Abstract
Intracranial aneurysms have an estimated prevalence of about 3%. A rare subgroup are aneurysms of the internal carotid artery that develop medially into the sellar region. Due to the risk of rupture with subsequent subarachnoid hemorrhage and of compression of surrounding structures, mechanical occlusion is advised. Hypopituitarism is not a rare disease and most often related to pituitary adenoma. Only 0.17% of cases with hypopituitarism are caused by unruptured intracranial aneurysms. Today, the predominant treatment of these aneurysms is endovascular coiling or application of flow diverting stents. We present the case of a 60-year-old female patient, who was treated with endovascular coiling for a right-sided, intracavernous, incidental internal carotid artery aneurysm. On postinterventional day 6, she was readmitted with contralateral third nerve palsy, mild hyponatremia und thyreotropic insufficiency. The symptoms recovered after anti-edematous treatment with corticosteroids; only an asymptomatic hyperprolactinemia persisted. To the best of our knowledge, this is the first case report of transient contralateral cranial nerve palsy combined with transient hypopituitarism after endovascular treatment of an internal carotid aneurysm. As treatment we propose corticosteroids, if necessary in combination with nonsteroidal anti-inflammatory drugs, in order to inhibit inflammatory reactions of the aneurysm wall compromising the nearby, partially compressed neural structures.
Introduction
Intracranial aneurysms occur with a prevalence of 3%–5%. 1 Most aneurysms remain asymptomatic, until rupture or mass effect occurs. The most dangerous complication is subarachnoid hemorrhage after rupture of the aneurysm. Therefore, depending on the risk of rupture, mechanical occlusion is usually necessary. Only 1%–2% of intracranial aneurysms are located in the sellar region close to the pituitary gland. 2 These aneurysms can become symptomatic by diplopia or vision impairment due to affection of cranial nerves as well as by endocrinological symptoms.
As
Usually, pituitary insufficiency is detected before treatment, or even initially leads to the diagnosis of the aneurysm. The therapy remains challenging, since in only three of the published cases hypopituitarism recovered after occlusion of the aneurysm9,29,33 (Table 1).
Case reports published after the reviews by
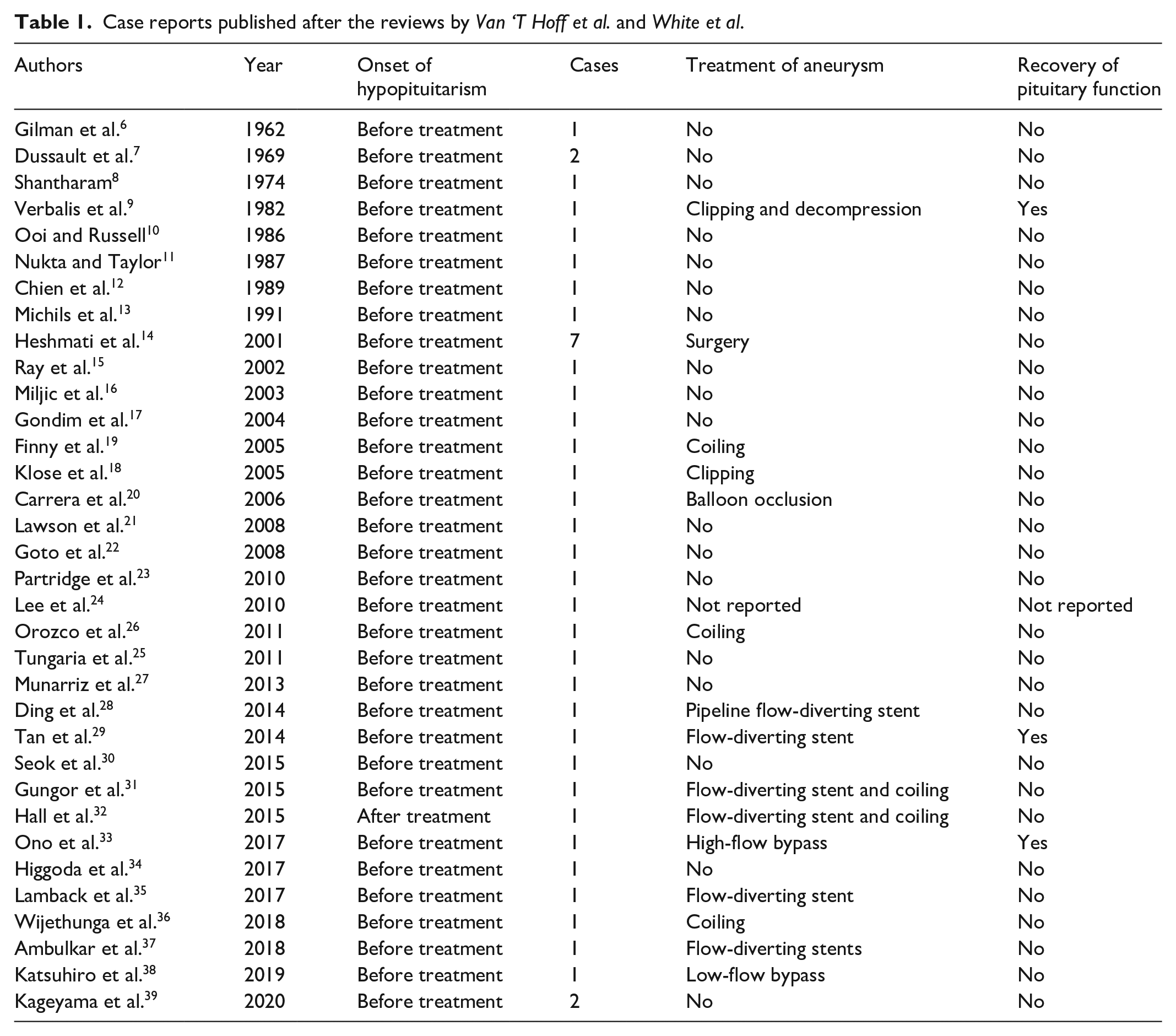
More generally, hypopituitarism is not an uncommon disease with a prevalence of 45.5/100.000 and can be accompanied by hyperprolactinemia due to pituitary stalk affection. Most cases (70%) are caused by tumorous lesions.40,41 Only 0.17% are caused by intracranial aneurysms. 14 While hypopituitarism can lead to unspecific symptoms such as chronic fatigue, loss of libido or hyponatremia, the accompanying hyperprolactinemia can cause galactorrhea, breast swelling or hypogonadism. Untreated hypopituitarism eventually ends with coma, cardiac arrythmia and death. The treatment of hypopituitarism consists in hormone substitution and, if possible, removal of the sellar tumor. This is especially true for pituitary adenomas, except for prolactinomas, which can be treated more efficiently with dopamine agonists.
Case report
A 60-year-old female presented to her general practitioner because of intermittent headaches. Computed tomography of the head brought up the suspicion of a right-sided internal carotid artery (ICA) aneurysm of 25 mm, protruding into the sellar region, which was confirmed by magnetic resonance imaging (MRI).
After digital subtraction angiography, endovascular coiling was recommended and the patient agreed to the intervention. Two months later, the aneurysm was densely packed with 24 platin coils (Penumbra 400) with assistance of a self-expanding stent (Neuroform EZ, 4.5 × 20). Final control showed complete occlusion of the aneurysm; no residual inflow could be detected (Figure 1). The patient was observed for four further days on our neurosurgical ward and then was dismissed home without neurological deficit.

Left: Anterior–posterior digital subtraction angiography of the right ICA before treatment. The aneurysm develops medially into the sellar region and measures 25 mm. After interdisciplinary discussion, endovascular treatment was recommended and performed with application of 24 coils (Penumbra 400) with assistance of a self-expanding stent (Neuroform EZ, 4.5 × 20). Right: Control series at the end of the first intervention.
On postinterventional day 6, she was readmitted to the emergency room because of diplopia. Clinical examination revealed unilateral palsy of the left oculomotor nerve, contralateral to the site of the aneurysm. Endocrinological work-up disclosed new mild hyponatremia (133 mmol/L) and new thyreotropic insufficiency (Table 2). MRI on postinterventional day 7 did not detect subarachnoid hemorrhage or increased compression of neural structures, but revealed inflammatory signals in the aneurysm wall with contact to the pituitary stalk and the left oculomotor nerve (Figure 2). Anti-edematous treatment with dexamethasone led to full recovery of clinical symptoms and normalization of sodium and thyroid-stimulating hormone (TSH) serum levels within 1 week.
Serum concentrations of sodium (Na+), thyroid-stimulating hormone (TSH), free triiodthyronine (fT3), and free thyroxine (fT4) before treatment (2013), on postinterventional day 6 (onset of symptoms, 2014), and after completion of treatment (2017, no symptoms).

TSH: thyroid-stimulating hormone.
Note that hyponatremia and thyreotropic insufficiency resolved after completion of treatment.

Magnetic resonance imaging on postinterventional day 7. The mass effect of the aneurysm compromises all structures in proximity to the sella turcica. Note the strong inflammatory signal close to the left oculomotor nerve and the pituitary stalk, as well as around the whole wall of the aneurysm. Top: Coronal contrast enhanced T1 sequence of the sellar region. A: giant aneurysm of the right ICA after occlusion, I: left ICA, C: optic chiasm, S: pituitary stalk, P: pituitary, O: left oculomotor nerve in the wall of the cavernous sinus, *: inflammatory signal. Bottom: Axial T2 sequence of the sellar region. A: giant aneurysm of the right ICA after occlusion, I: left ICA, B: head of the basilar artery, O: left oculomotor nerve, *: inflammatory signal.
Subsequently, the patient needed multiple treatments for reperfusion of the aneurysm. In August 2014, 6 months after first intervention, 25 additional platin coils (Penumbra 400) were applied. In October 2015, 20 months after first intervention, two flow-diverting stents (Phenox p64, 3.5 × 15 and 3.5 × 21) were inserted. In February 2017, 3 years after first intervention, another flow-diverting stent (Phenox p64, 3.5 × 18) was administered. Consecutively, the aneurysm then was permanently occluded, as shown in the digital subtraction angiography from August 2017, 42 months after first treatment (Figure 3). In contrast to the first intervention, the subsequent steps went without further complications.

Anterior–posterior digital subtraction angiography of the right ICA 42 months after first treatment. In addition to the first intervention, 25 platinum coils (Penumbra 400) and three flow-diverting stents (Phenox p64, 3.5 × 15, 3.5 × 21 and 3.5 × 18) have been applied in three additional interventions. In contrast to the first intervention, the subsequent steps went without further complications.
Meanwhile, the patient has received
Pituitary hormones after completion of treatment at 57 and 61 months after first coiling.
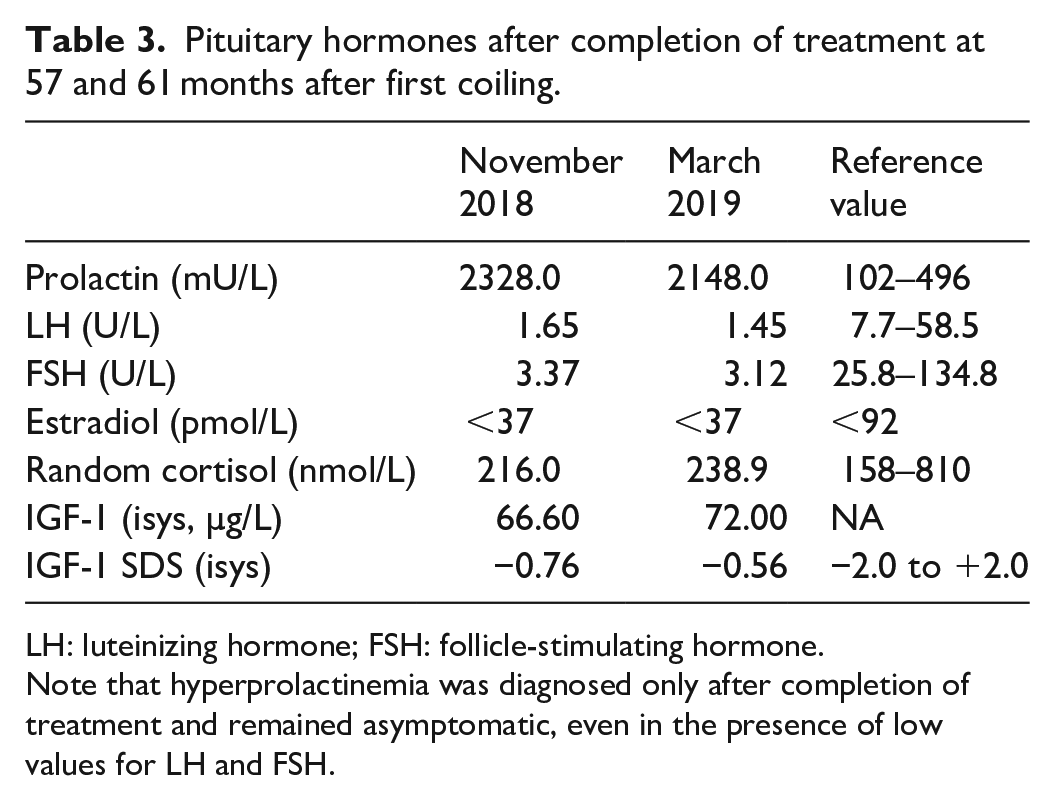
LH: luteinizing hormone; FSH: follicle-stimulating hormone.
Note that hyperprolactinemia was diagnosed only after completion of treatment and remained asymptomatic, even in the presence of low values for LH and FSH.
Discussion
Hypopituitarism and impairment of cranial nerves in the cavernous sinus are well-known symptoms when diagnosing a tumor of the sellar region. The case presented here points to an important differential diagnosis and fits well into the existing reports of ICA aneurysms provoking these deficits.
However, these symptoms usually stand at the beginning of the diagnostic work-up that finally detects the aneurysm, whereas in our case, the symptoms occurred within 1 week after endovascular treatment. Most likely, mass effect and inflammatory reactions of the aneurysmal wall lying in closest vicinity to the involved structures led to metabolic dysregulation and induced the deficits (Figure 2). This can explain the contralateral oculomotor nerve palsy and the resolution of symptoms by anti-edematous treatment with dexamethasone.
Literature research identified only one further case report, in which hypopituitarism occurred after treatment. 32 In that case, hypopituitarism was delayed by 18 months after the placement of a flow-diverting stent and did not recover, rather suggesting a late ischemic event than inflammation as etiology. However, compression of surrounding structures by coil material or induced by intra-aneurysmal microbleeding has to be taken into account, too, since non-ischemic cerebral enhancing lesions occur in only 0.5% of endovascular interventions. 42
Recovery of pituitary function after occlusion of an ICA aneurysm as the underlying cause is rare and relies on residual pituitary function before treatment. Interestingly, the mode of occlusion seems to be unrelated, since recovery has been achieved by clipping and decompression, 9 flow-diverting stent, 29 high-flow bypass surgery, 33 as well as, in our case, by anti-edematous treatment after endovascular coiling.
Conclusion
Hypopituitarism and cranial nerve impairment can occur after endovascular treatment of asymptomatic sellar aneurysms of the ICA. Treatment of choice comprises corticosteroids.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
We acknowledge support from Leipzig University for Open Access Publishing.
Informed consent
Written informed consent was obtained from the patient for publication of this case report.
