Abstract
Gastric mucosa-associated lymphoid tissue lymphoma is a rare clonal B-cell neoplasm that is usually associated with Helicobacter pylori infection. The presence of H. pylori should be confirmed via special stains and/or immunohistochemistry of gastric biopsies from multiple anatomic sites of the stomach. Mucosa-associated lymphoid tissue lymphoma is diagnosed with histopathologic examination and positive immunohistochemical staining for certain B-cell markers. Ancillary testing should be performed to determine any genetic abnormalities in H. pylori that increases its virulence. We report the case of a 49-year-old woman with recurrent epigastric pain and vomiting found to have chronic H. pylori gastritis despite appropriate rounds of treatment with first-line therapy. She was diagnosed with mucosa-associated lymphoid tissue lymphoma via histopathologic examination. She ultimately required oncological treatment due to H. pylori infection refractory to antibiotic treatment.
Keywords
Introduction
Gastric mucosa-associated lymphoid tissue (MALT) lymphoma constitutes 5%–8% of all B-cell lymphomas. 1 Concurrent Helicobacter pylori infection was initially demonstrated in over 90% of MALT lymphoma cases. 2 While most cases of H. pylori gastritis are uncomplicated and are easily treated with first-line triple therapy, refractory cases should prompt immunohistochemical evaluation and ancillary testing. Most cases of stages I and II MALT lymphoma regress with H. pylori eradication, thus underscoring the importance of a thorough gastric tissue analysis to diagnose MALT lymphoma with concomitant H. pylori infection. This report demonstrates a case of chronic H. pylori gastritis refractory to first-line treatment. Further diagnostic testing of gastric tissue revealed MALT lymphoma that required oncological treatment.
Case
A 49-year-old Caucasian female presented with worsening epigastric pain and vomiting that first began 3 months ago when she was diagnosed with H. pylori gastritis. She completed triple therapy with amoxicillin, clarithromycin, and pantoprazole, but follow-up esophagogastroduodenoscopy (EGD) showed persistent gastric ulcers with a biopsy positive for chronic active H. pylori gastritis. She denied weight loss, fever, malaise, hematemesis, or melena. She does not use tobacco, alcohol, or illicit drugs. Vital signs were within normal limits. Physical examination was significant for a tender epigastrium, but no rebound tenderness or guarding. No lymph nodes were palpated. Laboratory testing showed mild anemia with a hemoglobin of 11.8 g/dL. EGD with biopsy showed multiple antral ulcerations and a large posterior antral wall ulcer with heaped up edges with surrounding infiltrated-appearing mucosa (Figure 1). A hematoxylin and eosin (H&E) stain of the ulcerated tissue revealed a markedly dense lymphoid infiltrate, which was suspicious for lymphoma (Figure 2). A Giemsa stain demonstrated rare Helicobacter organisms. Immunohistochemical staining confirmed a B-cell predominance (Figure 3). Flow cytometry confirmed the clonal nature of this process consistent with a marginal zone lymphoma immunophenotype (Figure 4). Fluorescence in situ hybridization (FISH) analysis was negative for t(11;18)(q21;q21) (Figure 5). Positron emission tomography–computed tomography (PET-CT) demonstrated a hypermetabolic focus in the antrum of the stomach with no other hypermetabolic foci or enlarged lymph nodes (Figure 6). The patient was diagnosed with a gastric extranodal marginal zone lymphoma of MALT associated with H. pylori infection.

EGD showed a large posterior antral wall ulcer with heaped up edges and surrounding infiltrated-appearing mucosa.

H&E staining of the ulcerated region revealed a marked lymphoid infiltrate of monotonous, bland-appearing, small lymphoid cells with clear cytoplasm forming occasional lymphoepithelial lesions.
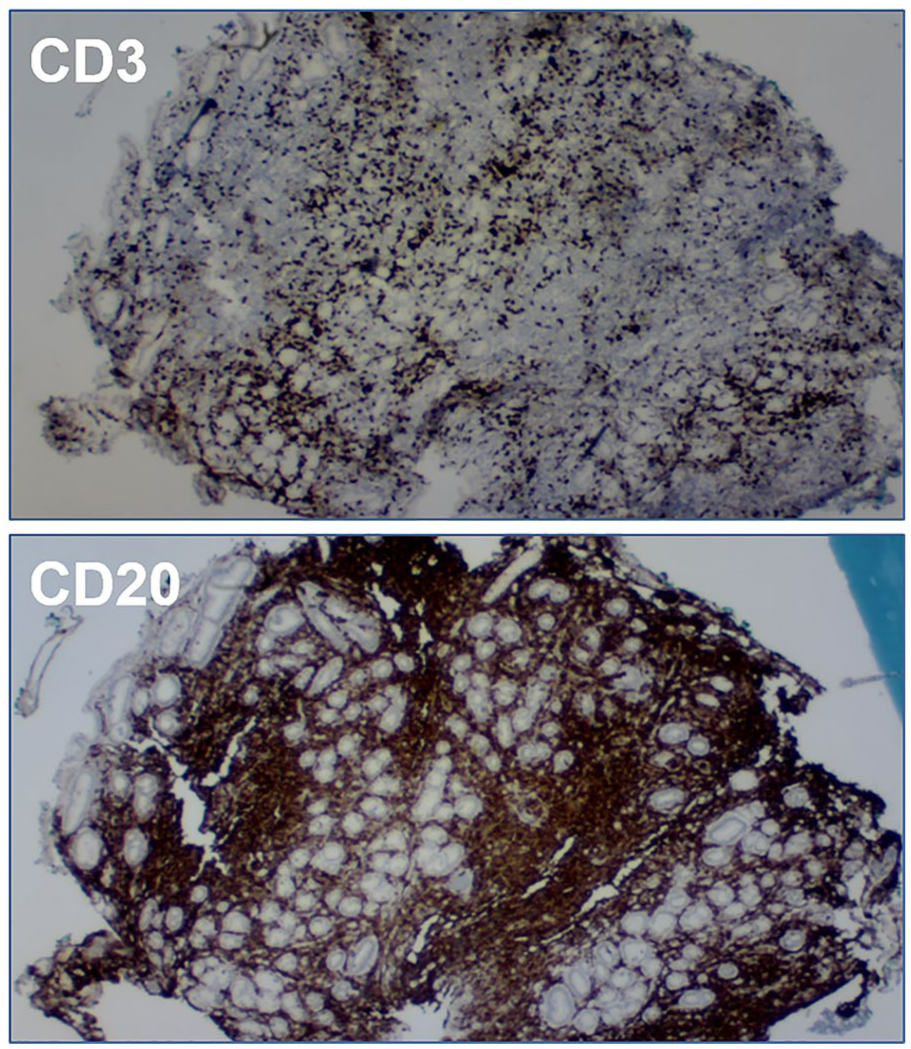
Immunohistochemical staining confirmed a B-cell predominance (CD20 positive stain) without co-expression of CD5 or CD10.
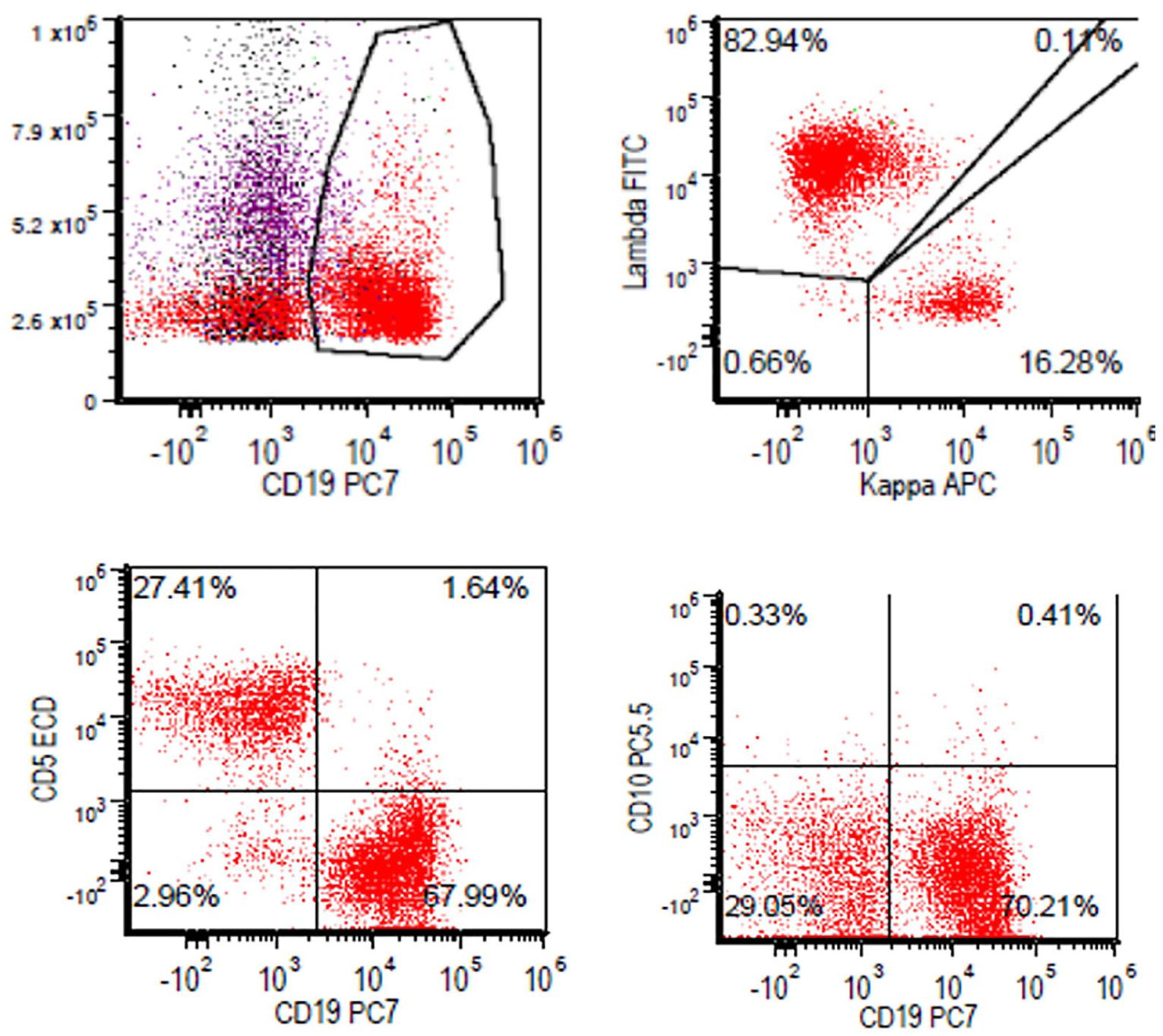
Flow cytometry revealed a prominent B-cell population with lambda light chain restriction without co-expression of CD5 or CD10, consistent with marginal zone lymphoma (MALT lymphoma).

FISH for BIRC3-MALT1 (11;18) fusion was negative. BIRC3 is represented in green and MALT1 is represented in orange.

PET-CT in the (a) axial and (b) sagittal views showed a hypermetabolic focus in the antrum of the stomach with no other hypermetabolic foci or enlarged lymph nodes.
A repeat course of triple therapy with levofloxacin, amoxicillin, and pantoprazole for 14 days was initiated. However, 2 months later, a urea breath test was positive for H. pylori detection. A repeat EGD with a Geimsa stain of gastric tissue was positive for Helicobacter organisms. Repeat pathological examination of gastric tissue demonstrated persistent MALT lymphoma. She was initiated on quadruple therapy with bismuth, tetracycline, metronidazole, and omeprazole for 10 days and referred to outpatient oncology. Two weeks after completion of quadruple therapy, a repeat urea breath test remained positive for H. pylori detection. After discussions regarding treatment options, she was initiated on rituximab therapy and completed 4 weeks of treatment. Follow-up evaluation with PET-CT and pathological analysis demonstrated full remission of MALT lymphoma. To this day, the patient remains in remission.
Discussion
MALT lymphoma is a rare clonal B-cell neoplasm that constitutes 5%–8% of all B-cell lymphomas. 1 The stomach is the most frequently affected organ where MALT lymphoma has been associated with prior or concurrent H. pylori infection resulting in chronic gastritis. 2 The gastric mucosa normally lacks lymphoid tissue, but the development of lymphoid tissue is stimulated in response to mucosal colonization by H. pylori. Rarely, this tissue can give rise to a MALT lymphoma. Epidemiologic studies show that the risk of gastric cancer was six to eight times higher for patients infected with H. pylori than for uninfected patients.3,4 A 15-year study in Shandong, China, an area of high rates of gastric cancer, found that eradication of H. pylori reduced the incidence of gastric cancer by 40%. 5
Since the majority of gastric MALT lymphomas are associated with H. pylori infection, gastric biopsies should be taken from various anatomic sites and routinely examined for H. pylori with special stains and/or immunohistochemistry. Various methods include immunostaining, Giemsa staining, or Alcian blue staining. Flow cytometry is used to confirm the clonal nature of the B-cells. Histopathologic examination reveals a dense lymphoid infiltrate with positive immunohistochemical staining for B-cell markers such as CD20 and CD79a. 6 The aberrant expression of CD43 can also help support the diagnosis of gastric MALT lymphoma. 6
Ancillary testing is not routinely done in simple H. pylori gastritis but should be done with gastric MALT lymphoma. The most common genetic abnormality detected in gastric MALT lymphoma is t(11;18)(q21;q21). This chromosomal translocation is associated with increased resistance of H. pylori to antibiotics and has been identified in 15%–40% of patients with gastric MALT lymphomas.7,8 Other genetic abnormalities include t(1;14)(p22;q32) and t(14;18)(q32;q21), but these translocations are not as common and their clinical relevance is unknown. Therefore, routine screening is not recommended. FISH for t(11;18)(q21;q21) is highly sensitive and specific and is routinely performed in newly diagnosed patients to direct treatment.
Another factor that increases the virulence of H. pylori is cagA-positive H. pylori. H. pylori that is positive for the cytotoxin-associated gene A (cagA) produces a toxin that can be injected into the intracellular junctions of the stomach mucosal lining, thus altering the stomach cell structure allowing the bacteria to attach more easily. This long-term exposure of the stomach lining to the toxin causes chronic inflammation and increases the incidence of MALT lymphoma. A meta-analysis of 16 case-control studies demonstrated that people infected with cagA-positive H. pylori had a two times increased risk of gastric cancer than those without cagA-positive H. pylori. 9
First-line treatment of MALT lymphoma is eradication of H. pylori with triple therapy. A second course of triple therapy or quadruple therapy is indicated if initial treatment fails. Successful eradication of H. pylori is followed by regression of stage I lymphoma in 78% of cases and stage II1 lymphoma in 56% of cases. 10 Repeated treatment failure is an indication for oncological treatment. Radiotherapy is highly effective in stage I/II1 disease, while chemotherapy with rituximab is suitable for advanced stages.11,12 Surgical resection is restricted to cases complicated by perforation or bleeding that cannot be managed endoscopically.11,12
Conclusion
The diagnosis of MALT lymphoma is made from a combination of clinical symptoms, endoscopic features, and histopathologic examination of biopsy tissue. H. pylori infection should be routinely established via stool antigen or serologic studies, as most cases of MALT lymphoma resolve with H. pylori eradication. Ancillary testing for drug sensitivity and FISH for t(11;18)(q21;q21) is recommended in cases of MALT lymphoma to further direct treatment. Repeated H. pylori treatment failure is an indication for oncological treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
