Abstract
A 73-year-old woman was referred to the pulmonology department for abnormal findings on chest computed tomography. She had undergone a laparoscopic staging operation including a hysterectomy, bilateral salpingo-oophorectomy, bilateral pelvic node and para-aortic node dissection, and concurrent chemo-radiation therapy for endometrial serous carcinoma stage IIIc cancer 15 months earlier. A follow-up chest computed tomography after the chemotherapy showed that the right lower lobe bronchus was obstructed, and it was necessary to differentiate a primary lung malignancy from a metastasis and secretion. A positron emission tomography revealed an intense hypermetabolic nodule in the right lower lobe bronchus and diffuse hypometabolism of the right lower lobe lung. Bronchoscopy revealed a tumor mass obstructing the right lower lobe bronchus, and an endobronchial biopsy confirmed a metastatic serous carcinoma from the endometrium. We described an endobronchial metastasis from an endometrial carcinoma with various diagnostic images and histology. To the best of our knowledge, this is the first report of an endobronchial metastasis from an endometrial carcinoma in Korea.
Introduction
Pulmonary metastases from extrathoracic malignancies are common. However, an endobronchial metastasis (EBM) is relatively rare and could be underestimated, since bronchoscopic examinations are not routinely performed in all patients presenting with pulmonary metastases.1,2 The incidence of an EBM varies (2%–50%) according to the definition of whether it includes invasion of the tracheobronchial tree by lung parenchymal masses and mediastinal lymph nodes or not.3,4 However, the incidence of an EBM has been increasing, as bronchoscopy has become more common, and the life expectancy of cancer patients increases.5,6
The most common extrathoracic malignancies associated with an EBM are breast, renal, and colorectal carcinomas.3,6 Pulmonary metastases are one of the typical sites of recurrent endometrial carcinomas, but on the other hand, an EBM is not a common metastatic form of an endometrial carcinoma. 7 EBMs from an endometrial malignancy have been reported by several investigators.7–9 Those EBMs were the types of bronchial invasions from lung parenchymal or mediastinal lymph node metastases. However, in our case, there were bronchoscopically visible lesions histopathologically identical to the extrapulmonary primary tumor, without any lung parenchymal or mediastinal lesions at the time of diagnosis.
Case
A 73-year-old female with continuous right lower quadrant abdominal pain accompanied by an elevated cancer antigen 125 had undergone a laparoscopic staging operation including a hysterectomy, bilateral salpingo-oophorectomy, and bilateral pelvic node and para-aortic node dissection due to endometrial carcinoma (serous carcinoma of the endometrium, high grade, stage IIIc) 15 months earlier. She was treated with concurrent chemo-radiotherapy with six cycles of cisplatin and 5400 cGy/6 weeks as a postoperative adjuvant therapy. Six cycles of chemotherapy with paclitaxel and carboplatin were followed due to multiple recurrences in the abdominal cavity. Four months after the last chemotherapy, computed tomography (CT) of the abdomen when the patient presented with lower abdominal pain showed bilateral hydronephrosis due to aggravation of the peritoneal seeding and a right percutaneous nephrostomy was performed. Although the patient was a nonsmoker and had complained of no respiratory symptoms, the chest CT revealed that the right lower lobe (RLL) bronchus was obstructed with segmental atelectasis (Figure 1(a) and (c)). It was a suspicious secretion stasis because the patient was nearly bed-ridden and weakened; however, positron emission tomography (PET)/CT revealed a hypermetabolic mass (SUVmax = 10.0) in the RLL bronchus with a diffuse hypometabolism of the RLL of the lung (Figure 1(b) and (d)). To evaluate the bronchial obstruction of the RLL, a flexible bronchoscopy was performed. The bronchoscopy revealed an endobronchial tumor obstructing the RLL bronchus (Figure 2(a)). To differentiate primary lung cancer from a metastasis, a bronchoscopic biopsy was performed and confirmed it was an endometrial cancer metastasis (Figure 2(b)). The pathology of the primary endometrial malignancy was reviewed, and it showed a similar pattern with an endobronchial lesion (Figure 2(c)). The immunohistochemical (IHC) study of the endobronchial lesion was negative for thyroid transcription factor-1, P40, and progesterone receptor (PR), and positive for p53. The IHC study for primary endometrial cancer was negative for Vimentin, Wilms’ tumor-1, and PR, and positive for p53. Based on the same pattern of the histology and IHC, which was negative for PR and positive for p53, the endobronchial mass was regarded as a metastasis from the endometrial malignancy. A multi-disciplinary team, including a gynecologist, radiologist, radio-oncologist, and medical-oncologist, discussed and decided on radiotherapy of the thorax considering the poor performance status (Eastern Cooperative Oncology Group 3 (ECOG 3)) of the patient. The patient received palliative radiotherapy of the right lung with 3900 cGy by 13 fractions. No additional chemotherapy or follow-up bronchoscopy was planned because of the patient’s generalized weakness and reluctance to undergo aggressive care. Accidently, the patient visited our emergency department because of abdominal pain and underwent a chest CT at a timing of 3 months after the conventional radiotherapy. A follow-up chest CT showed improvement in the atelectasis due to the EBM, and in contrast, multiple newly developed pulmonary and hepatic metastatic nodules were found. As the patient wanted hospice care, she received pain control and supportive care for her intestinal obstruction due to carcinomatosis, and then she was transferred to the hospice center. Nearly 10 months after EBM, she is still alive and takes hospice care.

(a) A chest CT displaying an endobronchial mass entirely occupying the right lower lobar bronchus. (b) PET/CT revealing a hypermetabolic nodule in the right lower lobar bronchus and diffuse hypometabolism (arrowheads) of the right lower lobe lung. (c) Coronal chest CT showing that the right lower lobar bronchus was obstructed by an endobronchial mass. (d) Coronal PET/CT showing that a hypermetabolic mass obstructs the right lower lobar bronchus and lobar hypometabolism (arrowheads). CT: computed tomography; PET: positron emission tomography.
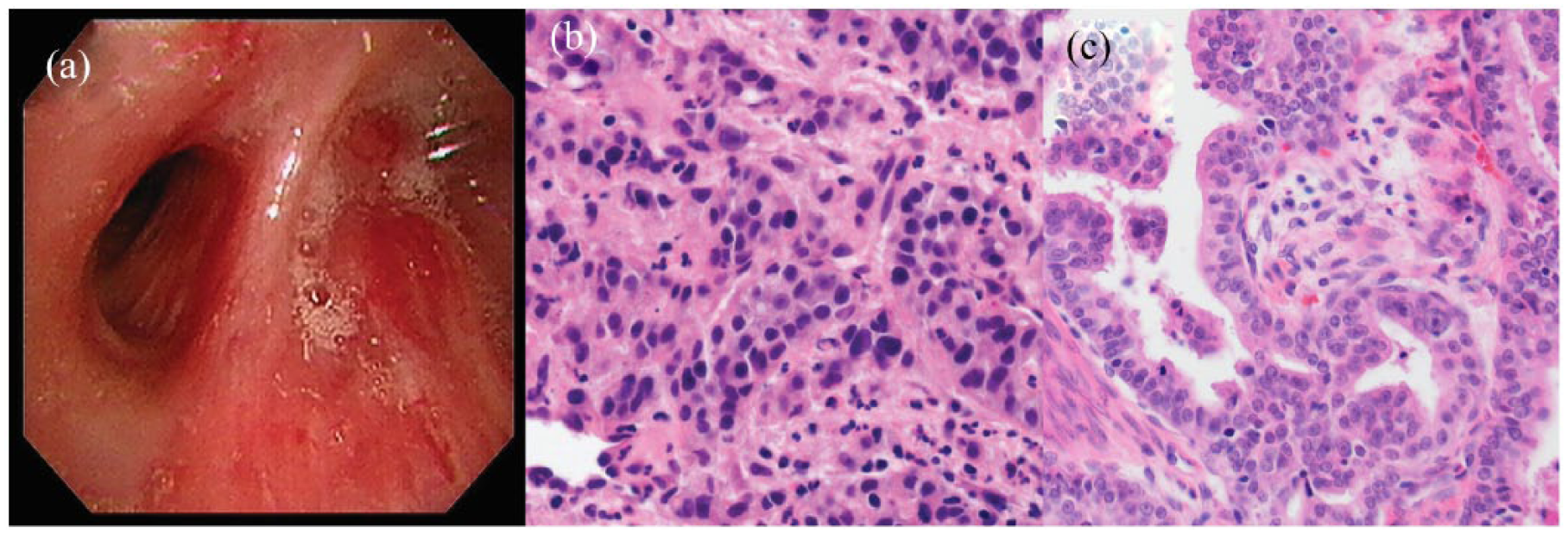
(a) Bronchoscopy showing an endobronchial mass occluding the right lower lobe bronchus. (b) A histological examination revealing atypical glandular cells similar to those of the endometrial carcinoma (hematoxylin and eosin staining, ×200). (c) Histology of the primary endometrial cancer showing pleomorphic tumor cells with prominent nuclear atypia (hematoxylin and eosin staining, ×200).
Discussion
A bronchoscopic biopsy confirmed an EBM from an endometrial carcinoma in this patient. There are typical and atypical metastatic sites of endometrial carcinomas, and pulmonary metastases are typical metastases of endometrial carcinomas, whereas EBMs are atypical metastases of that. 7 To the best of our knowledge, this is the first case report of an EBM from an endometrial carcinoma in Korea. Although one case was included in an investigation regarding an EBM for over 10 years, 6 this case differs from the previous reports in terms of the metastatic developmental types of EBMs.8,9 The developmental modes of EBMs were proposed as follows: type I, direct metastasis to the bronchus; type II, bronchial invasion by a lung parenchymal lesion; type III, bronchial invasion by a mediastinal or hilar lymph node metastasis; and type IV, peripheral lesions extending along the proximal bronchus. 3 The two previous cases with EBMs from endometrial carcinomas were types II and III, respectively.8,9 However, our case represented a type I, which was an endobronchial mass without a pulmonary metastasis nor mediastinal lymph node enlargement.
The mechanisms of EBMs have not been well established; however, lymphatic dissemination, a hematogenous spread, involvement from adjacent tumor tissue in the lung parenchyma or mediastinal lymph nodes and, rarely, aerogenous colonialization could be possible.3,10 It is difficult to prove the mechanisms of EBMs because there are a lot of things to consider, such as the histologic type of primary malignancy, biologic characteristics of the primary malignancy, anatomical structures, and vascular and lymphatic drains. Serous carcinomas, as in our case, are the most aggressive type of endometrial carcinomas, and vascular invasion is common. 11 This highly aggressive behavior of the primary malignancy could be a cause of the EBM. However, more research on EBM cases and further studies about the tumor biology are necessary to establish the mechanism of EBMs.
The usefulness of PET/CT in making a differential diagnosis between an endobronchial malignancy and benign stenosis in patients presenting with obstructive pneumonitis has already been reported.12,13 Chest CT can reveal accompanying pulmonary metastases or hilar-mediastinal lymphadenopathy, whereas CT is not always able to demonstrate luminal lesions. 1 In our case, PET/CT showed not only a hypermetabolic nodule in the RLL bronchus but also a diffusely decreased fludeoxyglucose uptake of the RLL. Clinically, it suggested that the RLL bronchus was obstructed by a malignant mass, and the metabolism of the RLL had decreased. This finding seemed to be a reflection of a decreased metabolism due to the EBM before any radiologically obvious atelectasis, and this could be an example of a functional image of PET. Notably, PET/CT is a helpful tool when an EBM without a pulmonary metastasis as in our case occurs.
The similarities in the clinical and radiological presentation between an EBM and primary lung cancer has been well described.1,2,10 Several patients may be totally asymptomatic and radiological findings are not particularly helpful in finding a differential diagnosis between endobronchial primary and metastatic tumors, even with secretion stasis. 4 As shown in our case, a bronchoscopic evaluation is an important tool for the differentiation between bronchogenic carcinomas and metastases. Furthermore, the identification of endobronchial histologic specimens together with previous histologic specimens from an extrathoracic primary tumor is necessary in order to reach a correct diagnosis.
There are variable treatment options for EBMs such as a surgical excision, local radiotherapy, chemotherapy, and interventional bronchoscopy including photodynamic therapy, electrocoagulation, and prosthetic stents.5,6 Since EBMs are a rare and heterogeneous condition and a randomized control trial is lacking, the treatment and management employed in EBMs should be individualized based on the primary tumor histology, anatomic location of the EBM, other metastases, and the patient’s performance status.1,14
Although the reports on EBMs from endometrial cancer are rare, this condition could become more frequent with the development of diagnostic imaging applications, which are clinically important in terms of airway patency issues. To discriminate primary lung cancers, a bronchoscopic biopsy is an important tool. In addition, a pathologic review with careful caution is necessary to achieve a correct diagnosis and an inter-department multi-disciplinary approach is required to make a proper therapeutic decision for the patients.
Conclusion
Although an EBM is rare, to make an accurate diagnosis, bronchoscopy plays a key role. Considering heterogeneous clinical conditions and various treatment modalities for endobronchial metastases, individualized treatment strategies are necessary.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2017 Yeungnam University Research Grant.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
