Abstract
Ovarian masses larger than 100 pounds are rarely encountered in developed countries given advancements in early diagnosis and treatment. Their successful resections pose unique surgical and anesthetic challenges. An otherwise healthy 38-year-old para 1 woman developed a 50 × 60 cm pelvic mass. An exploratory laparotomy, left salpingo-oophorectomy and anterior abdominal wall reconstruction were performed. A total of 60 L of cystic fluid were drained. Close monitoring of hemodynamics and massive volume resuscitation required intensive care. Inpatient physical rehabilitation reinstated independent mobility. Final pathology revealed benign ovarian mucinous cystadenoma. A multidisciplinary approach in the preoperative, intraoperative and postoperative stages of management optimizes patient outcomes.
Introduction
Surface epithelial tumors with mucinous differentiation account for 15% of all ovarian neoplasms in the United States and Europe. 1 About 80% of mucinous ovarian neoplasms are benign unilocular or multilocular cystadenomas, 10%–15% are borderline tumors and less than 10% are carcinoma. 1 Mucinous cystadenomas are characterized by endocervical or gastrointestinal tract epithelial cells and collect mucin in the cytoplasm. They occur mostly in the reproductive age group, are typically unilateral and can reach 30 cm or more in diameter with reports of weighing up to 135 kg. 2
Gigantic ovarian masses are rare in developed countries due to the availability of medical care and diagnostic imaging. Although patients may fear their malignant potential, most gigantic masses tend to be benign. From 1970 to 2006, there were 19 cases of gigantic ovarian cysts in the English literature defined as weighing more than 40 kg. 3 About 14 of these cases (73.6%) were benign mucinous cystadenomas ranging from 47 to 134 kg with the maximum fluid drained intraoperatively reported at 44 L. We present a case of a 60 L, 132-pound ovarian mucinous cystadenoma and discuss its successful removal.
Case report
A 38-year-old para 1-0-0-1 patient presented with accelerated abdominal growth and decreased mobility during the prior 3 months. The patient had no significant medical, gynecologic or family history. Physical exam demonstrated cachexia with temporal wasting, a mobile, firm 80–100 cm abdominal mass and anasarca.
Computed tomography of the abdomen and pelvis revealed a 50 × 60 cm cystic mass without definite complex elements or a clear origin of the mass (Figure 1(a) and (b)). The gastrointestinal tract, pancreas, adrenal glands and vasculature were poorly evaluated. The urinary bladder, uterus and adnexa could not be identified. The lungs indicated no pleural effusion and the liver was partially visualized with no focal lesions. The peritoneal surfaces appeared normal, and there was no definite adenopathy. CA 125 and CA 19-9 were 50 and 54 U/mL, respectively. Tumor markers carcinoembryonic antigen (CEA), inhibin A, inhibin B, human chorionic gonadotropin (HCG), lactate dehydrogenase (LDH) and complete metabolic panel were within normal range. Hemoglobin and hematocrit were 12.6 g/dL and 39.9%, respectively.
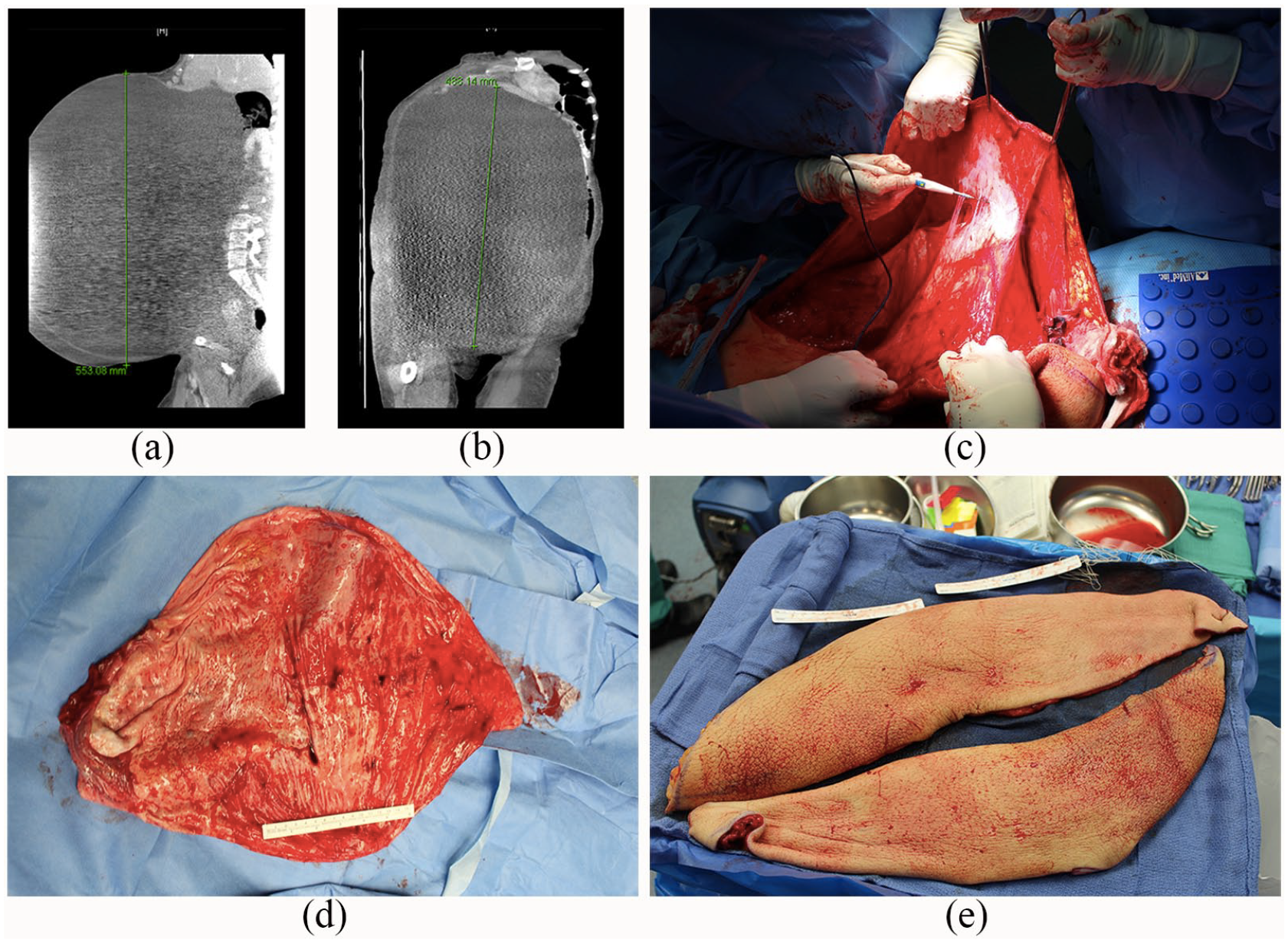
(a) Computed tomography of gigantic tumor (sagittal view), (b) computed tomography of gigantic tumor (coronal view), (c) densely adherent tumor wall dissected away from peritoneal surfaces, (d) left ovary and tumor wall measured 51 × 42 cm with a thickness ranging from 0.2 to 0.9 cm and (e) the excess pannus was removed in two elliptical portions measuring 54 × 15 × 2.0 cm and 57 × 15 × 2.5 cm, weighing 2 kg.
An echocardiogram indicated the right atrium was extrinsically compressed by the abdominal mass, and the ventricles were underfilled with an ejection fraction of 70%–75%. An anterior abdominal wall reconstruction was planned for by plastic surgery at the same time as the pelvic mass resection due to anticipated redundant skin and subcutaneous tissue.
Preoperatively, the patient weighed 154 kg. Epidural catheter placement was attempted for postoperative analgesia but without success. General anesthesia was administered. Arterial line and central venous pressure line were placed. Transesophageal echocardiogram was used as a real-time monitor of the patient’s cardiac function and fluid status. Her mean arterial pressure (MAP) was between 80 and 100 mm Hg.
The patient was placed in the left lateral recumbent position to avoid supine hypotension. Given her relatively stable condition, a midline vertical skin incision was made from pubic symphysis to the umbilicus. Careful dissection was carried down to the cyst wall, which appeared smooth but was densely adherent to the peritoneum. A 1-cm incision was made in the cyst wall and a pool suction was inserted to drain 60 L of clear, sero-mucinous fluid in four discrete fractions. The patient responded hemodynamically to removal of the first aliquot with an acute decrease in MAP to 65–70 mm Hg (Figure 2). A phenylephrine infusion was initiated and her MAP was maintained at 80–90 mm Hg for the duration of the procedure.
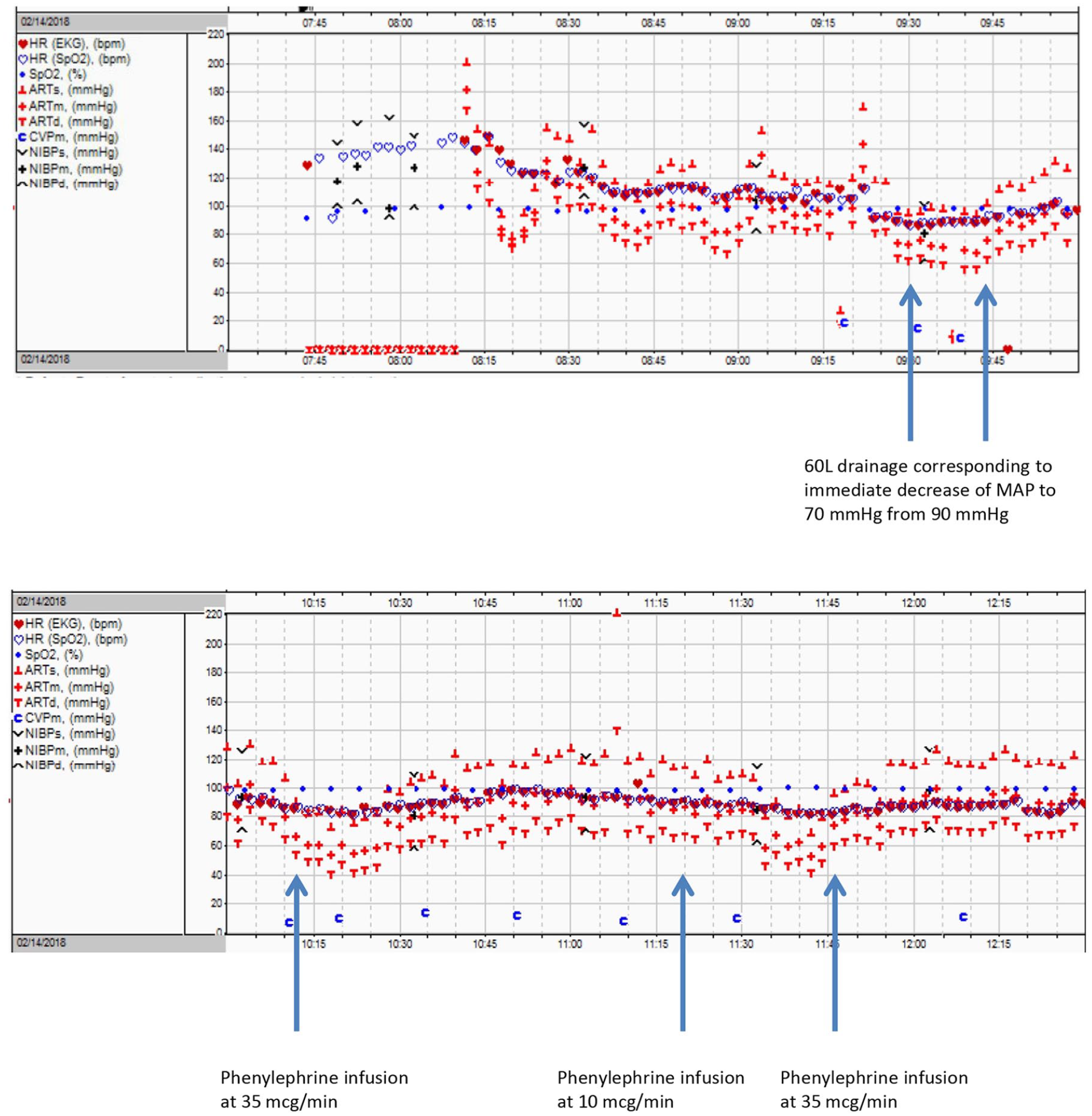
Cardiovascular status during cystic drainage.
Following complete aspiration, the skin incision was extended to the xiphoid process to perform a thorough exploration of the abdomen and pelvis, which revealed the gigantic cyst originating from the left ovary. The thin cystic wall was carefully dissected from the peritoneal surfaces (Figure 1(c)). A left salpingo-oophorectomy was performed with vessel sealer and free tie sutures. The left ovary and tumor wall measured 51 × 42 cm with a thickness ranging from 0.2 to 0.9 cm (Figure 1(d)). The uterus, right fallopian tube and right ovary were grossly normal in appearance. There were no gross liver or bowel lesions, ascites or carcinomatosis.
The excess pannus and peritoneum were removed in two elliptical portions measuring 54 × 15 × 2.0 cm and 57 × 15 × 2.5 cm, weighing 2 kg (Figure 1(e)). A vest-over-pants complex abdominal wall reconstruction was performed involving both leaves of the anterior rectus fascia. A Jackson-Pratt drain was placed, and the skin was closed with staples.
Final pathology of the left ovary revealed a benign mucinous cystadenoma. The ovarian cystic fluid was negative for malignant cells.
Total cystic fluid drained was 60,000 mL, which equates to approximately 60 kg (132 pounds) in weight. Remaining intraoperative fluid output was 1500 mL estimated blood loss, and 550 mL urine output. The patient was resuscitated with 500 mL intravenous crystalloid, 1300 mL packed red blood cells, 610 mL fresh frozen plasma and 500 mL 5% albumin. Blood loss was mostly during cyst wall dissection, lysis of adhesions and abdominal wall reconstruction.
The patient was admitted to the intensive care unit (ICU) for mechanical ventilation, weaning vasopressor medications and close monitoring of fluid shifts. Postoperatively, the patient weighed 98 kg. Prophylactic low molecular weight heparin was started for 4 weeks due to anticipated immobility. The patient developed an ileus, which was managed conservatively. After 2 weeks of inpatient physical rehabilitation, she was discharged at independent mobility level with a plan for continued outpatient physical therapy.
At 8 weeks follow-up, she had resumed regular menses and returned to work. She weighed 70 kg—a total weight loss of 83 kg from the day of surgery.
Discussion
Surgical technique of gigantic masses may involve drainage of cystic fluid, cystectomy, oophorectomy and/or hysterectomy. In our case, the massive dimensions required vertical laparotomy and initial drainage of cystic fluid in a controlled manner for better visualization of the pelvic anatomy. Given preoperative imaging indicated no solid or complex elements of the mass, there was low suspicion for malignancy potential. Although previous case series have discouraged cystic drainage to avoid possible malignant seeding, those reports indicated complex, multicystic masses, which were more easily rolled away from the patient after excision. In our case, the mass was encapsulated by a thin cyst wall containing almost 60 L of fluid, which would not have been able to be resected intact. Given the flimsy 1-cm cyst wall, attempt at intact removal would have more likely caused cyst rupture than with controlled drainage. There are reports that intraoperative drainage would have caused cardiac failure, dyspnea or pulmonary edema;3 –5 however, these are consequences that would also occur with resection of an intact gigantic mass. Furthermore, intraoperative controlled drainage may help prevent splanchnic vessels and inferior vena cava to be suddenly released as seen in resection of a mass. 3
Conclusion
A multidisciplinary approach was crucial to our successful patient outcome. Continued close monitoring of hemodynamic changes was provided by the intensive care and anesthesia teams. Complex abdominal wall reconstruction by plastic surgery ensured minimal disfigurement and postoperative body image satisfaction. Early physical therapy and long-term physical rehabilitation assisted with mobility as well as bowel and respiratory function. Our case reviews the surgical and anesthesia challenges that come with removal of gigantic masses and highlights that detailed planning of preoperative, intraoperative and postoperative management will optimize patient outcomes.
Footnotes
Copyright permission
All photographic images were taken by author A.S.M. and are owned by authors A.S.M. and V.A. Both authors have permitted the images to be reproduced in the publication of this manuscript. Based on a press release from the authors’ institution, there have been reports of this patient case in news media. In contrast, this manuscript describes the preoperative evaluation, intraoperative technique and postoperative management in a peer-reviewed format for the scientific community.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article. Informed consent was obtained to publish the patient information and images.
