Abstract
Background:
Perforator flaps have recently been used in the field of plastic surgery. Skin defects can be reconstructed to reach functional and cosmetic goals. With the development of reconstructive approaches, utilizing stem cells is a hopeful approach to enhance wound healing and tissue recovery. In this study, we assessed the effect of bone marrow mesenchymal stem cells on the perforator flap’s survival in rats.
Methods:
Perforator flaps (2.5 × 11 cm) were transplanted into rats and focused on the thoracodorsal, intercostal, and deep circumflex arteries, which were randomly divided into three groups: control, saline, and bone marrow mesenchymal stem cells (7 × 106 ml). Seven days after the surgery, tissue edema, inflammation, and discharge were observed and photographed. Histological analyses were performed to determine flap survival. Hematoxylin and eosin staining was performed to assess levels of microvascular density determined in skin flaps.
Results:
Rats in the bone marrow mesenchymal stem cells group exhibited higher average flap survival area, and higher microvascular density levels at the dynamical regions of the flaps compared with the other two groups. Subdermal injection of bone marrow mesenchymal stem cells significantly increased ischemic perforator flap survival due to stimulated neovascularization in rats.
Conclusion:
Our findings suggest the potential usefulness of bone marrow mesenchymal stem cells in preventing skin flap perforator tissue necrosis.
Keywords
Introduction
A flap is defined as a unit of tissue that is transferred from a donor site to a recipient site while its blood supply is preserved. Flaps exist in various forms, from simple advancement flaps to composite flaps. 1 A perforator flap is a flap that comprises a skin or subcutaneous fat. The supplying blood vessels are isolated perforators. 2 Perforator flap is considered a significant concept on reconstructive surgery. It provides an opportunity to perform less invasive and more complicated procedures by conserving essential blood vessels and muscles with intramuscular vessel dissection. The term “perforator” indicates that the vessel perforates the muscle, followed by the vessel perforating the deep fascia irrespective of muscle perforation. 3 Recognition of a perforating vessel and dissection in a retrograde fashion is necessary to harvest a perforator flap successfully.
In contrast to conventional flaps, the approach initiates from the distal part of circulation at the subcutaneous tissue layer continuing to the source vessel to harvest a perforator flap. The flap harvest is usually free-style, considering the anatomical variations of the pedicle. 4 Partial flap necrosis is the most frequent adverse outcome of flap surgery. The distal part of flaps is more likely to develop ischemic necrosis. 5 The underlying causes are inadequate arterial supply or inadequate venous outflow. 6 The most efficient way to prevent flap necrosis is to provide sufficient blood flow in the microcirculation. Many angiogenic factors affect the stimulation, promotion, and stabilization of new vessels, including vascular endothelial growth factors, fibroblast growth factors, angiopoietins, platelet-derived growth factors, monocyte chemotactic protein-1, and transforming growth factors. 5 Stem-cell-based therapy has also been suggested to be beneficial for stimulating early angiogenesis, which can effectively prevent skin flap necrosis. 7 Mesenchymal stem cells (MSCs) are adult multipotent stem cells with great potential in regenerative medicine due to their multilineage differentiation ability. 8 Bone-marrow-derived mesenchymal cells also secrete factors useful for tissue repair. 9 The main reason for this is their capacity to communicate with other cells and to carry a significant amount of bioactive molecules, such as proteins, nucleic acids, and lipids. These molecules facilitate wound healing by exerting anti-inflammatory effects, inhibiting apoptosis, promoting angiogenesis, and facilitating enhanced cell migration and proliferation. 10 Considering the reported influence of stem cells on tissue recovery and prevention of flap necrosis, this study was conducted to evaluate the effect of stem cells on the survival of the perforator flap in rats.
Methods and materials
Laboratory animals
This current experimental study was conducted at the Faculty of Medicine, Lorestan University of Medical Sciences, Khorram Abad, IRAN. 30 Albino Wistar rats with an average weight of 250–300 g were purchased from Lorestan Aftabavaran Company (Khorramabad, Lorestan, Iran). Before the start of the study, an ethical clearance from the Health Research Ethical Committee of Medical faculty was obtained (number: IR.LUMS.REC.1400.096). All procedures performed were under the standards outlined in the Guide for the Care and Use of Laboratory Animals Declaration. All authors declared that all legal and ethical aspects of the current study have been fulfilled before and during the study period. For animal identification, the pairing method was a selective method with standard equipment and laboratory animal handling tools such that racks for each group, cages for each animal, and cage cards to name were separate, all of which formed the basis of the identification process.
Cells cultures
The standard protocol was used to obtain bone marrow mesenchymal stem cells (BMMSCs), which were isolated from bone marrow obtained by perfusing the femur. In phosphate-buffered saline (PBS), sterile tubes with 50-IU/mL heparin and 0.25-mg/L gentamicin were used to transport the marrow aspirate to the laboratory, which was at a temperature of 20–25°C. After placing 10 mL of bone marrow aspirate into a centrifuge tube, it was pelleted at 350g for 5 min. After removing all excess water, the cell pellet was placed in 20 mL of lysis buffer (114 mM NH4Cl, 7.5 mM KHCO3, 0.1 mM EDTA) and allowed to dissolve for 3–5 min. After second centrifugation, the cell pellet was resuspended in a nutrient medium for BMMSCs (DMEM/F12 (Invitrogen, Waltham, MA, USA), 10% FBS (Invitrogen, Waltham, MA, USA), 0.4 μM insulin, 20 ng/mL bFGF, 10 nM dexamethasone, 100 μg/mL penicillin, and 100 μg/mL streptomycin (Invitrogen, Waltham, MA, USA)). The cells were passed through four times in accordance with a standard protocol. Briefly, 5–7 mL of a suspension of mononuclear cells (1.0–1.5 × 106 cells/mL) was sown in culture flasks (25 cm2) and incubated at 37 ©, 5% CO2 under high-humidity conditions. The culture medium was changed every 72 h. The cells were washed in Dulbecco’s PBS with no Ca2 and Mg2 ions from Sigma-Aldrich in Salisbury, UK after reaching 90% confluency, and washed with the same substance as the other ones.
Then, cells were removed from the culture plastic with triple solution (Invitrogen, Waltham, MA, USA), centrifuged at 350g for 5 min, the supernatant was collected, and the pellet was resuspended in a nutrient medium, next 1/3 of the cell suspension was placed on a new culture flask. Four times, the cycle was repeated. Flow cytometry analysis and documentation of stem cell features were performed on the fourth passage of BMMSCs. The flow cytometry analysis procedure was similar to that reported before. In summary, the Iranian Biological Resource Center in Tehran, Iran (code number IB RCC10164) characterized surface markers CD90, CD45, CD34, and CD105. The differentiation of MSC into multiple different lineages was induced by osteogenic differentiation in a α-MEM medium that contained 0.1 μM of dexamethazone, 50 μM of ascorbic acid, and 10 mM of glycerophosphate. Besides, adipogenic differentiation was induced in α-MEM with 0.5 μM of dexamethazone, 0.5 mM of 3-isobutyl-1methylxanthine, and 0.1 mM of indomethacine. 10 BMMSCs were labeled with red fluorescent molecule CM-DiI (C7000; Invitrogen). Diluting CM-Dil with 95% ethanol at 20% concentration produced the cell labeling solution, followed by selecting cells of 1 × 106 in 1 mL of DMEM. Each 1 mL of cell suspension was mixed with gentle pipetting after adding 5 mL of the cell labeling solution. Next, incubation was done for 20 min at 37°C, and then, the falcon in which the cells were suspended was placed along with CM-Dil for centrifuging at a speed 1500 rpm for 5 min, preferably at 37°C. Finally, after removing the supernatant, the cells were gently suspended in a warm environment (37°C) once again in the DMEM. 11 In order to estimate the percentage of live cells, trypan blue staining was utilized to distinguish between live and nonliving cells. To put it briefly, cell samples were diluted with normal saline. For viability assessment, the cell staining was done with a 0.4% Trypan blue solution (GibcoTM) and the counting was conducted in a Neubauer chamber. A Neubauer chamber was loaded with 10 μL of the solution after mixing trypan blue with cell suspension and pipetting it properly. Afterward, the cells were inspected under a microscope in all four hemocytometer squares, and the number of cells per square (106 g/mL) was determined.
Surgical method and cell implantations
The rats were anesthetized with intraperitoneal phenobarbital (60 mg/kg). 11 After anesthesia, the animals were fixed to the operating table in the prone position. The dorsal area was shaved and disinfected with betadine and 70% alcohol. A rectangle measuring 11 × 2.5 cm was drawn. The upper side of the rectangle was located between the spine of the scapula, and the lower side was located between the iliac crest. It was then dissected, and a skin flap was harvested. All skin connections except the particle were cut. There was a deep circumflex iliac artery on the right side of the flap. 12 There were the anatomical region (deep circumflex iliac), the dynamic region (intercostal artery), and the potential region (thoracodorsal artery) in the flaps. 11 After ligature of thoracodorsal and intercostal arteries, the flap was sutured to its original location (Figure 1(a)–(d)). 13

Performing perforator flap model in rat skin, ((a)–(d)) drawing a rectangle measuring 2.5 × 11 cm on animal dorsal skin ((b) and (c)), performing skin flap surgery and cutting all skin connections except the pedicle (d). Dorsal perforator flap. The perforator flap in rats shows intercostal artery, thoracodorsal artery, and deep circumflex iliac artery. Choke I is located between the anatomical and dynamic regions; Choke II is situated between the dynamic and potential areas.
In the subgroups receiving cell injection, BM-MSCs (7 × 106 cells per 0.25 mL of PBS) were injected intraoperatively with an insulin syringe into the area of flap implantation to ensure cell colonization of the wound surface (Figure 2), 14 the flap was sutured at a distance of 0.5 cm from the skin with nylon 4-0. In the saline receiving group, 50 ccs of 0.9% normal saline were injected at 8 points of the flap, and then the skin was sutured (Figure 2). In the control group, no intervention was done. Immediately after surgery, pictures were taken of each flap using a camera (Sony Song Jap). Samples were taken on the zero-day and day 7 after surgery.15–17 All rats were examined for complications like acute inflammation, abscess formation, serum accumulation, or necrosis on day 7 after flap surgery. The survival area, distinguishable from the necrotic area, was defined as an area with no color change or stiffness.

The minor black points show the injection site of MSCs and saline in flaps.
Measurement of the flap on day zero (surgery day)
After surgery on day 0, the flap size was measured to confirm that it was the same in all three groups of rats, and this range was transferred to a sterile, transparent sheet placed on the flap. 18 In the paper template area (PTA) method, patterns designed in checkered paper with a resolution of 300 dpi were scanned, and the total area of the flap was measured using ImageJ 1.52v software (LOCI, University of Wisconsin-Madison, USA). 19
Measurement of the flap on day 7 after surgery
The survival area in all three groups was recorded by the PTA method with a Sony jap camera, and then the photos were analyzed by ImageJ software. We measured the survival rate of each flap using the following formula. Then, the unit conversion to mm2 was performed.
Histological examination
All the rats were euthanized chloroform agents. Samples were extracted, fixed with 10% neutral buffered formalin, and embedded in paraffin 7 days after implanting. Serial paraffin sections with a thickness of 5 μm were stained with hematoxylin and eosin (H&E), for further morphological assessment using performed on 100 random fields in 10 samples from each group to count the number of vessels. The number of blood vessel densities is calculated based on stereological methods, which reports 3D-related information in terms of three-dimensional values. The framework of a field is arranged by a unique counting frame in the dermal area. Volume Density of the vessels from Vv (vessel/dermis) was counted as P (Vessel)/P (dermis), where the number of P (Vessel) was placed between the points of the pattern. 20
Statistical analysis
The statistical analysis of obtained data was carried out using GraphPad Prism 7.0 software (Boston, USA) with three-way analysis of variance (ANOVA) and post hoc analysis using Tukey’s mean. The use of nonparametric statistics (Spearman’s rank correlation coefficient) was utilized to search for correlations. A p-value below 0.05 was what the level of statistical significance was defined as.
Results
Gross explanations
All animals endured the flap surgery. None displayed any signs of complications, inflammation and/or transudation at the surgical site.
Marker expression
Flow cytometry showed that more than 82% BMMSCs expressed CD90 and CD105 and about 1% expressed CD34 and CD45.
Following of transplanted cells
On the seventh day after surgery, we noted that the CM-Dil-labeled BMMSCs could still be found in the region of the flaps in the BMMSCs group (Figure 3).

CM-Dil-labeled BMMSCs (arrows) distinguished in the skin flap by a fluorescent microscope at day 7 after surgery. BMMSCs, bone marrow mesenchymal stem cells.
Findings of a viable perforator flap
On the seventh day after surgery, the mean survival area was 62.5 mm2 in the control group, 56.4 mm2 in the saline receiving group, 75.8 mm2 in the BMMSCs receiving group, and 64.9 mm2 in all three groups. On the seventh day following surgery, there was no significant difference between the groups in the mean survival area of the perforator flap (p = 0.107) (Figure 4).
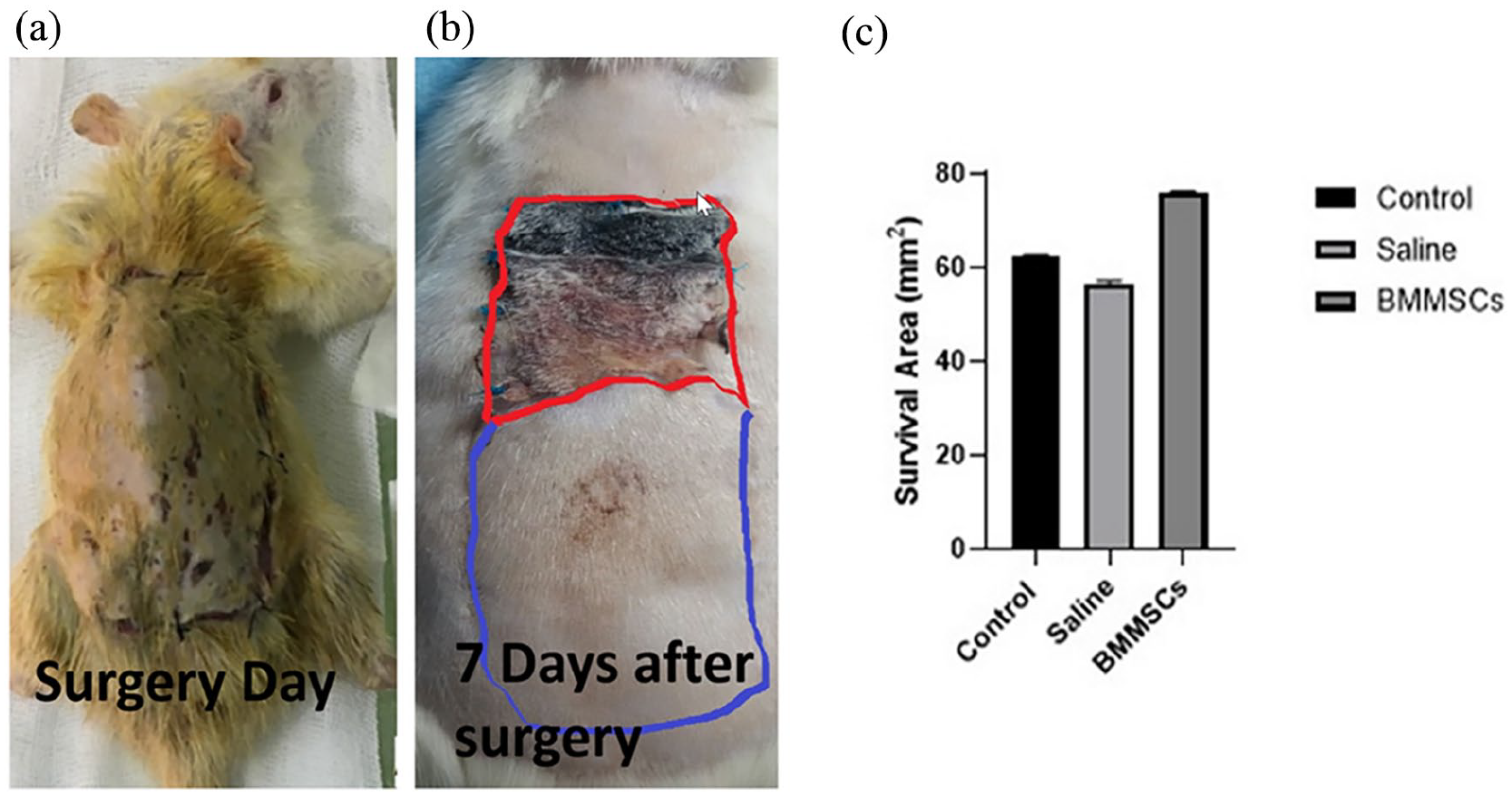
The macroscopic examination of the perforator flap on the day of surgery (a) and day 7 after surgery (b). In Figure (b), the necrotic area is marked with a red line, and the survival area is marked with a blue line. (c) Comparison of mean survival area of perforator flap tissue in all three groups on the seventh day after surgery.
Histopathological examinations of vessels
The control group averaged one vessels per group, while the saline receiving group averaged one vessels per group, and the MSC receiving group averaged two vessels per group. The mean number of vessels in all three groups was 1.3667. Post hoc Bonferroni correction showed that the number of vessels in the MSC group was significantly higher than in the saline and control groups (p < 0.001), but there was no significant difference between the number of vessels in the saline and control groups (p = 0.99) (Figure 5).

Histopathological evaluation demonstrates that d (dermis), v (vessels), and h (hair follicle) formation and maintains its original architecture. D Represents the comparisons of the number of vessels of perforator flap tissue between groups on the seventh day after surgery (Scale bar 50 µm).
Discussion
Ischemic necrosis resulting from inadequate blood supply is a crucial complication of flap surgery. The special techniques and advantages of perforator flaps can make any reconstructive procedure better for both functional and aesthetic purposes. In the beginning, the perforator flaps were used as open flaps, leading to a significant decrease in donor site morbidity. The flaps’ viability is not affected by the location of the perforator within the zone of injury, which can be a potential disadvantage. The capture of multiple adjacent perforasomes is possible through direct and indirect linking vessels by hyperperfusion in a perforator. 21 Venous congestion of the tip or the entire flap is a frequent problem with perforator flaps, which is caused by the insufficient flow in the perforator pedicle. The reason for this is either the perforator’s insufficient selection or dissection and mobilization, particularly around the vein. 22 Different surgical and non-surgical methods have been suggested to decrease the risk of ischemia. One of the surgical methods to deal with ischemia is to change the main position of the flap. The use of growth factors that stimulate angiogenesis has been proposed as a promising way to enhance the survival of flaps. 23 Furthermore, it has been established that bone-marrow-derived cells play a crucial role in both ischemic and non-ischemic wound healing processes. MSC therapy in particular is considered important agents against flap necrosis due to their angiogenic features. 24 The use of stem cells has its advantages, but it comes with limitations. Because they are not thought to be safer, have the potential for tumorigenesis, and carry the risk of embolism. 25 It is still important to address the issue of immune rejection in the clinical setting. Cell loss can cause a limited efficacy and a potential for serious adverse events in certain circumstances. The ability to repeat doses of cell therapy is limited by immune rejection, at the very least. Efficacy can be achieved only for a limited duration of cell survival, which is dependent on a short-term trophic effect for some indications. Many treatments aim to replace tissues or organs, which means that long-term cell survival and avoidance of host immune responses are required. 26 To address this challenge as well as inflammation and hyoxia, we proposed a strategy using BMMSCs in flap reconstruction type perforation with three main vessels for wound treatment, which did not result in a significant increase in the survival area due to the relatively limited lateral perfusion at the wound site, but was effective in the distribution and proliferation of micro vessels. The emerging field of regenerative medicine has rendered possible the bioengineering of a skin flap one type engineered vascularized biologic or composite tissue. Research on the bioengineering of skin flap to date mainly involves two aspects: the types of biomaterials used, and strategies for promoting neovascularization. 27
Investigated the influence of chicken embryo extract and BM-MSCs on skin flap survival in rats.
They demonstrated that transplantation of BM-MSCs and chicken embryo extract improved flap survival. Despite this, the biomechanical assessment revealed a slight contrast between the treatment and control groups. 28 To improve the hemostatic and antioxidant properties of wound dressings, a hybrid hydrogel was designed that uses polyvinyl alcohol, gelatin (GA), and borax. Therefore, it is well-suited foe wound that need recurrent extending and for use in parts of the body that are in continuous signal and can provide defense to wounds by efficiently fastening the wound site, falling the risk of more injury or corruption. 25
Chen et al. 28 evaluated factors released by BM-MSCs and their impact on the cells responsible for wound healing. They reported that BM-MSCs secreted a high amount of chemokines and cytokines, which could improve the wound healing process. Moreover, the BM-MSC-conditioned medium considerably increased the migration of keratinocytes, macrophages, and endothelial cells and the proliferation of endothelial cells and keratinocytes. Ding et al. 29 assessed the effects of BM-MSC on the survival of the three-territory perforator flaps in rats. BM-MSC injection led to a higher flap survival rate. Also, the diameter and density of microvessels increased in the BM-MSC group. Stem cell therapy can improve early angiogenesis by stimulating vascular endothelial growth factor (VEGF). The injection of bone-marrow-derived cells prevents skin flap necrosis and is an effective method for increasing the survival rate of skin flaps. 7
Conclusion
The present study results showed that BM-MSCs accelerated the wound healing process in rats. Using BM-MSCs in perforator flaps can be a promising concept in reconstructive surgery. Further research is needed to examine the impact of stem cells on flap survival and uncover the underlying mechanisms. Multiple functions, including hemostasis, antioxidant activity, oxygen delivery, and enhanced exosome transport, are combined in this advanced dressing in a synergistic way.
Footnotes
Acknowledgements
Our article is taken from a student thesis (Code; 2001) submitted for the award of a doctoral degree: Doctoral dissertation by Fatemeh Bazgir. This study was financed by grants from the Vice Chancellor of Research and Technology at Lorestan University of Medical Sciences, Khorramabad Iran (IR.LUMS.REC.1400.096).
Author contributions
Author contributions are as follows: F.C. designed this study, performed research, and revised the paper. F.B. performed the experiments; B.M. and F.C. analyzed the data; A.K.R. wrote the first draft of the manuscript. The final approval of the published version was the responsibility of all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) established without financial support and only Lorestan University of Medical Sciences provided academic support for this research.
Ethics approval
This study was conducted with the permission of the Research Ethics Committee of Lorestan University of Medical Sciences with the code IR.LUMS.REC.1400.096.
Animal welfare
All steps were completed based on the Guide for the Care and Use of Laboratory Animals.

