Abstract
A few cases of platypnea-orthodeoxia syndrome have been described in the literature, some of them after thoracic or upper abdominal surgeries. In most cases, hypoxemia in the upright or sitting position, which is the main clinical symptom for this uncommon diagnosis, is usually related to a dynamic right to left cardiac shunt induced by anatomical changes in the relative position between the inferior vena cava and the atria in the presence of a patent foramen ovale. In this case report, we describe a situation in which platypnea-orthodeoxia syndrome developed acutely before surgery but that became severely exacerbated after an open urologic surgery without a clear acute anatomical change that could be responsible for triggering the syndrome. This case might suggest that the pathophysiology of acute platypnea-orthodeoxia syndrome is not completely elucidated and that other possible triggers for acute clinical manifestation in addition to acute anatomical thoracic changes must be explored.
Introduction
Platypnea-orthodeoxia syndrome (POS) is a rare cause of hypoxemia in which dyspnea usually develops over weeks to years and is exacerbated in the upright position and relieved by the recumbent position.1,2 The most common pathophysiology of POS involves an intermittent and dynamic right-to-left cardiac shunt triggered by anatomical changes in the position of the atria in the presence of a patent foramen ovale (PFO) and usually in the absence of pulmonary hypertension. These anatomical changes may include a progressive dilation of the ascending aorta3,4 or can be induced more acutely by thoracic trauma5,6 and thoracic 7 or upper abdominal surgeries,8–10 among others. In this report, we describe a case of acute-onset hypoxemia due to POS in a patient before a surgical urologic intervention that became severe after surgery with no obvious cause.
Case presentation
An 88-year-old male patient was admitted to the intensive care unit (ICU) due to severe and acute hypoxemia 3 days after hospital admission. He had a diagnosis of urinary retention due to benign prostate hyperplasia and was treated for a urinary infection prior to surgical intervention. His past medical history included only cigarette smoking (discontinued 20 years before hospital admission). On the day of ICU admission, the rapid-response team was called to the ward because the patient had an SO2 of 67% while breathing room air. All previous measurements in the ward in those 3 days were above 90% while the patient being supine in the bed. The cause of hypoxemia was not evident. The patient was lying in bed and was not using antithrombotic prophylaxis. His blood pressure was 120/80 mmHg at that moment, with a heart rate of 56 beats/min, a respiratory rate of 20 breaths/min and no fever. Supplemental oxygen with a nasal catheter 2 L/min did not reverse the hypoxemia. The patient was immediately transferred to the ICU, although the hypoxemia reverted spontaneously. There were no signs of respiratory distress, and his lungs were clear. He was admitted to the ICU with an SO2 of approximately 95% in room air. Common alternative diagnostic hypotheses usually identified by a simple chest X-ray, such as pulmonary edema, pneumonia, or atelectasis, were excluded. A contrast-enhanced chest tomography additionally excluded pulmonary thromboembolism or arteriovenous malformations as causes of severe hypoxemia. The only relevant finding was an ectasia of the ascending portion of the aorta. The measured level of methemoglobin was 0.8%, while the patient had a hemoglobin level of 16.5 g/dL at ICU admission.
Two days after ICU admission, the patient was considered able to be subjected to open prostatectomy and cystolithotomy, although he was using supplemental low-flow (1–2 L/min) oxygen by a nasal catheter to maintain an SO2 above 90%. There was no intercurrence during the procedure (sedation + spinal anesthesia) except for deterioration in the patient’s SO2. He returned from the operating room with an SO2 of less than 80% using an oxygen mask with a reservoir bag (fractional inspired oxygen near 100%). The patient’s lungs remained clear, and there were no signs of respiratory distress. In the subsequent days, his SO2 oscillated from moderate (85%) to severe hypoxemia (75%) with no apparent cause, and he was not able to remain without the 100% oxygen mask even in the semi-recumbent position. He started to experience mild dyspnea. A high-flow nasal catheter barely improved his oxygenation, and non-invasive mechanical ventilation (BIPAP) reverted his hypoxemia, but only using high levels of fraction of inspired oxygen (FiO2 > 60%). A microbubble-enhanced transthoracic echocardiography in the semi-recumbent position revealed only ectasia of the ascending aorta and an increased left atrium, but no signs of pulmonary hypertension and no apparent intracardiac or pulmonary shunts (the quality of the exam was considered poor by the echocardiographist for anatomical reasons). Four days after surgery, the patient experienced an abrupt decrease in oxygenation while seated out of bed (until that moment, he had been constantly in bed in a 30° recumbent position). He developed dyspnea, cyanosis, and transient loss of consciousness, which improved soon after his return to bed while remaining in a lying position. After this episode, it was noted that the patient’s oxygenation improved significantly in the supine position and deteriorated in a more upright position.
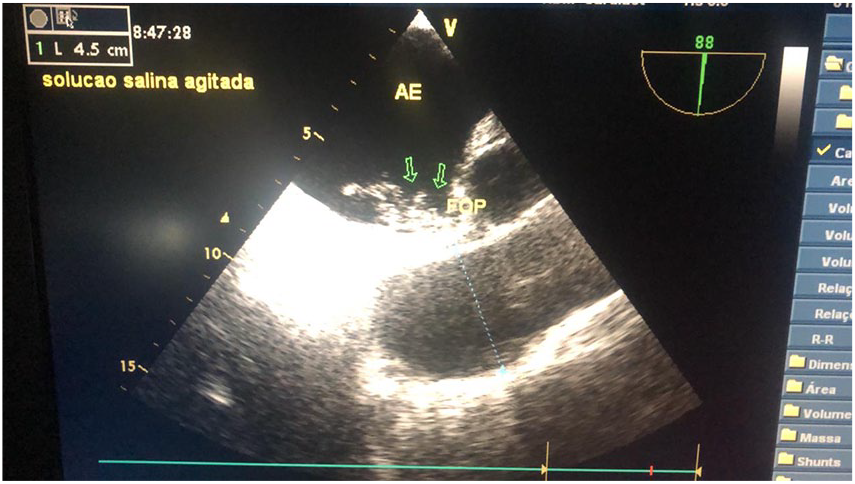
A microbubble-enhanced transesophageal echocardiography after endotracheal intubation (saline infusion made through the same venous access in the arm as in the transthoracic) revealed a right-to-left atrial shunt due to a PFO with a septal aneurysm (Figure 1) and confirmed an increased diameter of the ascending aorta (4.5 cm; Figure 2). During mechanical ventilation, the pattern of improved saturation in the lying position remained, and severe hypoxemic episodes did not recur, even with the intubated patient seated out of bed, although there was a need for an increased FiO2 in this position.

Transesophageal echocardiography image of the interatrial septal aneurism.
Transesophageal echocardiography image of the right-to-left shunt with spontaneous microbubble passage across the patent foramen ovale (PFO).
The PFO was closed percutaneously (using a CeraFlexTM PFO occluder) a few days later. Right heart catheterization prior to the PFO closure has shown a pulmonary artery pressure of 37/21 (26) mmHg, a pulmonary artery occlusion pressure of 17 mmHg and a right atrial pressure of 5 mmHg. The patient was extubated the next day after the procedure with no recurrence of hypoxemia and no need for supplemental oxygen. The rest of his hospital stay was unremarkable.
Discussion
POS is a rare syndrome, usually described in older patients (>70 years old), which leads to hypoxemia (sometimes severe) with no clinically relevant findings upon physical examination, except for a body position-dependent SO2 (significant decreases while sitting or standing). The pathophysiology of POS is still not completely elucidated, but ascending aortic dilation is frequently part of the thoracic findings, in combination with an intracardiac shunt (usually a PFO, which is present in 25% of the population and may remain asymptomatic for life).3,4 A combination of anatomical findings could lead to a functional and dynamic right-to-left cardiac shunt, which usually occurs in the absence of pulmonary hypertension. In our patient, the diagnosis required a large amount of clinical suspicion, which only occurred after the observation that a fast improvement occurred in the recumbent position. Most cases of POS previously described in the literature had a more progressive installation, with dyspnea being the most important complaint developing over weeks to years. Acute POS cases are usually associated with acute anatomical thoracic changes due to trauma5,6 or surgery.7,9,10 In this case report, we highlight the sudden and disproportional degree of hypoxemia in relation to the degree of symptoms. We do not know which factors led to the acute onset of frequent episodes of hypoxemia. Although the patient had a relatively high blood hemoglobin level for a man of his age at ICU admission (which might suggest some degree of previous chronic hypoxemia), the patient remained in the ward for 3 days before the first documented episode of desaturation in room air. This fact argues against the hypothesis that the patient was already experiencing such episodes (at least, severe episodes) for longer periods, including at his home. The reason for the patient’s hospital admission did not seem to have any link with these hypoxemic episodes. Moreover, the fact that the episodes were more frequent, severe, and sustained after surgery led to the hypothesis that surgery could have exacerbated this phenomenon in some way, but the pathophysiology was not clear. In surgical patients, cases of POS after pneumectomy 11 or single-lung transplantation 12 have been described, probably because there are a number of anatomical thoracic changes after these types of surgery that could favor right-to-left shunt in the presence of PFO. Previous cases of POS after upper abdominal surgery8–10,13 or nonsurgical conditions, in which the right diaphragm was elevated,14,15 have been reported, and this anatomical change was the common finding that could have favored the right-to-left atrial shunt in these patients. In our patient, no significant anatomical thoracic change could have been attributed to an open prostatectomy with cystolithotomy. His chest X-ray was very similar before and after surgery. Although it could have been merely a temporal coincidence, we think that a non-obvious phenomenon could have made the clinical postoperative scenario worse than that in the preoperative period. POS was already present at ICU admission, but caution is advised before any surgical procedure in patients with unresolved POS. To the best of our knowledge, we are the first to report an acute and sustained deterioration of hypoxemia due to POS after an open lower abdominal surgery.
Conclusion
The pathophysiology of POS remains not completely elucidated: acute and severe cases may occur even without obvious acute or recent anatomical thoracic changes and demands a high level of suspicion. The diagnosis is mandatory since the treatment (PFO occlusion) is relatively simple, effective and, in our patient, life-saving.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series. The patient’s daughter agreed with anonymous publication of the case.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
