Abstract
An 83-year-old Japanese man with Alzheimer’s disease was admitted to our hospital for treatment of hyponatremia resulting from water intoxication. During hospitalization, the patient developed focal impaired awareness seizures, focal to bilateral tonic-clonic seizures, and subsequent status epilepticus. Electroencephalogram during focal impaired awareness seizures showed rhythmic 5–9 Hz theta activity in the right frontotemporal region. Electroencephalogram during focal to bilateral tonic-clonic seizures showed bilateral polyspikes. Electroencephalogram during an interseizure period revealed sharp waves in the right frontal region. Continuous intravenous administration of midazolam was the only effective treatment for status epilepticus. The patient died of aspiration pneumonia on day 58. Hyponatremia-associated status epilepticus is rare; in the present case, multifocal epileptogenicity resulting from Alzheimer’s disease and hyponatremia-associated elevation of glutamate levels in the synaptic cleft may have contributed to the onset of focal to bilateral tonic-clonic seizures with subsequent status epilepticus.
Keywords
Introduction
The incidence of new-onset epilepsy in the elderly population is increasing; nearly 25% of new-onset seizures occur in patients aged over 65 years and the incidence in this population is twice as high as that in children. 1 According to a previous study, 1% of elderly individuals have epilepsy. 2 Regardless of the presence of precipitating factors, elderly people are more prone to develop seizures than younger people and elderly patients with epilepsy have two to three times higher mortality than the general population. 2 Moreover, 30% of acute seizures in elderly people may present as status epilepticus (SE), with a mortality rate of nearly 40%. 2 Most cases of new-onset epilepsy in elderly individuals are secondary to cerebrovascular disease, metabolic disturbances, dementia, traumatic brain injury, tumor, or pharmacotherapy. Seizures occur in approximately 10% of patients with Alzheimer’s disease (AD); two major seizure subtypes observed in this population are focal to bilateral tonic-clonic seizures (FTBTCS) and focal impaired awareness seizures (FIAS).2,3 However, FIAS in patients with AD can occur without physical movement, presenting instead as transient increased confusion. Therefore, the incidence of FIAS without physical movement may be underestimated.2–4
We herein present a case of refractory SE that began with epileptic seizures during correction of hyponatremia in a patient diagnosed with AD 2 years before admission.
Case presentation
An 83-year-old man was admitted to our hospital for evaluation of general malaise. He had a 10-year history of chronic atrial fibrillation and hypertension and had been treated with candesartan (8 mg/day) and warfarin (2 mg/day). Two weeks before presentation, the patient had visited a urologist because of transient urinary tract hemorrhage. Detailed examination had shown no abnormalities of the urinary tract. He had been advised to drink enough water to prevent urinary obstruction by blood clots for 1–2 weeks. In response, the patient started drinking about 4 L of water per day. One week later, he became aware of fatigability and gait disturbance but continued drinking extra water. His symptoms gradually worsened until he was admitted to the general medicine department of our hospital. The patient had no relevant medical or family history. He was a non-smoker and non-drinker.
On admission, the patient’s weight was 58 kg and his blood pressure was 102/72 mmHg. He was alert and afebrile. The patient complained of gait disturbance, but physical and neurological examination showed no abnormalities. Electrocardiography showed atrial fibrillation. Plain computed tomography of the neck, chest, and abdomen showed no significant abnormalities. Notably, laboratory tests showed a markedly decreased serum sodium level (105 mEq/L, normal: 138–145 mEq/L). Plasma and urine osmolality were 225 and 140 mOsm, respectively. Urine sodium level was undetectable. Levels of plasma adrenocorticotropic hormone, cortisol, and antidiuretic hormone were normal, suggesting the possibility of water intoxication. The patient received 0.5 L of normal saline solution in the emergency department and was transferred to a general medicine bed 4 h after emergency department presentation. His serum sodium level increased by 4 mEq/L over 4 h. Administration of 5% glucose solution was started at a rate matching urine output to prevent a further increase in serum sodium level. On day 2, the patient’s serum sodium level rose to 113 mEq/L. His serum sodium level on day 3 was 121 mEq/L. Thereafter his serum sodium level increased by 5 mEq/L per day.
On day 5, the patient’s serum sodium level was 130 mEq/L. He was alert and was improving. However, he suddenly developed twitching of the left arm lasting for 30 s, consistent with FIAS, followed by FTBTCS lasting for about 40 min. Assisted ventilation was started; intravenous diazepam (10 mg) was administered twice but was ineffective. Therefore, intravenous fosphenytoin sodium (1300 mg) was subsequently administered. FTBTCS stopped after administration of the first dose of fosphenytoin. However, intermittent twitching of the left arm with impaired awareness resulting from FIAS continued several times per hour, with each episode lasting more than 5 min. Moreover, the patient had five episodes of recurrent FTBTCS lasting for about 30 min each in the first 24 h with prolonged impaired awareness, despite the addition of continuous intravenous fosphenytoin (400 mg/day). Ictal electroencephalogram (EEG) during FTBTCS showed bilateral polyspikes but was obscured by artifact; ictal EEG during FIAS showed rhythmic 5–9 Hz theta activity in the right frontotemporal region (data not shown). EEG recorded during an interseizure period revealed irregular right frontal sharp waves with theta and delta activities (Figure 1). Magnetic resonance imaging (MRI) of the brain showed no obvious signs suggesting osmotic demyelination syndrome (ODS); however, marked atrophy of the hippocampus and temporal lobe was seen (Figure 2). Further questioning of the patient’s family revealed that he had been diagnosed with AD 2 years before admission. Although atrophy of the hippocampus and temporal lobe has been associated with seizures in patients with AD,2–4 no symptomatic epileptic seizures had been recognized before hospitalization. On day 6, the frequency of FTBTCS decreased, but intermittent FIAS and FTBTCS did not completely disappear. Correction of hyponatremia was suspended to lower serum sodium to 125 mEq/L. Fosphenytoin administration was discontinued and was replaced with intravenous levetiracetam (1000 mg/day); however, FTBTCS continued intermittently. The addition of perampanel (4 mg/day) and clonazepam (3 mg/day) to levetiracetam was ineffective at controlling seizures. Administration of lamotrigine (200 mg/day), carbamazepine (600 mg/day), and levetiracetam (1000 mg/day) on day 7 was also ineffective. Continuous intravenous infusion of midazolam (5 mg/h) was introduced to treat SE on day 8. Thereafter, both FIAS and FTBTCS subsided. Repeat MRI of the brain on days 7, 14, and 28 revealed no obvious lesions suggesting ODS on diffusion-weighted, T2-weighted, or fluid attenuation inversion recovery imaging. Reduction of the midazolam dose was not possible because of relapsing seizures. The patient’s general status gradually deteriorated; he died of aspiration pneumonia on day 58.
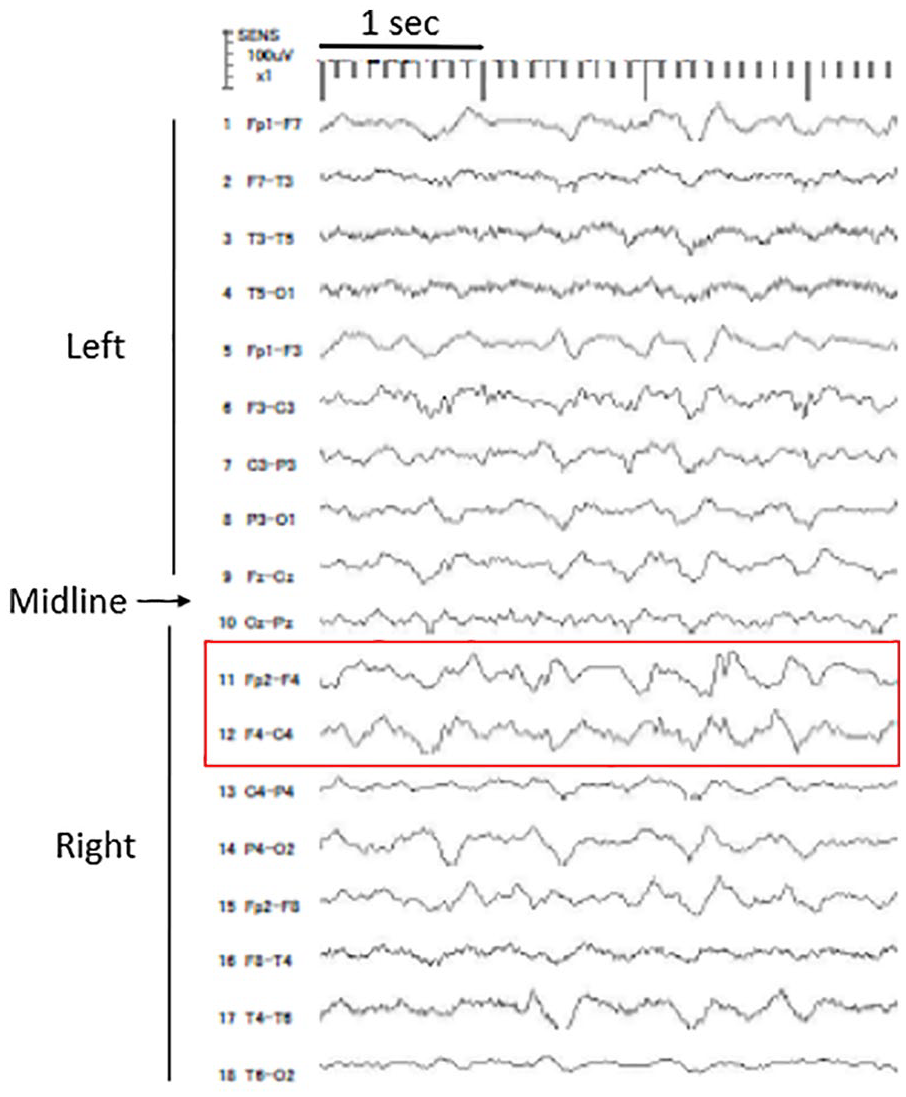
Electroencephalogram recorded during interseizure period on day 5. Sharp waves were recorded in the right frontal region (red box). These waveforms also showed disruption of background activity and subsequent emergence of slow waves. Low resolution and clipping traces are due to malfunction of the recording device.
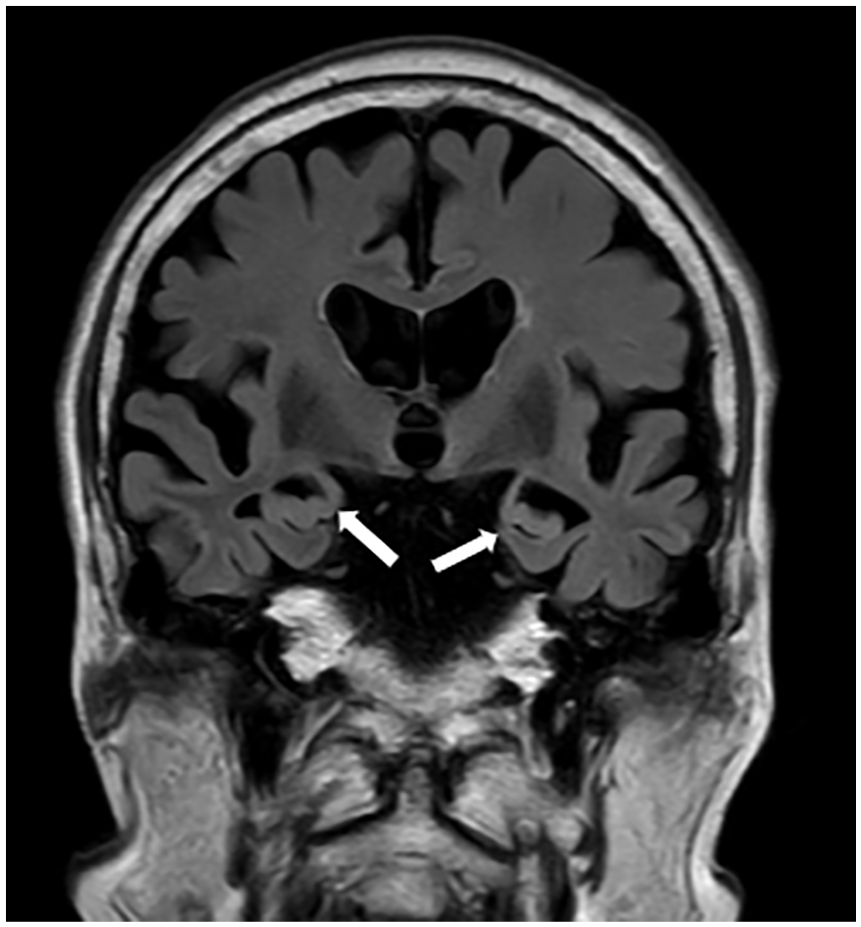
Coronal fluid attenuation inversion recovery magnetic resonance imaging of the brain on day 5. Bilateral atrophy of the hippocampus is observed (white arrows). No other clear abnormal findings, including ischemic leukoencephalopathy, were found. Diffusion-weighted imaging of the brain showed no significant abnormalities.
Discussion
In the present case, refractory FIAS and FTBTCS with subsequent SE occurred during correction of hyponatremia resulting from polydipsia. Hyponatremia may lead to acute symptomatic seizures, which are usually generalized tonic-clonic. 5 In addition, acute epileptic seizures and focal neurological deficits are identified concomitantly in 5% of patients with severe hyponatremia (<125 mEq/L). 5 When correcting hyponatremia, ODS may also cause seizures, 6 but no lesions suggesting ODS were found in the present case. Okazaki et al. 7 reported the effects of polydipsia-induced hyponatremia on pre-existing FIAS in five patients. In that report, the frequency of symptomatic FIAS increased when serum sodium fell below 127 mEq/L; four of the five patients had hippocampal atrophy or sclerosis and developed FIAS or FTBTCS. This finding suggests that hyponatremia may aggravate both FIAS and FTBTCS in patients with hippocampal atrophy or sclerosis.
It should be noted that hyponatremic seizures per se are not rare events and are often observed in children and women of childbearing age; 5 however, SE associated with hyponatremia is rare. To the best of our knowledge, only nine patients with SE associated with hyponatremia have been reported in the PubMed database in the past 30 years.8–15 Characteristics of the nine reported patients and our patient are shown in Table 1. Among these 10 patients, six were male and four were female. Patient age ranged from 10 to 83 years; serum sodium levels ranged from 90 to 126 mEq/L. The causes of hyponatremia were, in the order of descending prevalence, polydipsia (four cases), syndrome of inappropriate secretion of antidiuretic hormone (SIADH, three cases), cerebral salt wasting (two cases), and adrenal insufficiency (one case). Hence, polydipsia and SIADH may be leading causes of hyponatremia-associated SE. Regarding seizure types, generalized tonic-clonic seizures were most common (four cases); complex partial seizures (two cases) and non-convulsive SE (two cases) were also observed. Eight patients had no past history of epileptic seizures. No patients developed ODS during the treatment of hyponatremia. In the nine previously reported cases, antiepileptic drugs effectively stopped SE; these patients all recovered without sequelae. Our patient was the only one with AD and whose outcome was death.
Patients with status epilepticus secondary to hyponatremic seizures.
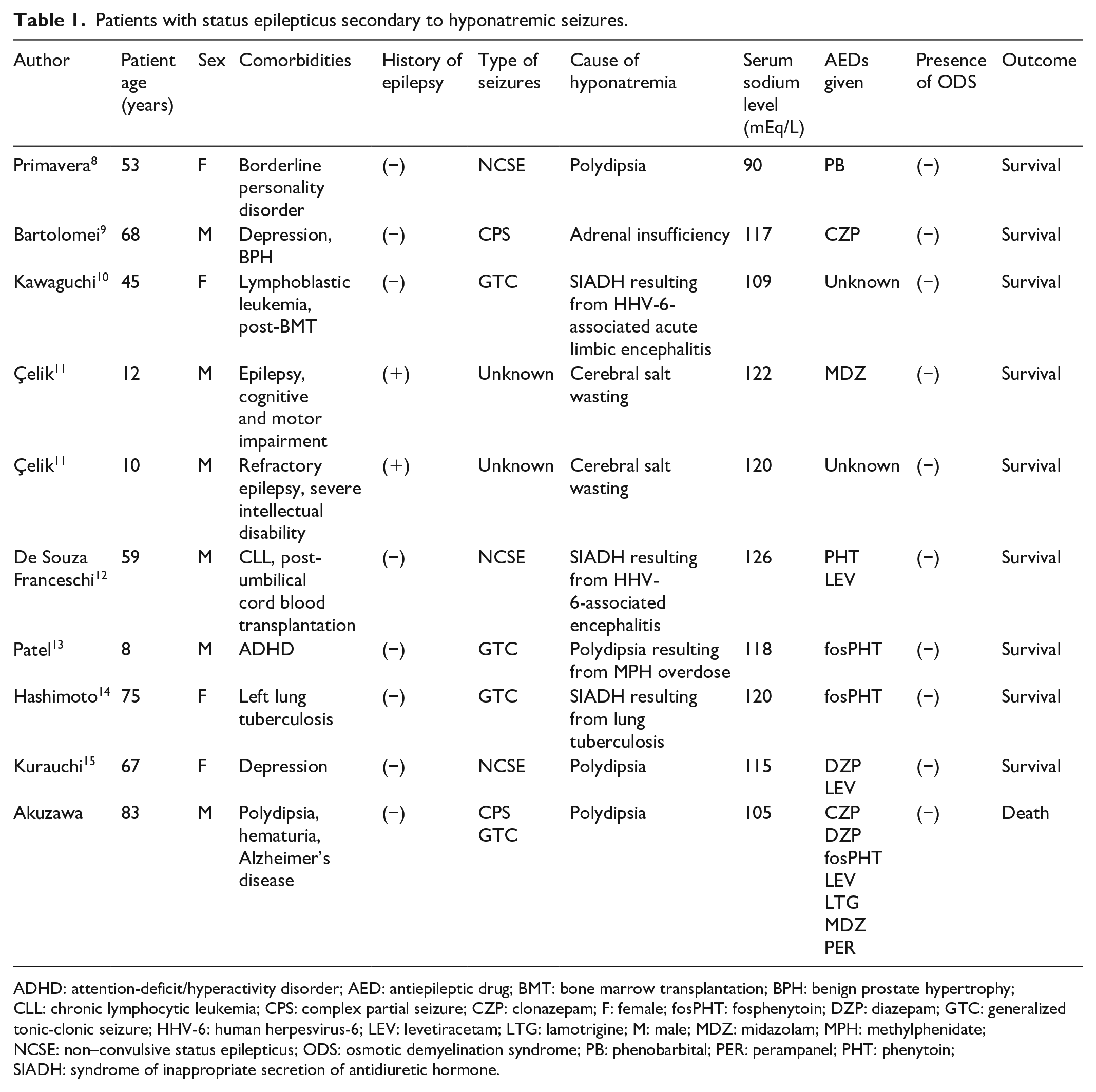
ADHD: attention-deficit/hyperactivity disorder; AED: antiepileptic drug; BMT: bone marrow transplantation; BPH: benign prostate hypertrophy; CLL: chronic lymphocytic leukemia; CPS: complex partial seizure; CZP: clonazepam; F: female; fosPHT: fosphenytoin; DZP: diazepam; GTC: generalized tonic-clonic seizure; HHV-6: human herpesvirus-6; LEV: levetiracetam; LTG: lamotrigine; M: male; MDZ: midazolam; MPH: methylphenidate; NCSE: non–convulsive status epilepticus; ODS: osmotic demyelination syndrome; PB: phenobarbital; PER: perampanel; PHT: phenytoin; SIADH: syndrome of inappropriate secretion of antidiuretic hormone.
Interestingly, our patient showed rhythmic 5–9 Hz theta activity in the right frontotemporal region during FIAS. This finding is similar to the typical ictal EEG of temporal lobe epilepsy and is specific for hippocampal seizures. 16 In contrast, EEG during a seemingly interseizure period showed irregular sharp waves with theta and delta activities in the right frontotemporal region; this pattern resembles the ictal pattern of temporal lobe epilepsy associated with neocortical seizures. 16 These observed EEG patterns may also indicate multiple epileptogenic foci. According to previous studies, hippocampal sclerosis and multifocal spikes on EEG may be associated with FTBTCS in patients with temporal or frontal lobe epilepsy.17,18 Moreover, AD is a leading cause of frontal or temporal epilepsy in the aged population. AD can cause SE, but SE in our patient was unresponsive to first-, second-, and third-line therapies and recurred for ⩾24 h.19–22 These characteristics meet the criteria of super-refractory SE, which has a distinctively high mortality (about 40%).23–25 To our knowledge, no similar super-refractory SE cases have been reported among AD patients. Factors related to the onset of super-refractory SE in our patient are unclear, but hyponatremia might have accelerated neuronal hyperexcitability resulting from AD-related presynaptic glutamate release, leading to SE.4,4,26
The limitations of our report include the lack of EEG information before hospitalization. Regarding EEG data during hospitalization, a mechanical breakdown of recording devices prevented capture of complete data and therefore we could not show ictal EEG data during FIAS and FTBTCS. In addition, although this patient was diagnosed with AD 2 years before admission, the precise time of AD onset was unclear. Regarding imaging, follow-up MRI of the brain was not performed after day 28 because of deterioration of the patient’s general status. However, this case highlights interesting mechanisms of FIAS proceeding to FTBTCS or SE and may also alert physicians to the potential risk of triggering SE under hyponatremic conditions.
In conclusion, we have presented a case of super-refractory SE during correction of chronic hyponatremia. Physicians should be aware of the potential risk of triggering SE when correcting hyponatremia in patients with AD.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient and legally authorized representatives including the patient’s wife and son for anonymized patient information to be published in this article. A copy of this written consent is available for review by the Editor-in-Chief of this journal.
