on pelvic ultrasound (US) of an SMAA with further investigations demonstrating a CMT.
Case description
A 39-year-old female, previously healthy, presented initially for diffuse abdominal pain associated with menorrhagia of 1-month duration. Pelvic US was initially obtained as part of the regular workup with findings consistent with multiple endometrial polyps and an incidental finding suspicious of SMAA.
This prompted further evaluation with computed tomography angiography (CTA) scan of the chest, abdomen, and pelvis, which demonstrated celiacomesenteric trunk with severe narrowing of the celiac trunk, and saccular aneurysmal dilatation of the SMA measuring up to 1.3 cm in the anteroposterior (AP) diameter, 1.5 cm in the transverse axis, and 1.8 cm in height (Figures 2–4). In addition, there was compression of the left renal vein between the aorta and the SMA raising the possibility of renal Nutcracker syndrome.

CT angiogram of the abdomen, axial cut, demonstrating the aorta (blue arrow), the CMT (green arrow), and celiac trunk stenosis (black arrow). The left gastric artery (red arrow) and hepatic artery (yellow arrow) are also seen.
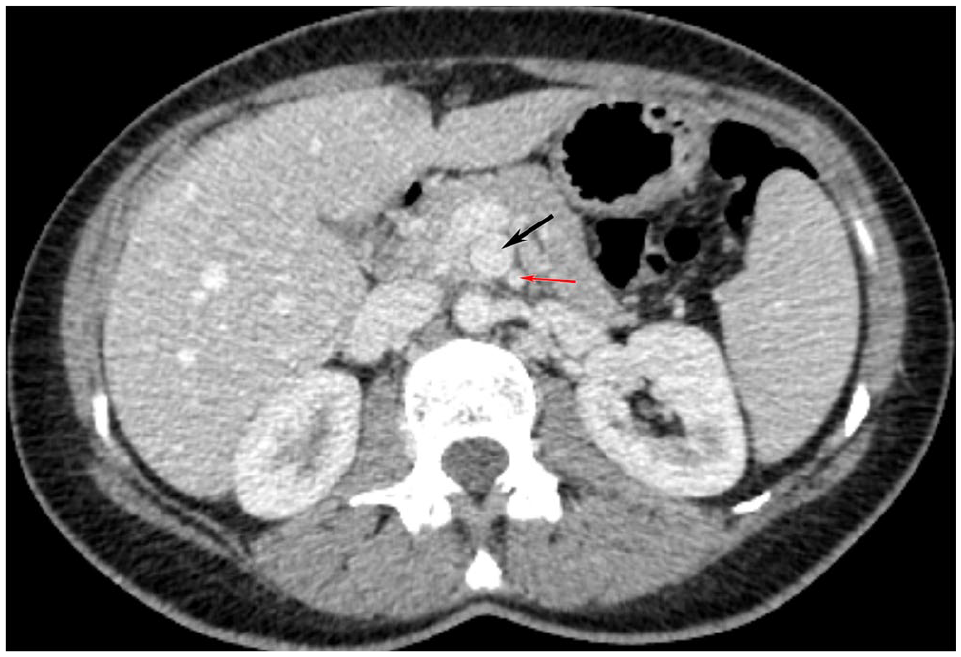
CT angiogram of the abdomen, axial cut, demonstrating superior mesenteric artery aneurysm (black arrow) and the superior mesenteric artery (red arrow).

3D reconstruction from CT angiography of the chest, abdomen, and pelvis, demonstrating aneurysm in the superior mesenteric artery (black arrow). Black circle shows the CMT. The splenic artery is highlighted by the yellow arrow, and the hepatic artery, by the red arrow. The SMA is highlighted by the gray arrow, and the green arrow shows the enlarged inferior pancreaticoduodenal artery.
No evidence of rupture was found, and the combination of abdominal pain and abnormal uterine bleeding was attributed to the endometrial polyps, with subsequent polypectomy performed and symptom resolution.
The decision to intervene in the aneurysm was related to the patient’s age and high probability of future pregnancy.
Considering the elective nature of the intervention, and the hemodynamic stability of the patient, endovascular rather than surgical approach was chosen.
Selective superior mesenteric and celiacomesenteric arteriograms, with coil embolization of SMAA, were done.
The right femoral access was obtained. Using the right-sided approach, a 5-French Cobra catheter was advanced over a 0.035 angled Glidewire and was used to attempt cannulation of the SMA. Multiple attempts were done using the tip of the Cobra catheter phase, but it was unsuccessful. The Cobra catheter was exchanged for a 5-French SOS 2 catheter which was successfully used to cannulate the SMA. Contrast injection revealed an approximately 2.5 × 2.0 cm saccular aneurysm arising from the mesenteric aspect of the celiacomesenteric trunk. Large pancreaticoduodenal artery was noted to feed the celiac axis distribution in a reverse manner (Video 1 in Supplemental Material).
A 1.7-French microcatheter was advanced in a coaxial manner through the sustentaculum over a 0.014 microwire. Multiple attempts at cannulating the aneurysm neck were finally successful in entering the aneurysm sac. Subsequently, the aneurysm sac was embolized with two 22-mm/65-cm Cosmos microcoils until the neck of the aneurysm was completely sealed (Figure 5, Video 2 in Supplemental Material). Follow-up CTA of the abdomen and pelvis revealed no residual filling of the aneurysm sac with persistent flow in the SMA, pancreaticoduodenal artery, and celiacomesenteric trunk.

Aneurysm sac embolized with two 22-mm/65-cm Cosmos microcoils with complete seal of the neck of the aneurysm. Notice the enlarged inferior pancreaticoduodenal artery (white arrow).
Discussion
Most commonly, SMA involvement presents with abdominal pain, with around 50% of SMAAs being diagnosed in the emergency department due to manifestations of spontaneous rupture, with a mortality rate of 30%. 6
Around one third of SMAAs are caused by mycotic embolization from nonhemolytic streptococci, staphylococci, and gram-negative bacteria. 6 Other causes include atherosclerosis, pancreatitis, and trauma. 7
In general, specific criteria exist concerning when to initiate treatment of VAAs, but in the case of pancreaticoduodenal artery aneurysms, intervention is always recommended due to high risk of rupture. 3 Both open surgical and endovascular techniques exist as treatment modalities, with endovascular coiling the preferable approach for SMAAs. 8
During the fourth week of gestation, visceral arteries begin development. Variations in the origin of the celiac artery and SMAs are therefore attributed to disturbances in the normal embryologic processes. In rare cases, the celiac artery and SMAs may arise from a common trunk of the abdominal aorta, a condition known as a CMT which occurs in less than 1% of all anomalies involving the celiac axis. 9
As medical imaging technologies evolve, and as imaging studies are increasingly utilized, a growing number of VAAs are detected before their rupture. These imaging studies primarily include duplex ultrasound (DUS), and CTA among others. 2 These splanchnic artery aneurysms are classified according to their anatomical locations, with 60% found in the splenic artery and 5.5% in the SMA. 10 Aneurysms involving the CMT are even rarer. 11 Only around 0.09%–2.00% of the general population present with true aneurysms,1,12 and depending on the location of the aneurysm, they are associated with a mortality rate of 25%–70%. 12 Importantly, no correlation has been found between the size of the VAA and its risk of rupture; therefore, rupture can occur regardless of the size. 13
Celiac artery occlusion or stenosis has been identified in 12.5%–49% of all individuals who have undergone abdominal angiography, 14 a finding that is not clinically significant by itself due to the collateral pathways that develop from the SMA via the inferior pancreaticoduodenal artery to provide retrograde flow through the gastroduodenal artery (GDA) to the liver. 14 This increased flow can potentially lead to the formation of a Gastroduodenal artery aneurysm (GDAA). 11
Clinical presentation
Most VAAs are asymptomatic. In case where symptoms do occur, the most frequent presentation is abdominal pain. Symptomatic presentation depends on three factors: size, location, and etiology. When rupture occurs, symptoms of hemorrhagic shock occur. 15 Fever and abdominal pain are seen with mycotic aneurysms.11,14
Symptoms associated with SMAAs are abdominal pain, nausea, vomiting, and even weight loss. 15
Diagnosis
Most VAAs are found incidentally on imaging, most commonly on abdominal US or x-ray. 15
In symptomatic patients, especially those with a pulsatile abdominal mass, prompt evaluation with either noninvasive imaging studies such as DUS, CTA or magnetic resonance angiography (MRA), or contrast arteriography should be done. 2
Signs found on US include a dilated artery and thickened wall with the possible presence of atherosclerotic plaque or thrombus. In addition to its convenience and noninvasiveness, ultrasonography also enables morphological assessment of the wall and sac of the aneurysm and the hemodynamic evaluation of blood flow in their region.1,7
When considering treatment of an incidentally found VAA, imaging through CTA or MRA should be obtained for proper planning.6,8 Cross-sectional imaging obtained with CT can help localize the aneurysm and determine its size and shape along with its wall characteristics. Importantly, it also helps evaluate collateral flow. 2
Management
Per the German Society for Vascular Surgery, indications for interventions for VAAs are asymptomatic VAAs >2 cm in size, increase in size ⩾0.5 cm/year, symptomatic and ruptured VAAs, and pseudoaneurysms. 15
In contrast, pancreaticoduodenal aneurysms are prone to rupture and should be treated at any size. 3
Similarly, the European Society of Vascular Surgery (ESVS) recommends observation of aneurysms <25 mm in size with imaging at a 3-year interval and treatment of symptomatic aneurysms. 16
Considering the high mortality rate observed in cases of SAA rupture, 70% and 90% in pregnant women and fetuses, respectively, the ESVS also recommends intervention in women of childbearing age with VAAs. 16
Management of SMAAs includes surgical ligation, endovascular embolization, and stent graft placement. The selection of open or endovascular approach is based on patient characteristics, aneurysm location and size, and expertise. 16 There are no randomized trials or prospective studies comparing open and endovascular repairs. 16
In light of its minimal invasiveness, endovascular approach has become the predominant treatment modality over open surgical techniques. 6 Most commonly, the right femoral access is obtained and coiling of the aneurysm is done to induce thrombosis, along with stenting to exclude the aneurysm.4,6
Although endovascular approach includes either coil embolization or stenting, coil embolization is a more used technique as it has shown promising success rates when used and has a wider applicability.2,3,16
Although percutaneous transcatheter coil embolization techniques have shown a success rate ranging from 96% to 98%,1,2,8 endovascular approach can still present with its own complications that include visceral ischemia caused by sacrifice of the involved visceral artery, coil migration with resultant embolization of nontreated artery, end-organ thrombosis, vessel recanalization, persistence of the aneurysm, stent thrombosis, or restenosis. 2
Conclusion
Although rare, VAAs carry a significant mortality rate of 10%–90% after rupture depending on the size and location. Their detection has become increasingly prevalent with the advance of imaging techniques and are frequently identified incidentally rather than through symptomatic presentations. Asymptomatic cases with an aneurysm diameter of less than 25 mm are usually monitored, whereas symptomatic cases or cases involving pregnant patients are treated. Intervention is mainly through endovascular techniques in hemodynamically stable patients, with coiling the preferred method due to its high success rate and widespread availability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval is not required by our institution for case reports or case series
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article
Supplemental material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
